Soil carbon, microbial activity and nitrogen availability in
agroforestry systems on moderately alkaline soils in northern India
B. Kaur
a, S.R. Gupta
a,∗, G. Singh
baDepartment of Botany, Kurukshetra University, Kurukshetra 136119, India bCentral Soil Salinity Research Institute, Karnal, India
Received 6 November 1998; received in revised form 28 April 2000; accepted 28 April 2000
Abstract
The present investigation aimed to analyze the role of agroforestry systems in improving soil organic matter status, microbial activity and nitrogen availability with a view to effective management of the fertility of moderately alkaline soils. The study
site was located at Karnal (29◦
59′
N, 76◦
51′
E, 250 m.s.l.) and the systems were characterized by a rice–berseem crop rotation; agrisilvicultural systems of Acacia, Eucalyptus and Populus along with rice–berseem and single species tree plantations. Soil microbial biomass was measured using the fumigation extraction technique and nitrogen mineralization using the aerobic
incubation method. Microbial biomass carbon was low in rice–berseem crops (96.14mg g−1soil) and increased in soils under
tree plantations (109.12–143.40mg g−1soil) and agrisilvicultural systems (133.80–153.40mg g−1soil). Microbial biomass
was higher by 42% (microbial C) and 13% (microbial N) in tree-based systems as compared to monocropping. Microbial biomass immobilized 2.32–2.57% of the soil carbon and 4.08–4.48% of the soil nitrogen in tree-based systems. Soil carbon increased by 11–52% due to integration of trees along with the crops for 6–7 years. Cropland management practices and tree
species influenced CO2-C production, biomass specific respiratory activity, and nitrogen mineralization rates. In tree-based
systems, soil inorganic N levels were higher by 8–74% and nitrogen mineralization by 12–37% as compared to monocropping. On the basis of increased soil organic matter content, enlarged soil microbial biomass pool and greater soil N availability, agrisilvicultural systems have been found to be ecologically sustainable land-use systems for utilizing moderately alkaline soils. © 2000 Elsevier Science B.V. All rights reserved.
Keywords: Carbon dioxide; Microbial biomass; Microbial activity; Nitrogen mineralization; Soil carbon; Soil nitrogen
1. Introduction
Saline and alkaline soils are of widespread oc-currence in arid and semiarid regions of northern India. More than 2.5 million ha of otherwise arable lands in the Indo-Gangetic plains have become un-suitable for cultivation due to soil sodicity (Abrol
∗Corresponding address. Tel.:+91-1744-20196(501); fax:+91-1744-20277.
E-mail address: [email protected] (S.R. Gupta).
and Bhumbla, 1971). These soils are character-ized by high pH throughout the soil profile, high exchangeable sodium and low soil organic mat-ter content (Gupta et al., 1984), a sparse cover of natural vegetation (Rana and Parkash, 1987) and low microbial activity (Kaur et al., 1998; Pathak and Rao, 1998). The productive capacity of al-kaline soils has been found to improve by grow-ing plants adapted to sodic soils (Gupta et al., 1990). Reclamation agroforestry systems have been reported to improve biological production and
ameliorate sodic conditions of soils by increas-ing soil organic matter content and availability of soil inorganic nitrogen (Singh, 1995; Singh et al., 1997).
The soil microbial biomass is a labile pool of organic matter and comprises 1–3% of total soil or-ganic matter (Jenkinson and Ladd, 1981). The soil microbial biomass acts as a source and sink of the plant nutrients (Singh et al., 1989; Smith and Paul, 1990) and regulates the functioning of the soil sys-tem. Plant cover through its effects on the quantity and quality of organic matter inputs influences the levels of soil microbial biomass (Wardle, 1992). The specific respiratory activity of soil microbial biomass has been used to analyze the effects of en-vironmental factors, crop management, and organic inputs on the microbial populations (Anderson and Domsch, 1990, 1993; Campbell et al., 1991). It is sensitive to the changes in the quantity and quality of soil organic matter and ecosystem stability (Insam, 1990).
In saline and alkaline soils, excessive amounts of salts have an adverse effect on biological activity including soil enzyme activity (Frankenberger and Bingham, 1982; Rao and Pathak, 1996), nitrogen min-eralization (McClung and Frankenberger, 1985) and soil microbial biomass (Sarig and Steinberger, 1994; Batra and Manna, 1997; Kaur et al., 1998). The bio-logical activity of alkaline soils has been found to im-prove under a crop, grass or tree cover (Rao and Ghai, 1985). Nitrogen mineralization is an essential func-tion of the soil microbial system (Ellenberg, 1971). Pathak and Rao (1998) reported that added organic matter has a favorable effect on nitrogen mineraliza-tion rate in saline and alkaline soils under laboratory conditions. In Acacia, Eucalyptus and Populus based agroforestry systems, Singh et al. (1997) reported an increase in soil organic carbon and soil available nitrogen.
Soil biological activity in relation to plant growth and land-use practices is poorly understood for salt-affected soils. The present investigation aims to analyze the effects of cropping, forestry and agroforestry on soil organic carbon, total nitrogen, soil microbial biomass and net nitrogen mineraliza-tion and to consider implicamineraliza-tions for the long-term management of the fertility of moderately alkaline soils.
2. Materials and methods
2.1. Study site
The agroforestry systems selected for study were on the Central Soil Salinity Research Institute experi-mental farm at Karnal (29◦59 N′, 76◦51′E, 250 m.s.l.). The climate of the study area is semiarid and mon-soonic and characterized by hot dry summers and cold winters. The mean annual rainfall of the area is about 700 mm, about 80% of which is received dur-ing July and September. Geologically the area consti-tutes a part of the Indo-Gangetic alluvial plains and belongs to the Pleistocene age. The soils are classi-fied as Haplic-Salonetz, very strongly alkaline, loam to clay loam in A and B horizons. They are sandy loam in texture in the surface 0–5 cm layer and clay loam at 5–85 cm. Initially the soils of the study site were highly sodic with moderately low permeability and had a sparse cover of salt-tolerant grasses.
