www.elsevier.com / locate / bres
Short communication
Involvement of GABA neurons in allylnitrile-induced dyskinesia
a ,
*
a b aHideji Tanii
, Xiao-ping Zang , Naoaki Saito , Kiyofumi Saijoh
a
Department of Hygiene, Kanazawa University School of Medicine, Kanazawa, Japan
b
Laboratory of Molecular Pharmacology, Biosignal Research Center, Kobe University, Kobe, Japan
Accepted 19 September 2000
Abstract
Nitriles are a class of compounds with potential relevance to human health. Allylnitrile, one of nitriles, induces persistent behavioral abnormalities in mice. To explore what type of neuronal system is involved in these behavioral abnormalities, five neuronal markers,
g-aminobutyric acid (GABA), tyrosine hydroxylase, serotonin, the serotonin transporter and choline acetyltransferase were immuno-histochemically examined within various brain structures in allylnitrile and vehicle-treated mice. Allylnitrile induced changes in the immunolabelling of GABA in the medial habenula, interpeduncular nucleus, substantia nigra, dorsal raphe nucleus and median raphe nucleus; the amount of immunolabelling decreased in all of these brain structures except the medial habenula at 2 days postdosing, and increased in all of these structures at 14 days postdosing. Allylnitrile also induced changes in the amount of immunolabelling of tyrosine hydroxylase in the arcuate nucleus, substantia nigra pars compacta, locus coeruleus and caudoventrolateral reticular nucleus at either 2 or 14 days postdosing, depending on the structures. No immunohistochemical change was seen for serotonin, serotonin transporter and choline acetyltransferase. The present results suggest that the GABAergic systems through the medial habenula-interpeduncular nucleus-ascending raphe nuclei relay and through the substantia nigra may be involved in allylnitrile-induced behavioral abnormalities.
2000 Elsevier Science B.V. All rights reserved.
Theme: Disorders of the nervous system
Topic: Neurotoxicity
Keywords: Allylnitrile; GABA; Tyrosine hydroxylase
Nitriles are a class of neurotoxic compounds with (DA) in the brain of animals following allylnitrile
treat-potential relevance to human health. Nitriles are used ment [20,21,23]. Apoptotic changes are also shown to
broadly in the manufacture of plastics, solvents and occur in the brain following allylnitrile treatment [26]. The
synthetic intermediates. It has been shown that thermal interrelationship between these changes remains to be
degeneration of acrylonitrile-based plastics leads to the explored, and little is known about the full mechanism
emission of a great variety of nitriles [19]. Recently, we underlying persistent behavioral abnormalities.
have found that allylnitrile (CH25CHCH CN) induces in2 On the basis of these findings, we believe that
allylnit-rodents persistent behavioral abnormalities similar to the rile may produce either a temporal or long-term change in
excitement, choreoathetosis and circling syndrome (ECC neurotransmitters of these systems, resulting in the
persis-syndrome) described by Selye [18], i.e., head twitch, tent behavior changes observed. To test this hypothesis, we
circling, increased locomotor activity, occasional backward have investigated changes in neuronal expression of g
-pedaling and alteration in reflexive behavior [20,22]. We aminobutyric acid (GABA), NA, DA, serotonin and
acetyl-have studied the mechanism by which allylnitrile induces choline(Ach) in the mouse brain following allylnitrile
persistent behavioral abnormalities, and shown temporal treatment. GABA was chosen because it is ubiquitously
alterations in the activation of serotonergic activity, con- expressed throughout the brain being the primary inhibitor
tent of noradrenaline (NA) and metabolism of dopamine neurotransmitter, and has been implicated in the
patho-genesis of behavioral disorders [3].
Male ddy mice (Japan SLC Co., Shizuoka, Japan)
*Corresponding author. Fax:181-76-234-4232.
E-mail address: [email protected] (H. Tanii). weighing 25–30 g were used throughout the experiments.
