OPTIMIZING BIODIESEL PRODUCTION FROM RUBBER SEED OIL USING CONVENTIONAL METHOD
SARVIN RAM A/L SERIRAMULU
This report is submitted in partial fulfilment of the requirements for the award of the degree in Bachelor of Mechanical Engineering (Thermal-fluid)
Faculty of Mechanical Engineering University Teknikal Malaysia Melaka
i
SUPERVISOR DECLARATION
“I hereby declare that I have read this thesis and in my opinion this report is sufficient in terms of scope and quality for the award of the degree of
Bachelor of Mechanical Engineering (Thermal-Fluid)”
Signature : ……….
Supervisor : Pn. Nur Fathiah Binti Mohd Nor
ii
STUDENT’S DECLARATION
“I hereby declare that the work in this report is my own except for summaries and quotations which have been duly acknowledged.”
Signature : ………..
Author : Sarvin Ram a/l Seriramulu
iii
iv
ACKNOWLEDGEMENT
v
ABSTRACT
vi
ABSTRAK
vii
TABLE OF CONTENT
CHAPTER CONTENT PAGE
SUPERVISOR DECLARATION i
STUDENT DECLARATION ii
DEDICATION iii
ACKNOWLEDGEMENT iv
ABSTRACT v
ABSTRAK vi
TABLE OF CONTENTS vii
LIST OF TABLES x
LIST OF FIGURES xi
LIST OF SYMBOLS xiii
LIST OF ABBREVIATIONS xiv
CHAPTER 1 INTRODUCTION 1
1.1 Project Background 1
1.2 Problem Statement 3
1.3 Project Objective 4
viii
CHAPTER 2 LITERATURE REVIEW 5
2.1 Rubber Seed Oil 5
2.2 Basic Chemical Reactions 6
2.3 Biodiesel Production Processes 7
2.3.1 Raw Material Properties 7
2.3.1.1 Gas Chromatography (GC) 8
2.3.1.2 Acid Value and FFA 9
2.3.1.3 Saponification Number 9
2.3.1.4 Iodine Value (IV) 10
2.3.1.5 Kinematic Viscosity 10
2.3.2 Catalyst and Alcohol 11
2.3.3 Esterification Process 13
2.3.4 Mixing and Neutralization 13
2.3.5 Transesterification and Separation 14
2.4 Conventional Method 15
2.5 Properties of Biodiesel 16
2.6 Optimization of Biodiesel 17
2.6.1 Reaction Time 21
2.6.2 Methanol to Oil Ratio 21
2.6.3 Catalyst Weight Percentage 22
CHAPTER 3 METHODOLOGY 23
3.1 Introduction 23
3.2 Acid Value Experiment 23
3.3 Saponification Number Experiment 25
3.4 Iodine Value (IV) Experiment 25
3.5 Density Measurement 26
3.6 Kinematic Viscosity Experiment 27
ix
3.8 Transesterification Process 29
3.9 Flash Point Test 30
3.10 Minitab Software Analysis Process 32
3.11 Biodiesel Production and Optimization Flow Chart
39
CHAPTER 4 RESULTS AND DISCUSSION 40
4.1 Properties of Rubber Seed Oil 40
4.1.1 Gas Chromatography for Rubber Seed Oil
42
4.1.2 Average Molecular Weight of RSO 43
4.2 Acid Esterification and Transesterification of
Rubberseed Oil
44
4.3 Properties of Biodiesel from Rubber Seed Oil 45
4.4 Manipulated Parameters and Yield for Biodiesel 46
4.5 Analysis of Minitab Software 47
4.6 Main Effect and Interaction Plot for Yield 50
4.7 Relationship of Parameters and Response 53
4.7.1 Yield vs. Methanol, Catalyst 53
4.7.2 Yield vs. Time, Methanol 55
4.7.3 Yield vs. Time, Catalyst 56
4.8 Response Optimization for Yield 57
CHAPTER 5 CONCLUSION & RECOMMENDATIONS 59
x
LIST OF TABLES
NO. TITLE PAGE
2.1 Comparison between the types of catalyst 12
2.2 ASTM D6751 biodiesel standard 17
2.3 Structural Design of CCD for 3 factors 20
3.1 Density value of distilled water based on temperature 27
4.1 Properties of Rubber Seed Oil 40
4.2 Gas Chromatography Result for Rubber Seed Oil 42
4.3 Fatty Acid Molecular Weight 43
4.4 Comparison of Biodiesel Properties with ASTM Standard 45