The present study is part of a long-term field ex-periment initiated in 1989 at the exex-perimental farm of the CSSRI, Karnal. The treatments in this experiment were three tree species, viz., Acacia nilotica Delile sub sp. Indica (Benth.) Brenan, Eucalyptus tereticornis Smith, Populus deltoides Bartr. Ex Marsh, in the main plots and four inter-crop treatments, viz., rice–wheat (Triticum aestivum), rice–berseem (Trifolium alexan-drium), pigeonpea (Cajanus cajan)–mustard (Brassica juncea) and no crop in the sub-plots. The 12 treatment combinations were replicated three times in a spilt plot design. Soil carbon for various treatments varied from 0.22–0.28% and the soil pH was 9.26–9.34.
Saplings of Eucalyptus, Acacia and Populus were planted during September 1989 and February 1990 in augerholes. These augerholes were filled with the original soil mixed with gypsum (3 kg) and farmyard manure (8 kg). The size of plots was 12 m×12 m and the distance between rows of trees was 4 m and be-tween trees in a row 2 m from 1989 to 1995. Inter-row space was also amended with gypsum before the first inter crop was planted in the summer of 1990.
1997). The rice crop was planted using nursery-raised 20 days old seedlings on 28 June 1995 and harvested on 12 October 1995. Nitrogen fertilizer was applied at the rate of 150 kg N ha−1and ZnSO
4was added at
the rate of 50 kg ha−1during the crop growing season.
After preparing the field, berseem was sown on 25 October 1995 with a basal dose of 25 kg N ha−1. Green
herbage of berseem was cut during the last weeks of December, February and March and finally the mature crop was harvested on 29 May 1996. The inter-crops without trees were also grown in a separate field adja-cent to the main experiment with similar management and agronomic practices. In this area, the intercrop sequences were replicated three times. An unweeded control (fallow for 5 years) was also maintained.
In the present study, soil samples were taken from only one inter-crop treatment of the experiment, i.e. rice–berseem. Therefore, the treatments compared in this study were: rice–berseem cropping, rice–berseem with tree species of A. nilotica, E. tereticornis, P. deltoides; Acacia, Eucalyptus and Populus without rice–berseem, fallow with naturally growing plants as a control. At the start of this study during 1995, the soil pH (1:2) varied from 8.72 to 9.17. The soil or-ganic carbon content in different treatment plots var-ied from 0.42 to 0.68% and total soil nitrogen ranged from 0.046 to 0.086%.
2.2. Soil sampling and preparation
To analyze the effects of tree and crop species on soil organic matter, soil microbial biomass and nitrogen mineralization, soil samples were collected during July and December 1995 and March 1996 from the various treatment plots. The soil was sam-pled (0–10 cm depth) using a soil corer (12 cm diam.). For obtaining a representative sample five soil cores were collected along a transect for each of the three replicates of the eight treatments. Soil samples for the three replicates from the respective treatments were also composited for analysing soil carbon and nitro-gen, microbial biomass and nitrogen mineralization.
During May 1996, the effect of soil depth and hor-izontal spatial variability (distance from the rows of trees) was also studied on soil microbial activity and nitrogen mineralization. After removing the ground floor litter and plant residues, the underlying soil was sampled using a soil corer (12 cm diam.) at depth
intervals of 0–7.5, 7.5–15, and 15–30 cm to obtain three pooled samples at 1, 2 and 3 m distance from the trees within the alleys. A total of three compos-ite samples for each depth and distance from the trees was obtained. Samples were transported to the laboratory in polyethylene bags and stored at 4◦C until analysis. The soil was pre-conditioned before measuring soil microbial biomass by spreading it overnight in a thin layer within folded polyethylene sheets, with moisture content adjusted to 40% of wa-ter holding capacity. The measurement of microbial biomass carbon and nitrogen was completed within a few days of sieving the soil. Subsamples of air dried soil were used for determining soil pH (1:2), organic carbon (Kalembasa and Jenkinson, 1973), and total nitrogen (Bremner, 1965). Soil bulk density was also determined under field conditions.
2.3. Microbial biomass
Microbial biomass C and N were determined us-ing the fumigation extraction methods (Brookes et al., 1985; Vance et al., 1987). The sieved, field moist soil sub-samples (equivalent to 50 g oven dry soil) were fumigated with alcohol free chloroform in vacuum desiccators and stored in the dark for 24 h. After re-moving the fumigant (by repeated de-evacuation of chloroform from the soils), the samples were extracted with 200 ml 0.5 M K2SO4 for 30 min on a shaking
machine. The unfumigated soil samples were extracted similarly at the start of experiment.