All animal experiments were conducted according to the biotinylated horse anti-mouse antiserum in PBS containing
Guidelines of the Committee on Animal Experimentation 5% normal horse serum (for GABA; Vector Laboratories,
of Kanazawa University, Takara-machi Campus. Animals Burlingame, Calif., USA) or 1:200 biotinylated goat
anti-were maintained on a 12:12 h light:dark cycle, and allowed rabbit antiserum in PBS containing 5% normal goat serum
free access to laboratory food and tap water. The tempera- (for TH, ChAT, serotonin and the serotonin transporter,
ture was maintained at 22.0628C. Animals received a Vector Laboratories), washed in 5% normal horse serum or
single oral dose of allylnitrile (Tokyo Kasei Co., Tokyo, 5% normal goat serum in PBS for 1 h, and were then
Japan) (84 mg / kg) or the vehicle olive oil (4 mg / kg). incubated for 1 h in avidin–biotin complex reagents (ABC
From previous studies, this dose level of nitrile was shown kit, Vector Laboratories) containing 5% normal horse
to induce full behavioral abnormalities [26]. Changes in serum or 5% normal goat serum. After washes in PBS and
the spontaneous and reflex behaviors, documented in our in Tris–HCl buffered saline (TBS, pH 7.4), the sections
previous work [26], were observed over a period of 14 were incubated in TBS containing 0.04%
diaminoben-days. Briefly, observation of behavior was done between zidine, 0.08% nickel ammonium sulfate and 0.003% H O2 2
0900 and 1100 h. The animals were placed on a table top, for 10 min. The sections were washed in TBS, and then
and their behavior was observed. The dyskinetic behaviors PBS followed by distilled water. They were mounted on
assessed were circling, retropulsion, head twitch and gelatinized slides, dehydrated and prepared for
micro-alteration in tail hanging. Circling consisted of stereotyped scopic observation. Some sections were prepared without
circling, retropulsion was defined as displacement of the incubation with the primary or secondary antibodies
animal towards his back, and head twitch was backward serving as a negative control; these sections showed no
head movement. When normal animals were hung by the apparent staining.
tail and slowly lowered to the surface of a table, they The brain structures could be differentiated without
landed on their forelimbs with the head turned dorsad. The counterstaining, and were identified according to Paxinos
mice with diskinetic syndrome bent ventrally, resulting in and Watson [16]. Cells on both the left and right sides of
occipital landing. the brain that were immunostained for the different
Immunohistochemical studies were conducted on brains markers in each section were counted by one of the authors
from animals killed at 0 (immediately after treatment), 2 who had no knowledge of the treatments. The labelled
and 14 days postdosing with allylnitrile or olive oil. The cells were counted regardless of the staining intensity. The
animals were anesthetized with 100 mg / kg sodium pen- cell count was the sum of observations from at least four
tobarbital, and perfused transcardially with 0.01 M phos- sections per brain structure per animal with the number of
phate buffer containing 0.9% sodium chloride (PBS, pH cells immunostained per structure given as the
7.4) followed by cold fixative containing 4% paraformal- mean6S.E.M. for three animals. The mean number of
dehyde, 0.5% glutaraldehyde, and 0.2% picric acid in 0.1 immunopositive neurons per brain structure as a function
M phosphate buffer (PB, pH 7.4). The brains were of the postdosing survival periods was analyzed using
removed and postfixed in 4% paraformaldehyde and 0.2% analysis of variance, followed by Scheffe’s multiple
com-picric acid in PB overnight at 48C, and then placed in 30% parison. A significance level of P,0.05 was used.
