Atherosclerosis 150 (2000) 443 – 444
Letter to the Editor
www.elsevier.com/locate/atherosclerosis
Upregulation of the interleukin-8 system in hypercholes-terolemic patients. Does inhibition of the mevalonate pathway lower interleukin-8 levels in the vessel wall?
Recently, Porreca et al. [1] reported a comprehensive study about peripheral blood mononuclear cell (PBMC) production of interleukin-8 (IL-8) and IL-8-dependent neutrophil function in hypercholesterolemic patients. The authors found an increase in the amount of IL-8 transcript in PBMC from hypercholesterolemic patients, in non-stimulated and in lipopolysaccharide (LPS) stimulated cultures, respectively. Correspond-ingly, a significant increase of IL-8 immunoactivity in the conditioned medium of PBMC from hypercholes-terolemic subjects compared with controls was found. As the authors state, these results indicate an upregula-tion of the IL-8 system in dyslipidemic patients and provide further evidence for ongoing in vivo IL-8-de-pendent activation of inflammatory cells during hyperc-holesterolemia. Other groups reported that oxidized LDL and acetylated LDL were capable of inducing IL-8 synthesis in THP-1 macrophages, whereas native LDL had no impact on the production of the cytokine [2]. Furthermore, macrophages isolated from athero-sclerotic plaques have been found to produce IL-8 [3]. In addition to its initial characterization as a proinflam-matory cytokine, IL-8 has been shown to be synthe-sized by a variety of cell types and accordingly, to have a variety of functions. With regard to the constituents of the vessel wall, IL-8 plays an important role as a mitogen and chemoattractant for vascular smooth mus-cle cells (SMC) [4]. SMC migration can be stimulated by IL-8 to values 20-fold over those of controls. Since SMC proliferation and migration are crucial steps in the development of vessel wall thickening during atherosclerosis, these data raise the importance of IL-8 for the pathologic changes of the vessel wall.
As Porreca et al. [1] reported, increased levels of cholesterol are linked to an upregulation of the IL-8 system, which in turn may explain leukocyte activation during hypercholesterolemia and cause the inflamma-tory response during atherosclerosis. The authors also point out that inhibition of 3-hydroxy-3-methylglutaryl coenzyme A (HMG-CoA) reductase is an important means to inhibit atherosclerotic disease. In a rabbit
atherosclerosis model, atorvastatin has been shown to reduce the neointimal inflammation, which could con-tribute to the stabilization of the atherosclerotic plaque [5]. However, it is widely unknown by which pathways HMG-CoA reductase inhibitors affect the pathogenesis of atherosclerosis apart from simply lowering the cholesterol. The interesting question now arises, whether the lowering of cholesterol levels is followed by a decrease of IL-8 levels. Monocytic THP-1 cells have been found to produce markedly decreased IL-8 levels following mitogenic stimulation when the cells were pretreated with HMG-CoA reductase inhibitors such as lovastatin or compactin at concentrations of 10 mmol/l
[6]. In contrast, another group found that stimulated or non-stimulated IL-8 synthesis of PBMC is not affected by inhibition of the mevalonate pathway using simvas-tatin at doses up to 10 mmol/l [7]. However, IL-8
synthesis by blood cells does not directly reflect the IL-8 content in the vessel wall. Studies with Ca2+
channel blockers on SMC revealed increased levels of IL-8 due to the treatment with the compounds [8]. Among a variety of effects, Ca2+ channel blockers
have been shown to downregulate the HMG-CoA re-ductase gene in fibroblasts [9]. In view of the unclear action of HMG-CoA reductase inhibitors on IL-8 ex-pression, we studied the influence of lovastatin and simvastatin on IL-8 synthesis in human coronary SMC (hcSMC) under in vitro conditions by applying 5 – 20
mmol/l of the compounds — doses comparable to those
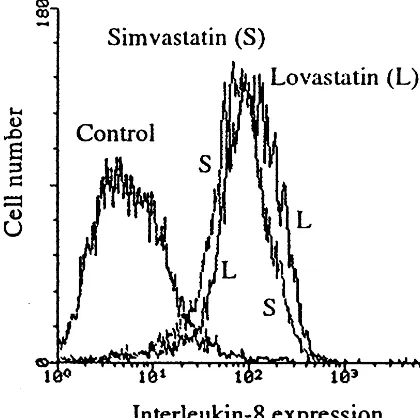
used in the above mentioned studies on blood cells — for an incubation time of 24 – 48 h. Intracellular IL-8 levels were determined by flow cytometry employing cell preparation methods as described previously [10] using a monoclonal antibody directed against recombi-nant human IL-8 (Endogen, Cambridge, MA). In con-trast to the decreased production of IL-8 in human THP-1 monocytes under HMG-CoA reductase in-hibitor treatment, we observed a marked increase of intracellular IL-8 levels up to 23-fold (relative fluores-cence intensity) due to incubation with lovastatin or simvastatin (Fig. 1). Since HMG-CoA reductase in-hibitors are known antiproliferative compounds, it may be discussed whether upregulation of IL-8 could be an autocrine production of a mitogen to overcome the cell
Letter to the Editor 444
Fig. 1. Flow cytometric analysis of intracellular IL-8 levels in hcSMC. The cells were cultured to subconfluency in the presence of DMEM media containing 10% FCS. Simvastatin (5mmol/l) or lovastatin (5 mmol/l) were then added to the culture media and incubated for 48 h. Cells were fixed and perforated using 1% formaldehyde (5 min) following ice-cold pure methanol (30 min) and subsequent treatment with 0.1% Triton X-100 and 0.1% sodium citrate in phosphate buffered saline on ice (45 min), before IL-8 was indirectly stained using a monoclonal antibody specific for recombinant human IL-8. The data shown were generated in the same experiment.