The filtered soil extracts of both fumigated and un-fumigated samples were analyzed for organic C using the acid dichromate method (Vance et al., 1987). To-tal nitrogen in K2SO4soil extract was determined by
acid digestion and Kjeldahl distillation (Brookes et al., 1985). Soil microbial biomass carbon (MBC) was es-timated as BC=2.64 EC (Vance et al., 1987), where EC (extractable carbon) is the difference between car-bon extracted from fumigated and unfumigated sam-ples, both expressed in the same measurement unit. Soil microbial biomass nitrogen (MBN) was estimated as BN=EN/0.54 (Brookes et al., 1985), where EN (extractable nitrogen) is the difference between N ex-tracted from fumigated and unfumigated samples.
incubated in 2 l air tight jars at room temperature (27–35◦C) for 30 days. The CO
2produced from the
soil was absorbed in 0.5 N NaOH and determined titri-metrically with 0.1 N HCl using phenolphthalein as indicator (Gupta and Singh, 1981). The CO2evolution
rates were determined at an interval of 2–5 days. Car-bon mineralization rates were determined from CO2-C
evolved from the soil samples. Specific respiratory ac-tivity of soil microbial biomass carbon was estimated by dividing the CO2-C produced (i.e. mineralizable
carbon) by the value of soil microbial biomass fol-lowing Campbell et al. (1991). Mineralizable carbon was estimated from the quantity of CO2-C produced
during 10 days incubation of the soil under laboratory conditions.
2.4. Soil inorganic N and nitrogen mineralization rates
Inorganic N levels were determined using the Kjeldahl distillation method. Sieved, moist soil sub-samples (equivalent to 50 g oven dry weight) were extracted with 0.5 M K2SO4(4 K2SO4: 1 soil).
Inorganic N levels were determined in the soil fil-trate by MgO and Devardas alloy distillation. Similar sub-samples of soil were incubated under laboratory conditions at ambient temperature. After periods of 10, 20 and 40 days the incubated soil samples were extracted with 0.5 M K2SO4. Net nitrogen
mineral-ization rates were determined by subtracting initial soil mineral N from final mineral N at 40 days.
2.5. Statistics
Data on soil carbon, nitrogen, microbial biomass and nitrogen mineralization rates were analyzed using one way analysis of variance (ANOVA). Least signif-icant difference (LSD) values at the 5% levels of sig-nificance (p<0.05) and Duncan’s Multiple Range Test values (DMRT) were calculated following Gomez and Gomez (1984).
3. Results
3.1. Soil carbon and nitrogen
Variations in soil carbon and nitrogen with soil depth and horizontal spatial variability (distance from
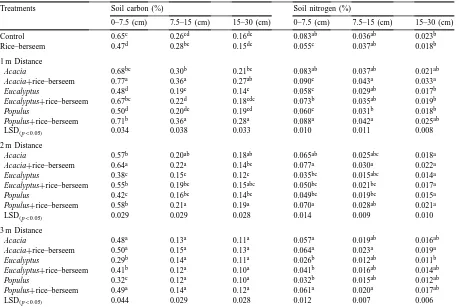
rows of trees) are shown in Table 1. Soil carbon and nitrogen concentration at 0–7.5 cm depth were higher than at 7.5–15 and 15–30 cm. Soil carbon and nitrogen concentration varied significantly with soil depth (p<0.01) and distance from the trees (p<0.01) in all the treatments. Organic carbon and nitrogen accretion in the soil was significantly higher when trees were associated with agricultural crops. Max-imum build up of organic carbon in soil occurred under Acacia based systems followed by Populus and Eucalyptus based systems. The nitrogen content increased by 8–46% (0–7.5 cm soil depth) due to integration of trees with crops; the increase being maximum in the case of Acacia based systems. Av-eraged across the distance from the trees at 0–7.5 cm soil depth, carbon and nitrogen concentrations were found to be higher in the case of Acacia based sys-tems (0.57–0.63% C; 0.068–0.077% N) and Populus based systems (0.41–0.59% C; 0.047–0.073% N) as compared to Eucalyptus based systems (0.38–0.54% C; 0.039–0.054% N). It is evident that the ameliora-tive effect of trees was more conspicuous at 1 and 2 m distance from trees and in upper soil layers (0–7.5 cm soil depth). There were no significant differences due to tree species in organic carbon and total nitrogen at 3 m distance from the trees (Table 1).
3.2. Microbial biomass
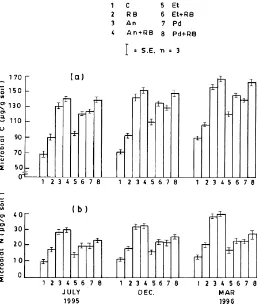
The levels of soil microbial biomass carbon and nitrogen in rice–berseem cropping, tree plantations and agroforestry systems during July 1995–March 1996 are shown in Fig. 1. Soil microbial biomass C (90.56–168.0mg g−1 soil) and biomass N (13.16–
35.59mg N g−1 soil) were maximum during the
Table 1
Soil carbon and nitrogen concentrations at different soil depths and distances from rows of tree (1, 2 and 3 m) in different systemsa
Treatments Soil carbon (%) Soil nitrogen (%)
0–7.5 (cm) 7.5–15 (cm) 15–30 (cm) 0–7.5 (cm) 7.5–15 (cm) 15–30 (cm)
Control 0.65c 0.26cd 0.16de 0.083ab 0.036ab 0.023b
Rice–berseem 0.47d 0.28bc 0.15de 0.055c 0.037ab 0.018b
1 m Distance
Acacia 0.68bc 0.30b 0.21bc 0.083ab 0.037ab 0.021ab
Acacia+rice–berseem 0.77a 0.36a 0.27ab 0.090c 0.043a 0.033a
Eucalyptus 0.48d 0.19e 0.14e 0.058c 0.029ab 0.017b
Eucalyptus+rice–berseem 0.67bc 0.22d 0.18cde 0.073b 0.035ab 0.019b
Populus 0.50d 0.20de 0.19cd 0.060c 0.031b 0.018b
Populus+rice–berseem 0.71b 0.36a 0.28a 0.088a 0.042a 0.025ab
LSD(p<0.05) 0.034 0.038 0.033 0.010 0.011 0.008
2 m Distance
Acacia 0.57b 0.20ab 0.18ab 0.065ab 0.025abc 0.018a
Acacia+rice–berseem 0.64a 0.22a 0.14bc 0.077a 0.030a 0.022a
Eucalyptus 0.38c 0.15c 0.12e 0.035bc 0.015abc 0.014a
Eucalyptus+rice–berseem 0.55b 0.19bc 0.15abc 0.050bc 0.021bc 0.017a
Populus 0.42c 0.16bc 0.14bc 0.049bc 0.019bc 0.015a
Populus+rice–berseem 0.58b 0.21a 0.19a 0.070a 0.028ab 0.021a
LSD(p<0.05) 0.029 0.029 0.028 0.014 0.009 0.010
3 m Distance
Acacia 0.48a 0.13a 0.11a 0.057a 0.019ab 0.016ab
Acacia+rice–berseem 0.50a 0.15a 0.13a 0.064a 0.023a 0.019a
Eucalyptus 0.29b 0.14a 0.11a 0.026b 0.012ab 0.011b
Eucalyptus+rice–berseem 0.41b 0.12a 0.10a 0.041b 0.016ab 0.014ab
Populus 0.32c 0.12a 0.10a 0.032b 0.015ab 0.012ab
Populus+rice–berseem 0.49a 0.14a 0.12a 0.061a 0.020a 0.017ab
LSD(p<0.05) 0.044 0.029 0.028 0.012 0.007 0.006
aFor each column, values not marked with the same letter are significantly different (DMRT; p<0.05).