sucrose in PB at 48C for a minimum of 2 days. The brains Consistent with our previous report [26], mice given
were rapidly frozen and sectioned coronally in a cryostat at allylnitrile exhibited behavioral abnormalities from day 1
20mm. We used a mouse anti-GABA monoclonal antibody to 14 after treatment, while the control mice displayed no
(0.1 ng IgG / ml, Sakaue et al. [17]), a rabbit anti-tyrosine behavioral abnormality. Circling and retropulsion were not
hydroxylase(TH) polyclonal antibody (1:5000; Chemicon,
Temecula, CA, USA), a rabbit anti-choline acetyltrans- Table 1
GABA-positive cell counts in brain structures of mice following
allylnit-ferase(ChAT) polyclonal antibody (1:1000, Chemicon), a a
rile treatment
rabbit anti-serotonin polyclonal antibody (1:8000; Sigma,
Regions Days after treatment
St. Louis, Mo, USA) and a rabbit anti-serotonin transporter
polyclonal antibody (1:10 000, Yamamoto et al. [25]). 0 2 14
Sections were placed for 4 days in PBS containing 0.3% MHb n.d. n.d. 12.263.2
Triton X-100 (PBST, pH 7.4) at 48C, and preincubated IP 58.3613.0 n.d. 183620.0* P50.0018
with 0.3% (v / v) H O2 2 and 5% normal horse serum in SNr 136626.5 20.065.6* 290630.9* P50.0002
SNc 16.963.2 n.d. 59.4611.7* P50.0050
PBST for GABA or 5% normal goat serum in PBST for
DR 2.860.9 4.662.2 19.664.5* P50.0013
TH, ChAT, serotonin and serotonin transporter to block
MnR 2.160.7 n.d. 17.068.2* P50.0355
endogenous peroxidase activity and non-specific binding of
a
Each value represents the mean6S.E.M. (n53). n.d. shows no GABA
antibodies. The sections were then incubated for 7 days at
detection. *Significantly different from the mean number from animals at
48C with the primary antibody diluted in PBST containing
0 day postdosing(P,0.05). MHb medial habenular nucleus, IP
inter-5% normal horse or goat serum. For GABA immuno- peduncular nucleus, SNr substantia nigra pars reticulata, SNc substantia
histochemistry Triton X-100 was omitted. After washing nigra pars compacta, DR dorsal raphe nucleus, MnR median raphe
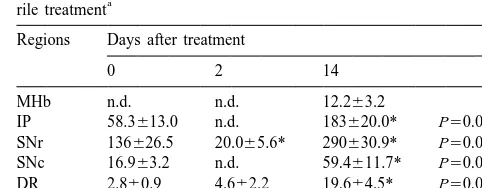
always observed during the assessment, but these be- Allylnitrile produced various changes in the
immuno-haviors often appeared when returning the mice to their histochemical staining of GABA-positive cells in brain
home cages. Head twitch and alteration in tail hanging structures dependent upon the days after treatment (Table
continued from day 1 to 14 after treatment. Increased 1). The medial habenular nucleus had no
GABA-immuno-locomotor activity was also observed around 2 days reactive cells at 0 and 2 days postdosings (Fig. 1A,B), but
following allylnitrile treatment. showed some GABA-positive cells at 14 days postdosing
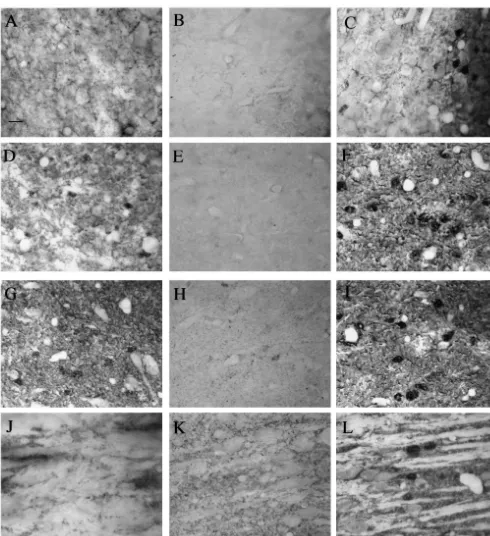
Table 2 area and dorsal raphe nucleus the number of TH-positive Tyrosine hydroxylase-positive cell counts in brain structures of mice cells tended to decrease by 2 days postdosing, but this
a
following allylnitrile treatment
decrease was not significant when compared to the number
Regions Days after treatment of cells seen at 0 day postdosing. This was also true for the
0 2 14 caudate putamen, globus pallidus, periventricular
hypo-thalamic nucleus and the paraventricular hypohypo-thalamic
VTA 307638.9 256617.7 293634.3 P50.2055
nucleus. The amount of TH immunolabelling in the arcuate
Arc 122611.3 51.0615.7* 128622.8 P50.0034
SNc 321641.7 269644.2 462651.0* P50.0060 nucleus, locus coeruleus (Fig. 2A–C) and caudoventrola-LC 195622.8 62.0619.3* 174625.4 P50.0013 teral reticular nucleus decreased significantly by 2 days DR 70.4613.2 56.369.3 77.6613.8 P50.1768
postdosing, and recovered by 14 days postdosing. The
CVL 33.561.7 25.762.6* 34.562.9 P50.0094
amount of TH immunolabelling in the SNc remained
a
Each value represents the mean6S.E.M. (n53). *Significantly different unchanged at 2 days postdosing, and was significantly from the mean number from animals at 0 day postdosing(P,0.05). VTA
higher at 14 days postdosing. In the control olive
oil-ventral tegmental area, Arc arcuate nucleus, SNc substantia nigra pars
treated mice the observed immunohistochemical labelling
compacta, LC locus coeruleus, DR dorsal raphe nucleus, CVL
caudoven-trolateral reticular nucleus. at 0, 2 and 14 days postdosings was similar to that seen for allylnitrile-treated animals at 0 day postdosing.