References
[1] Porreca E, Sergi R, Baccante G, Reale M, Orsine L, Di Febbo C, Caselli G, Cuccurullo F, Bertini R. Peripheral blood mononu-clear cell production of interleukin-8 and IL-8 dependent neu-trophil function in hypercholesterolemic patients. Atherosclerosis 1999;146:345 – 50.
[2] Wang N, Tabas I, Winchester R, Ravalli S, Rabbani LE, Tall A. Interleukin 8 is induced by cholesterol loading of macrophages and expressed by macrophage foam cells in human atheroma. J Biol Chem 1996;271:8837 – 42.
[3] Liu Y, Hulten LM, Wiklund O. Macrophages isolated from human atherosclerotic plaques produce IL-8, and oxysterols may have a regulatory function for IL-8 production. Arterioscler Thromb Vasc Biol 1997;17:317 – 23.
[4] Yue TL, Wang X, Sung CP, Olson B, McKenna PJ, Gu JL, Feuerstein GZ. Interleukin-8. A mitogen and chemoattractant for vascular smooth muscle cells. Circ Res 1994;75:1 – 7. [5] Bustos C, Hernandez-Presa MA, Ortego M, Tunon J, Ortega L,
Perez F, Diaz C, Hernandez G, Egidio J. HMG-CoA reductase inhibition by atorvastatin reduces neointimal inflammation in a rabbit model of alherosclerosis. J Am Coll Cardiol 1998;32:2057 – 64.
[6] Terkeltaub F, Solan J, Barry M Jr, Santoro D, Bokoch GM. Role of the mevalonate pathway of isoprenoid synthesis in IL-8 generation by activated monocytic cells. J Leukocyte Biol 1994;55:749 – 55.
[7] Fukuo Y, Nagashima M, Soya-Ohmura N, Takagi S, Terashi A, Ikejima T. Effects of an HMG-CoA reductase inhibitor on cytokine production by human monocytes/macrophages. Nippon Ika Daigaku Zasshi 1995;62:386 – 90.
[8] Rodler S, Roth M, Nauk M, Tamm M, Block LH. Ca(2 +)-channel blockers modulate the expression of interleukin-6 and interleukin-8 genes in human vascular smooth muscle cells. J Mol Cell Cardiol 1995;27:2295 – 302.
[9] Block LH, Matthys H, Emmons LR, Perruchoud A, Erne P, Roth M. Ca(2+)-channel blockers modulate expression of 3-hy-droxy-3-methylglutaryl-coenzyme A reductase and low density lipoprotein receptor genes stimulated by platelet-derived growth factor. Proc Natl Acad Sci USA 1991;88:9041 – 5.
[10] Sindermann J, Weigel KA, Breithardt G. A simple method for the flow cytometric analysis of intracellular antigens in whole smooth muscle cells. Quantification of cyclin-dependent kinase 2. J Immunol Methods 1997;202:205 – 12.
18 October 1999
12 January 2000 Ju¨rgen R. Sindermanna,b, Annette Schmidta,
Gu¨nter Breithardta,b a
Di6ision of Molecular Cardiology,
Institute for Arteriosclerosis Research,
Uni6ersity of Mu¨nster,
48149 Mu¨nster, Germany bDepartment of Cardiology and Angiology, Uni6ersity of Mu¨nster Medical School,
Domagkstraße 3,
48149 Mu¨nster,
Germany
cycle arrest. However, our studies on another com-pound known to be capable of inhibiting SMC-prolifer-ation such as paclitaxel revealed no substantial modulations of IL-8 levels. Taking the fact into account that IL-8 is released from cells, an increased immunore-activity of IL-8 in whole cells may be explained by a different distribution of the cytokine from intracellular to a more membrane located form. Therefore, we also treated hcSMC with 2.5mmol/l monensin for 6 h in the
presence of lovastatin or simvastatin in order to prevent IL-8 being released from the cells. Even under these conditions we found an increase of IL-8 in hcSMC.
Despite the known upregulation of IL-8 during hy-percholesterolemia [1], it remains unclear whether the inhibition of HMG-CoA reductase causes a downregu-lation of IL-8 among various cell types. Our data may indicate an upregulation of the IL-8 system by inhibit-ing the mevalonate pathway in SMC. Whether com-parable results can be found under in vivo conditions in the vessel wall remains to be shown. If so, the known clinical benefits of HMG-CoA reductase inhibitors could not be explained by primarily lowering the proinflammatory cytokine IL-8 in the muscular com-partment of the vessel wall.