Averaged across the sampling dates, microbial biomass C and N varied from 76.1 to 153.4mg C g−1
soil and 11.1 to 32.6mg N g−1 soil, respectively
(Table 2). The tree species had a significant effect on the levels of microbial carbon and nitrogen. The
Table 2
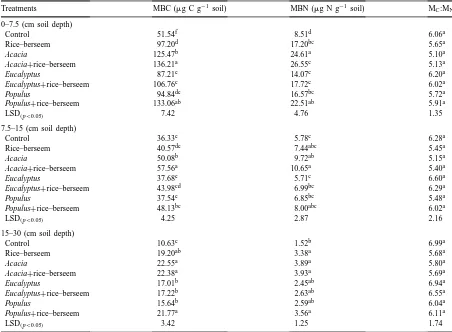
Soil microbial biomass carbon (MBC) and microbial biomass nitrogen (MBN) averaged across the seasons and their relationships with total soil carbon and nitrogen in different management systemsa
Treatments MBC (mg g−1 soil) MBN (mg g−1soil) Microbial C:N MBC Soil C (%) MBN Soil N (%)
Control 76.13f 11.06e 6.88a 1.21c 1.51d
Rice–berseem 96.14e 18.61cd 5.16cd 2.29cd 4.04ab
Acacia 143.40b 31.45a 4.56d 2.31b 4.08ab
Acacia+rice–berseem 153.40a 32.57a 4.71d 2.26d 3.79bc
Eucalyptus 109.12d 15.78de 6.92a 2.32ab 3.36c
Eucalyptus+rice–berseem 133.80c 21.78bc 6.14ab 2.23b 3.69bc
Populus 131.10c 21.70c 6.04bc 2.57a 4.48a
Populus+rice–berseem 150.13a 25.62b 5.86bc 2.24b 3.28c
LSD(p<0.05) 4.86 3.38 0.731 0.194 0.525
aFor each column, values not marked with the same letter are significantly different (DMRT; p<0.05).
Fig. 1. Seasonal variation in soil microbial biomass Carbon (a) and Nitrogen (b) in different treatments (C=control; RB=rice–berseem; An=Acacia nilotica; An+RB=Acacia nilotica+rice–berseem; Et=Eucalyptus tereticornis; Et+RB=Eucalyptus tereticornis+rice–berseem; Pd=Populus deltoides; Pd+RB=Populus deltoides+rice–berseem).
42% (microbial C) and 13% (microbial N) in the case of tree-based systems as compared to monocropping. The ratio of microbial C:microbial N varied from 6.92 to 5.86 in Eucalyptus and Populus based systems, whereas in Acacia based systems the microbial C:N was in the range of 4.56–4.71.
The proportions of microbial carbon and nitrogen to total soil C and N were higher in the case of mono-cultures indicating higher C and N immobilization in microbial biomass (Table 2). The ratio of microbial C to total soil carbon was comparatively higher in mono-cultures of Eucalyptus and Populus (2.32–2.57%). The proportion of microbial N to total soil N varied from 4.08 to 4.48% in Acacia and Populus plantation.
The effect of soil depth on biomass carbon was sig-nificant, reflecting the steep decline in soil organic matter content in sub-surface layers of soil (Table 3).
Distance from trees also had a significant effect on mi-crobial biomass C and N at 0–7.5 cm and 7.5–15 cm soil depths (p<0.01). Significantly higher amounts of microbial biomass carbon and nitrogen were found in soil at 1 m from the trees than at 2 and 3 m. Aver-aged across the distance from the trees, the microbial biomass carbon and nitrogen were found to be higher under systems with Acacia (125 –136mg C g−1 soil;
25–27mg N g−1 soil) and Populus (95–133mg C g−1
soil; 17–23mg N g−1 soil) (Table 3). In general, the
microbial biomass carbon was significantly higher in agroforestry as compared to tree plantations. Micro-bial biomass nitrogen did not differ significantly be-tween soils under trees alone and agroforestry systems (Table 3).