We examined ChAT, serotonin and the serotonin
trans-(Fig. 1C). In the interpeduncular nucleus, substantia nigra porter immunohistochemically. However, allylnitrile did
pars compacta (SNc) and median raphe nucleus, GABA- not induce any change at 2 or 14 days postdosing. No
positive cells were observed at 0 day postdosing (Fig. effect on ChAT immunolabelling was observed in the
1D,1J), were not seen at 2 days postdosing (Figs. 1E,K), caudate putamen (Fig. 3A–C) or on serotonin
immuno-and significantly increased at 14 days postdosing (Fig. labelling in median raphe nucleus (Fig. 3D–F), nor was
1F,L). The substantia nigra pars reticulata (SNr) showed any change observed in serotonin transporter
immuno-some GABA immunolabelling at 0 day postdosing (Fig. labelling (not shown).
1G), a significantly decreased immunolabelling at 2 days In the present study, changes in the neuronal markers
postdosing (Fig. 1H), and significantly increased immuno- GABA, TH, serotonin, serotonin transporter and ChAT
labelling at 14 days postdosing (Fig. 1I). In the dorsal were investigated in mice exhibiting behavioral
abnor-raphe nucleus, a higher level of the GABA immuno- malities similar to the ECC syndrome following a single
labelling was recorded at 14 days postdosing compared dose of allylnitrile. Among 5 neuronal markers, the amount
with the levels seen at 0 and 2 days postdosings. Although of GABA and TH immunolabelling changed in the various
the hippocampus and cerebral cortex displayed GABA brain structures at the different time points after allylnitrile
immunolabelling, the amount of immunolabelling re- treatment, while the ChAT, serotonin and serotonin
trans-mained unchanged over the experimental period. In the porter immunolabellings did not. The specific changes seen
control olive oil-treated animals the observed immuno- in this study provide support for some biochemical
altera-histochemical labelling at 0, 2 and 14 days postdosings tions seen in our previous studies [20,21,23], and suggest
was similar to that seen for allylnitrile-treated animals at 0 that the GABAergic system is involved in
allylnitrile-day postdosing. induced behavioral abnormalities.
In some brain regions, there was a difference in the Allylnitrile induced changes in GABA immunolabelling
amount of TH labelling between allylnitrile-treated mice in the medial habenular nucleus, interpeduncular nucleus,
and control animals (Table 2). In the ventral tegmental SNr, SNc, dorsal raphe nucleus and median raphe nucleus.
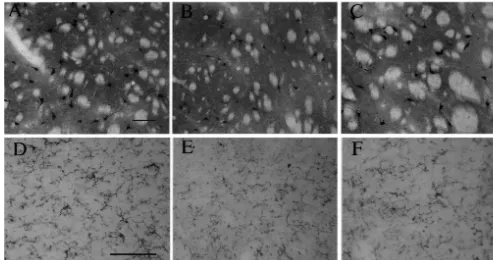
Fig. 3. ChAT immunolabelling in the caudate putamen (A–C) and serotonin immunolabelling in the median raphe nucleus (D–F) from allylnitrile-treated mice (84 mg / kg) at 0 (A,D), 2 (B,E) and 14 (C,F) days postdosings. Scale bar on A for A–C: 100mm. Scale bar on D for D–F: 100mm. No effect on ChAT labelling was observed in the caudate putamen or serotonin labelling in the median raphe nucleus at 2 and 14 days postdosings, when compared with the control (0 day postdosing).