4.5 Parameter Manipulation during Transesterification Process 46
4.6 Comparison between Experimental Yield and Theoretical
Yield
xi
LIST OF FIGURES
NO. TITLE PAGE
2.1 Schematic view of the chemical reaction during
transesterification
7
2.2 Process Flow Chart of a GC Machine 9
2.3 The example points of a Central Composite design with
three factors
18
3.1 Cool titration process 24
3.2 Brookfield Digital Viscometer model DV-II+ 28
3.3 Constant temperature electric heater with magnetic stirrer 30
3.4 Main components of flash point tester 31
3.5 Interface of Minitab Software for Create Response Surface
Design
32
3.6 Parameter Manipulation for Structural Design of CCD 33
3.7 Interface of Minitab Software for Analyze Response Surface
Design
34
3.8 Interface of Minitab Software for Yield Prediction 35
3.9 Interface of Minitab Software for Factorial Plots 36
3.10 Interface of Minitab Software for Graph Generation 37
3.11 Interface of Minitab Software for Response Optimizer 38
3.12 Flow Chart for Biodiesel Production 39
xii
4.2 Separation from transesterification process 44
4.3 Model Summary and Coded Coefficients 48
4.4 Graph for Normal Probability Plot 48
4.5 Regression Equation for Response Surface Model 49
4.6 Graph of Main Effects Plot for Yield 51
4.7 Graph of Interaction Plot for Yield 52
4.8 Surface Plot Graph of Yield vs. Catalyst, Methanol 53
4.9 Contour Plot Graph of Yield vs. Catalyst, Methanol 54
4.10 Surface Plot Graph of Yield vs. Time, Methanol 55
4.11 Contour Plot Graph of Yield vs. Time, Methanol 55
4.12 Surface Plot Graph of Yield vs. Time, Catalyst 56
4.13 Contour Plot Graph of Yield vs. Time, Catalyst 57
xiii
LIST OF SYMBOLS
OC = degree Celsius
g = grams
ml = milliliter
N = Normality
cm = centimeter
ρ = density
mg = milligram
g/mol = gram per mol
m2/s = meter squared per second
mm2/s = millimeter squared per second
mgKOH/g = milligram KOH per gram
kg/L = kilogram per liter
g Iodine/100g = gram Iodine per 100 gram
% = percent
xiv
LIST OF ABBREVIATIONS
FAME = Fatty Acid Methyl Esters
TAG = Triacylglycerol
RSO = Rubber Seed Oil
CaO = Calcium Oxide
FFAs = Free Fatty Acids
wt.% = Weight Percentage
RSM = Response Surface Methodology
AV = Acid Value
GC = Gas Chromatography
N2 = Nitrogen
H2 = Hydrogen
He = Helium
KOH = Potassium Hydroxide
IV = Iodine Value
I2 = Iodine
cST = Centistokes
RCOOR’ = Ester
H2SO4 = Sulphuric Acid
ASTM = American Society and Testing and Material
DMAIC = Define, Measure, Analyze, Improve, and Control
DOE = Design of Application
CCD = Central Composite Design
xv
R-sq = Regression squared
1
CHAPTER 1
INTRODUCTION
1.1 PROJECT BACKGROUND
The rapid increase in the use of the world’s crude oil reserve and diminishing of the natural source has accelerated the hunt for renewable energy. Renewable energy is energy source that use natural resources which have the potential to produce energy with minimum or zero emissions of both air pollutants and greenhouse gases. The predicted shortage of the crude oil encouraged the search for substitutes for petroleum derivatives which lead to an alternative fuel called “biodiesel”. Biodiesel has proven to be able to provide energy as per the requirement of the sectors such as agriculture, transportation, commercial and industrial sectors of the economy (Shafiee and Topal. 2009).