Table 3
Variations in soil microbial biomass carbon (MBC) and microbial biomass nitrogen (MBN) with soil depths in different treatmentsa
Treatments MBC (mg C g−1 soil) MBN (mg N g−1 soil) MC:MN
0–7.5 (cm soil depth)
Control 51.54f 8.51d 6.06a
Rice–berseem 97.20d 17.20bc 5.65a
Acacia 125.47b 24.61a 5.10a
Acacia+rice–berseem 136.21a 26.55c 5.13a
Eucalyptus 87.21e 14.07c 6.20a
Eucalyptus+rice–berseem 106.76c 17.72c 6.02a
Populus 94.84de 16.57bc 5.72a
Populus+rice–berseem 133.06ab 22.51ab 5.91a
LSD(p<0.05) 7.42 4.76 1.35
7.5–15 (cm soil depth)
Control 36.33e 5.78c 6.28a
Rice–berseem 40.57de 7.44abc 5.45a
Acacia 50.08b 9.72ab 5.15a
Acacia+rice–berseem 57.56a 10.65a 5.40a
Eucalyptus 37.68e 5.71c 6.60a
Eucalyptus+rice–berseem 43.98cd 6.99bc 6.29a
Populus 37.54e 6.85bc 5.48a
Populus+rice–berseem 48.13bc 8.00abc 6.02a
LSD(p<0.05) 4.25 2.87 2.16
15–30 (cm soil depth)
Control 10.63c 1.52b 6.99a
Rice–berseem 19.20ab 3.38a 5.68a
Acacia 22.55a 3.89a 5.80a
Acacia+rice–berseem 22.38a 3.93a 5.69a
Eucalyptus 17.01b 2.45ab 6.94a
Eucalyptus+rice–berseem 17.22b 2.63ab 6.55a
Populus 15.64b 2.59ab 6.04a
Populus+rice–berseem 21.77a 3.56a 6.11a
LSD(p<0.05) 3.42 1.25 1.74
aFor each column, values not marked with same letter are significantly different (DMRT; p<0.05).
microbial carbon to total soil carbon varied from 1.98 to 2.59% (trees with or without crops) and from 1.45 to 2.06% (rice–berseem cropping). Microbial N ac-counted for 2.61–3.60% of the total soil nitrogen in the surface layers of soil (0–15 cm). With increasing depth, the proportion of microbial C and N in the to-tal soil organic pool decreased appreciably, the values being 1.15–1.42% for C and 1.42–1.60% for N.
The proportion of microbial C and N to soil organic C and N was found to be greater in tree-based systems. Immobilization of nitrogen was 2.61–3.60% (0–15 cm soil depth) in soils receiving organic inputs in the form of litterfall and fine roots from the trees. In the case of rice–berseem cropping or the fallow, relatively less nitrogen was immobilized in microbial biomass in the soil.
3.3. Carbon dioxide evolution and biomass specific activity
The CO2-C production from the soil varied with
tree species, soil depth and distance from trees. CO2-C production decreased significantly with
in-crease in soil depth (p<0.01). For the various treat-ments, carbon dioxide evolution rates for the three soil depths (mg CO2-C g−1 soil per day) were:
10.75–21.73 (0–7.5 cm); 2.92–5.78 (7.5–15 cm) and 1.94–3.00 (15–30 cm). The CO2 evolution rates
were significantly higher at 1 m from the rows of trees than at 2 m for the soil from 0–7.5 cm depth (p<0.05). With increase in soil depth, the effect of distance from the trees on CO2-C production was not
Table 4
Specific soil microbial activity (mg CO2-C produced mg−1 MBC per day) in soils of different management systems
Treatments Distance (m) mg CO2-C produced mg−1MBC per day LSD(p<0.05)
0–7.5 (cm) 7.5–15 (cm) 15–30 (cm)
Control – 208.57 85.60 218.25 35.98
Rice–berseem – 111.94 71.97 39.55 40.36
Acacia 1 122.90 52.81 98.29 14.43
2 105.85 67.44 113.40 17.18
Acacia+rice–berseem 1 125.31 59.17 108.10 29.07
2 123.59 93.31 120.03 17.20
Eucalyptus 1 118.91 57.96 108.24 42.78
2 106.23 67.87 117.29 27.03
Eucalyptus+rice–berseem 1 118.95 56.81 115.21 16.78
2 96.63 64.96 123.61 33.58
Populus 1 114.61 65.23 112.30 19.76
2 100.70 67.67 132.38 43.72
Populus+rice–berseem 1 119.43 72.25 100.07 11.03
2 103.62 93.93 119.47 18.80
LSD(p<0.05) 1 14.74 19.84 41.13
2 13.32 22.39 36.23 –
Microbial biomass specific respiratory activity did not vary significantly with tree species (Table 4). Spe-cific respiratory activity was comparatively greater at 0–7.5 and 15–30 cm than at 7.5–15 cm. Biomass spe-cific respiratory activity was higher in the control soil than in the tree plantations and agroforestry systems.
3.4. Soil inorganic N and nitrogen mineralization
There were significant variations in levels of soil inorganic nitrogen due to treatments (p<0.01, Fig. 2a). During all three seasons, soil inorganic N (15.89–17.37mg N g−1 soil) was maximum in the
case of Acacia+rice–berseem. The levels of soil in-organic N were 41–74, 8–30 and 32–42% higher, respectively, for Acacia, Eucalyptus and Populus based agroforestry systems than for rice–berseem. Soil inorganic N showed appreciable increases in the agroforestry systems as compared with the tree plantations. It is interesting to note that this increase was greater in the case of the Eucalyptus+crop sys-tem (17–47%) than in the Acacia+crop (7–13%) or Populus+crop system (6–16%).
There were significant variations in nitrogen min-eralization due to tree species, soil depth and seasons (p<0.01) (Table 5). Nitrogen mineralization was max-imum in July (6.69–31.03mg g−1soil) and minimum
in December (1.55–4.26mg g−1soil) (Fig. 2b).
Maxi-mum nitrogen mineralization during the rainy season corresponded with the active growth period of plants. In winter months, the presence of dead roots in soil cores together with low temperature caused low rates of N-mineralization. Nitrogen mineralization rates for different agroforestry systems(mg g−1soil) were:
Aca-cia (4.26–31.03), Populus (3.84–26.04) and Eucalyp-tus (3.13–22.87). In the agroforestry systems nitrogen mineralization increased by 12–37% as compared to monocultures.
On the basis of seasonal N mineralization rates, to-tal annual nitrogen release has been calculated. Toto-tal N release was highest in the Acacia+rice–berseem system (312.90 kg N ha−1) followed by Acacia alone
(297.14 kg N ha−1) and the other systems studied
(182.54–268.58 kg ha−1). Annual release of
nitro-gen was 13–95% higher under tree-based systems as compared to monocropping. Total annual nitrogen mineralization accounted for 24–31% of total soil N in tree/tree-cropping systems, 23% in rice–berseem cropping and 6.36% in the control treatment.
Due to higher biological activity, N mineraliza-tion rates were highest at 0–7.5 cm and decreased with increasing soil depth (Table 5). The rates were highest under Acacia (38.17–40.60) and Pop-ulus (28.31–39.12). The mineral nitrogen (NH4+-N
and NO3−-N) concentrations under the various
Fig. 2. Variations in (a) initial levels of soil inorganic N and (b) nitrogen mineralization rates in soils of different treatments (C=control; RB=rice–berseem; An=Acacia nilotica; An+RB=Acacia nilotica+rice–berseem; Et=Eucalyptus tereticornis; Et+RB=Eucalyptus tereticornis+rice–berseem; Pd=Populus deltoides; Pd+RB=Populus deltoides+rice–berseem).
Table 5
Variations in nitrogen mineralization rates at various soil depths in control, cropping system, tree plantation and agroforestry systems
N-mineralization (mg N g−1 soil) LSD(p<0.05)
Treatments 0–7.5 (cm) 7.5–15 (cm) 15–30 (cm)
Control 7.39 3.02 1.71 0.87
Rice–berseem 23.02 12.96 4.16 0.78
Acacia 38.17 15.28 5.68 1.34
Acacia+rice–berseem 40.60 16.91 5.89 2.06
Eucalyptus 22.06 10.67 4.77 2.41
Eucalyptus+rice–berseem 30.99 14.74 5.54 1.03
Populus 28.31 13.81 5.65 0.96
Populus+rice–berseem 39.42 15.27 5.69 1.19
LSD(p<0.05) 1.38 1.03 0.96 –
Table 6
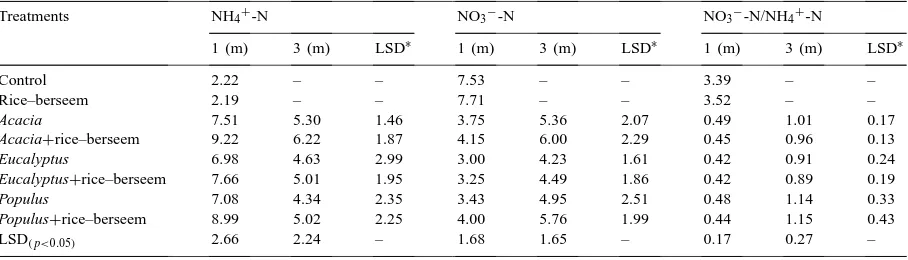
Variations in levels of NH4+-N and NO3−-N (mg g−1 soil) at two distances from rows of trees (averaged across the soil depths) in cropping, forestry and agroforestry systems
Treatments NH4+-N NO3−-N NO3−-N/NH4+-N
1 (m) 3 (m) LSD∗ 1 (m) 3 (m) LSD∗ 1 (m) 3 (m) LSD∗
Control 2.22 – – 7.53 – – 3.39 – –
Rice–berseem 2.19 – – 7.71 – – 3.52 – –
Acacia 7.51 5.30 1.46 3.75 5.36 2.07 0.49 1.01 0.17
Acacia+rice–berseem 9.22 6.22 1.87 4.15 6.00 2.29 0.45 0.96 0.13
Eucalyptus 6.98 4.63 2.99 3.00 4.23 1.61 0.42 0.91 0.24
Eucalyptus+rice–berseem 7.66 5.01 1.95 3.25 4.49 1.86 0.42 0.89 0.19
Populus 7.08 4.34 2.35 3.43 4.95 2.51 0.48 1.14 0.33
Populus+rice–berseem 8.99 5.02 2.25 4.00 5.76 1.99 0.44 1.15 0.43
greater amounts of ammonium were present and the NO3−-N/NH4+-N ratios were significantly lower
closer to the trees (p<0.05). The consistently lower NO3−-N/NH4+-N ratio near the trees (0.42–0.49)
than far from trees (0.89–1.11) in all the treatments indicated an efficient uptake of nitrate-N by tree roots.