2
energy required during transesterification process by using catalyst. The non-edible vegetable oil used in this study is rubberseed oil (RSO)
The RSO has high free fatty acids (FFAs) content, which means that cockle based heterogeneous catalyst is desirable for usage. Heterogeneous base catalysts have pluses of being recycled, noncorrosive, have better tolerance to water and FFAs in feedstock, improve biodiesel yield and purity, a simpler purification process for glycerol and are easy to separate from the biodiesel product (Kawashima et al. 2008). The cockle is processed so the calcium oxide (CaO) can be extracted and used as catalyst. The use of biodiesel replacing the conventional diesel would slow the development of global warming by reducing the emissions of sulphur, hydrocarbon and carbon oxides. Because of economic benefits and more power output, biodiesel is often blended with diesel fuel in ratios of 2, 5 and 20% (Vasudevan and Briggs. 2008). In Malaysia B7 biodiesel was implemented where 7% of biodiesel is blended with 97% of pure diesel. The carbon dioxide emission can be minimized by increasing the ratio of biodiesel compared to diesel (Fukuda et al. 2001).
Optimizing is improvement on the performance of a system, a process, or a product so that the maximum value from the source can be obtained. The term optimization has been commonly used in analytical chemistry as a method of finding out circumstances at which to apply a practice that provides the best possible outcome (Bezerra et al. 2008). The optimization of analytical procedures has been carried out by using multivariate statistic technique which is response surface methodology (RSM). Response surface methodology is a collection of mathematical and statistical methods that depends on the fit of a polynomial equation to the experimental data, which must describe the behaviour of a data set with the objective of making statistical previsions. RSM can be well utilized when a response or a set of responses of interest are inclined by several variables.
3
from renewable and its sustainability. In future, biodiesel will be one of the leading energy sources for all sectors of the industry.
1.2 PROBLEM STATEMENT
4
1.3 PROJECT OBJECTIVE
This report is obliged according to the title of the project which is Optimizing Biodiesel Production from Rubberseed Oil using conventional method. Before conducting the project study, objectives of this study have to be defined clearly and specifically. This is very essential in order to carry out this study successfully. The objectives of this study are:
1. To produce biodiesel from rubber seed oil using waste heterogeneous catalyst (cockle) and conventional method.
2. To statically evaluate and optimize the yield of the biodiesel production using Response Surface Method (RSM).
3. To study the feedstock characteristics such as FFAs and fatty acid composition
that may influence the final properties of the biodiesel.
1.4 PROJECT SCOPE
The scope of the study is defined so that a specified study can be conducted and the study does not go off the track of the study. Scope also ensures that the study is parallel with the objective. The scope of this study is:
1. The raw material used to produce biodiesel is Rubber seed oil.
2. The biodiesel production involves only Acid Esterification and Base Catalysed
Transesterification process.
3. Heating equipment used for Acid Esterification and Base Catalysed
Transesterification process is Constant temperature electric heater with magnetic stirrer.
4. The type of base catalyst used in Base Catalysed Transesterification process is
5
CHAPTER 2
LITERATURE REVIEW
2.1 RUBBER SEED OIL
6
2.2 BASIC CHEMICAL REACTIONS
7
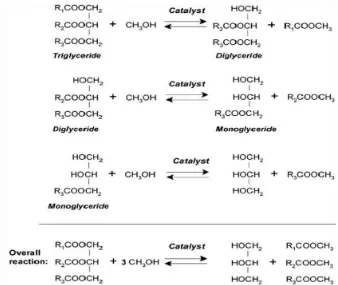
Figure 2.1: Schematic view of the chemical reaction during transesterification (Ngamcharussrivichai, C. et al., 2010)
2.3. BIODIESEL PRODUCTION PROCESSES
2.3.1 Raw Material properties
8
properties of the produced biodiesel. Section 2.3.1.1 until 2.3.1.5 explains in detail regarding the chemical properties of the rubber seed oil.
2.3.1.1 Gas Chromatography (GC)
GC is a common analytic method used in many research fields as identification and quantitation equipment for compounds in a mixture. The reason that GC is more favourable technique is because the detection of compound with very small quantities is possible with GC. A small amount sample of the mixture that is being analysed will be injected into the GC machine via a syringe. The mixture usually brought into the GC machine in liquid form where at latter stage in the column oven the components of the mixture are heated and instantly vaporize. Before the mixture is heated, a carrier gas is injected to the mixture to be tested. The carrier gas only serves a sole purpose which is to help the gases in mixture to move through the column after heated and vaporized. The
most common carrier gases are nitrogen gas (N2), hydrogen gas (H2), and helium gas