4. Discussion
The integration of trees with crops on moderately alkali soils improved soil carbon and total nitrogen status. This was due to organic matter inputs in the form of litter fall and fine roots from the tree species. The level of increase in soil organic matter was influ-enced by the tree species as there were marked varia-tions in the quantity and quality of litterfall among the tree species (Kaur, 1998). A. nilotica alone or in com-bination with crops caused greater improvement of soil organic carbon content closely followed by Pop-ulus and Eucalyptus based systems. While studying the organic matter dynamics in these systems, Kaur (1998) found that the litter accumulation on the ground floor was higher in Acacia based systems (3465 kg dry matter ha−1per year) as compared to agroforestry
systems with Populus (2722 kg dry matter ha−1 per
year) and Eucalyptus (2107 kg dry matter ha−1 per
year). Besides litterfall, the fine roots also contributed to soil organic matter, which accounted for 27–23% of the total dry matter input to the soil. Fine root biomass in the surface soil layers close to the trees varied from 1.0 to 2.4 Mg ha−1per year. Various
stud-ies on sodic soils have shown appreciable build up of organic carbon status of alkali soils when planted to trees (Singh and Gill, 1992; Singh et al., 1994; Bhojvaid et al., 1996). In the control plot, high or-ganic matter content could be due to undisturbed soil conditions and greater stability of soil organic mat-ter as contributed by the litmat-ter and roots of the native vegetation.
Soil microbial biomass and its activity increased due to the ameliorative effects of trees and organic matter inputs. According to Rao and Pathak (1996), carbon is an important factor influencing microbial activity in alkali soils. In the agroforestry systems, microbial C and N increased by 10–60 and 17–43%, respec-tively. It seems likely that litter quality regulated the
levels of soil microbial biomass in tree-based systems. The leaf litter of Acacia and Populus were character-ized by medium N concentration (1.52–2.51%) and low lignin content (6.13–9.0%), favoring the growth of organisms and resulting in higher soil micro-bial biomass (Kaur, 1998).
The maintenance of a large microbial biomass pool has been reported to be dependent on soil organic matter (Powlson et al., 1987). The availability of car-bonaceous materials and substrates such as sugars, amino acids and organic acids to the soil from the roots is important for supplying maintenance energy for microbial populations (Bowen and Rovira, 1991). The importance of root exudates in maintaining a larger microbial biomass closer to the trees has also been reported by Browaldh (1997). The rapid de-cline in microbial biomass with depth in the soil and distance from the trees in the present study could be attributed to the quantity and quality of organic inputs.
An increased proportion of microbial carbon and nitrogen in the total soil organic carbon pool of agro-forestry systems indicates greater potential nutrient availability to the plants. Carbondioxide evolution rates and biomass specific respiratory activity were influenced by organic carbon build-up in the soil under different management systems. Sarig and Stein-berger (1994) reported that microbes invest more energy in microbial metabolism to overcome salt stress at high soil pH. At moderate soil pH, it may be the quantity and quality soil organic matter which have a dominating influence on carbon mineraliza-tion, specific microbial activity and soil nitrogen availability.
Higher N availability under agroforestry systems could be attributed to high organic inputs from the trees. For certain tropical agroforestry systems, Haggar et al. (1993) showed that higher rates of ni-trogen mineralization resulted from the build up of readily mineralizable organic N in the soil over 7 years of tree mulch application. In this study, maxi-mum rates of N-mineralization under A. nilotica may be attributed to high foliar N and rapid circulation of N through the litterfall. The ratio of NO3−-N and
NH4+-N was low closer to trees indicating an efficient
5. Conclusions
1. The integration of trees with crops improved the productivity and fertility of moderately alkaline soils. The agroforestry systems improved soil con-ditions by increasing total soil organic matter, soil nitrogen availability and biological activity after 6–7 years of tree growth.
2. Organic matter inputs from trees in the form of lit-terfall and fine roots contributed to increased soil organic matter content. The organic matter inputs into the soil for different tree species was in the order: Acacia>Populus>Eucalyptus. Soil microbial biomass and nitrogen mineralization rates showed appreciable increase in agroforestry systems. An increased proportion of microbial carbon and ni-trogen in the total soil organic pool indicate higher nutrient availability to the plants.
3. On the basis of soil microbial parameters, agro-forestry has been found to be a viable land-use option for utilizing moderately alkaline soils for meeting the needs of food, fodder and fuelwood as well as for maintaining soil fertility.
Acknowledgements
Financial assistance in the form of Senior Research Fellowship to B. Kaur from the Council of Scien-tific and Industrial Research, New Delhi is gratefully acknowledged.
References
Abrol, I.P., Bhumbla, D.R., 1971. Saline and alkali soils in India; their occurrence and management, World Soil Resources Reports. FAO, Rome, Vol. 41, pp. 42–51.
Anderson, T.H., Domsch, K.H., 1990. Application of eco-physiological quotients (qCO2 and qD) on microbial
biomass from soils of different cropping histories. Soil Biol. Biochem. 22, 251–255.
Anderson, T.H., Domsch, K.H., 1993. The metabolic quotient for CO2(qCO2) as a specific activity parameter to assess the effects
of environmental conditions, such as pH, on the microbial biomass of forest soils. Soil Biol. Biochem. 25, 393–395. Batra, L., Manna, M.C., 1997. Dehydrogenase activity and
microbial biomass carbon in salt-affected soils of semiarid and arid regions. Arid Soil Res. Rehab. 11, 295–303.
Bhojvaid, P.P., Timmer, V.R., Singh, G., 1996. Reclaiming sodic soils for wheat production by Prosopis juliflora (Swartz) DC afforestation in India. Agrofor. Syst. 34, 139–150.
Bremner, J.M., 1965. Total nitrogen. In: Black, C.A., Evans, D.D., White, J.L., Ensminger L.E., Clark, F.E. (Eds.), Methods of Soil Analysis, Vol. 2. American Society of Agronomy, Madison, pp. 595–624.
Brookes, P.C., Landman, A., Pruden, G., Jenkinson, D.S., 1985. Chloroform fumigation and the release of soil nitrogen: a rapid direct extraction method to measure microbial biomass nitrogen in soil. Soil Biol. Biochem. 17, 837–842.
Browaldh, M., 1997. Change in soil mineral nitrogen and respiration following tree harvesting from an agrisilvicultural system in Sweden. Agrofor. Syst. 35, 131–138.
Bowen, G.D., Rovira, A.D., 1991. The rhizosphere. The hidden half of the hidden half. In: Waisel, Y., Eshel, A., Kafkafi, U. (Eds.), Plant Roots: The Hidden Half. Marcel Dekker, New York, pp. 641–669.
Campbell, C.A., Biederbeck, V.O., Zentner, R.P., Lafond, G.P., 1991. Effect of crop rotations and cultural practices on soil organic matter, microbial biomass and respiration in a thin black chernozem, microbial biomass and respiration in a thin black chernozem. Can. J. Soil Sci. 71, 363–376.
Ellenberg, H., 1971. Nitrogen content, mineralization and cycling. In: Duvigneaud, P. (Ed.), Productivity in Forest Ecosystems. UNESCO, Paris, France, pp. 509–514.
Frankenberger Jr., W.T., Bingham, F.T., 1982. Influence of salinity on soil enzyme activities. Soil Sci. Soc. Am. J. 46, 1173–1177. Gomez, K.A., Gomez, A.A., 1984. Statistical Procedures for Agricultural Research, 2nd Edition. Wiley, New York, 680 pp. Gupta, R.K., Bhumbla, D.R., Abrol, I.P., 1984. Effect of soil pH, organic matter and calcium carbonate on dispersion behaviour of alkali soils. Soil Sci. 137, 245–251.
Gupta, S.R., Singh, J.S., 1981. Soil respiration in a tropical grassland. Soil Biol. Biochem. 13, 261–268.
Gupta, S.R., Sinha, A., Rana, R.S., 1990. Biomass dynamics and nutrient cycling in a sodic grassland. Int. J. Ecol. Environ. Sci. 16, 57–70.
Haggar, J.P., Tanner, E.V.J., Beer, J.W., Kass, D.C.L., 1993. Nitrogen dynamics of tropical agroforestry and annual cropping systems. Soil Biol. Biochem. 25, 1363–1378.
Insam, H., 1990. Are the soil microbial biomass and basal respiration governed by the climate regime? Soil Biol. Biochem. 22, 525–532.
Jenkinson, D.S., Ladd, J.N., 1981. Microbial biomass in soil: measurement and turnover. In: Paul, E.A., Ladd, J.N. (Eds.), Soil Biochemistry, Vol. 5. Marcel Dekker, New York, pp. 415–471.
Kalembasa, S.J., Jenkinson, D.S., 1973. A comparative study of titrimetric and gravimetric methods for the determination of organic carbon in soil. J. Sci. Food Agric. 24, 1085–1090. Kaur, B., 1998. Organic matter dynamics and nitrogen
mineralization in agroforestry systems. Ph.D. Thesis, Kurukshetra University, Kurukshetra, India, 161 pp.
McClung, G., Frankenberger Jr., W.T., 1985. Soil nitrogen transformations as affected by salinity. Soil Sci. 139, 405–411. Pathak, H., Rao, D.L.N., 1998. Carbon and nitrogen mineralization from added organic matter in saline and alkali soils. Soil Biol. Biochem. 30, 695–702.
Powlson, D.S., Brookes, P.C., Christensen, B.T., 1987. Measurement of soil microbial biomass provides an early indication of changes in total soil organic matter due to straw incorporation. Soil Biol. Biochem 19, 159–164.
Rana, R.S., Parkash, V., 1987. Floristic characterization of alkali soils in north-western India. Plant Soil 99, 447–451. Rao, D.L.N., Ghai, S.K., 1985. Urease and dehydrogenase activity
of alkali and reclaimed soils. Aust. J. Soil Res. 23, 661–665. Rao, D.L.N., Pathak, H., 1996. Ameliorative influence of organic
matter on biological activity of salt affected soils. Arid Soil Res. Rehab. 10, 311–319.
Sarig, S., Steinberger, Y., 1994. Microbial biomass response to seasonal fluctuation in soil salinity under the canopy of desert halophytes. Soil Biol. Biochem. 26, 1405–1408.
Singh, G., 1995. An agroforestry practice for the development of salt lands using Prosopis juliflora and Leptochloa fusca. Agrofor. Syst. 29, 61–75.
Singh, G., Gill, H.S., 1992. Ameliorative effect of tree species on characteristics of sodic soils at Karnal. Ind. J. Agric. Sci. 62, 142–146.
Singh, G., Singh, N.T., Abrol, I.P., 1994. Agroforestry techniques for the rehabilitation of salt affected soils in India. Land Degradation Rehab. 5, 223–242.
Singh, G., Singh, N.T., Dagar, J.C., Singh, H., Sharma, V.P., 1997. An evaluation of agriculture, forestry and agroforestry practices in a moderately alkali soil in northwestern India. Agrofor. Syst. 37, 279–295.
Singh, J.S., Raghubanshi, A.S., Singh, R.S., Srivastava, S.C., 1989. Microbial biomass acts as a source of plant nutrients in dry tropical forest and savanna. Nature 338, 499–500.
Smith, J.L., Paul, E.A., 1990. The significance of soil microbial biomass estimations. In: Bollag, J.M., Stotzky, G. (Eds.), Soil Biochemistry, Vol. 6. Marcel Dekker, New York, pp. 357–396. Vance, E.D., Brookes, P.C., Jenkinson, D.S., 1987. An extraction method for measuring soil microbial biomass Carbon. Soil Biol. Biochem. 19, 703–707.