It has been shown that there is an anatomical and func- labelling was observed at 0 and 2 days postdosings, but we
tional linkage between the medial habenular, interpeduncu- encountered a significant amount of the GABA
immuno-lar and ascending raphe nucleus; the habenula has a labelling at 14 days postdosing. To our knowledge, there
modulatory effect on the anterior raphe nuclei, and exerts a has been no report demonstrating the presence of GABA
tonic inhibitory influence on mesocortical, mesolimbic and immunoreactivity in the medial habenular nucleus,
al-mesostriatal dopaminergic neurons through the habenuloin- though what the GABA immunolabelling of this nucleus
terpeduncular pathways [9,13]. In the interpeduncular and means is not clear at present. The present changes in
raphe nucleus GABA interneurons have been thought to be GABA immunolabelling in the SNr suggest an
in-controled by or mediate input to these nuclei [4,10]. volvement of the GABAergic system in allylnitrile-induced
Decreased amount of GABA immunolabelling in the dyskinesia, since this brain structure contains GABA
interpeduncular and median raphe nucleus at 2 days neurons which project to the thalamus, superior colliculus
postdosing likely reflects decreased input from the medial and pedunculopontine nuclei [1,5,6], forming one of the
habenular nucleus to the interpeduncular nucleus and to the major outputs from the basal ganglia. These GABA
median raphe nucleus. Allylnitrile is known to induce neurons thus play a role in the extrapyramidal motor
apoptotic changes in the medial habenula 2 days after functions processed through the basal ganglia. The
de-treatment [26]. Additionally, the present result of de- crease in GABA immunolabelling in the SNr at 2 days
creased GABA immunolabelling explains the previous postdosing may be due to a functional impairment of the
finding that allylnitrile produces an activation of serotoner- striatonigral pathway. It has been shown that quinolic
gic system around 2 days after administration [21], be- acid-induced degeneration of the striatonigral pathway
cause GABA exerts an inhibitory control over central causes a marked loss of GABA immunolabelling in the
serotonergic neurons via GABA receptors located in the SNr [12], although no functional change has been reported
raphe nuclei [4,14]. What the increased amount of GABA in the striatonigral pathway from mice given allylnitrile.
immunolabelling in the interpeduncular, dorsal raphe and The increase in GABA immunolabelling in the SNr at 14
median raphe nucleus at 14 days postdosing means is not days postdosing indicates that the brain of an animal given
clear, although a long-term increment in glutamic acid allylnitrile requires GABA to suppress a state of excitation,
decarboxylase activity has been shown in the inter- since the tonically active inhibitory output from the SNr
peduncular nucleus following lesion of the habenula [10]. normally keeps down-stream movement generators in
connections of the substantia nigra in the rat, J. Comp. Neurol. 207
[2,11]. What the change in the GABA immunolabelling of
(1982) 283–303.
the SNc implies is not clear at present, but might be related
[6] A.R. Granata, S.T. Kitai, Inhibitory substantia nigra inputs to the
to change in the SNc or to the change in SNr observed at pedunculopontine neurons, Exp. Brain Res. 86 (1991) 459–466.
14 days postdosing. Previous studies have demonstrated [7] F.G. Gonon, Nonlinear relationship between impulse flow and
dopamine released by rat midbrain dopaminergic neurons as studied
direct projections from the GABAergic neurons of the SNr
by in vivo electrochemistry, Neuroscience 24 (1988) 19–28.
to the DA cells of the SNc [8,24].
[8] M. Hajos, S.A. Greenfield, Synaptic connections between pars
Allylnitrile induced changes in TH immunolabelling in 4 compacta and pars reticulata neurons: electrophysiological evidence
brain structures at either 2 or 14 days postdosing although for functional modules within the substantia nigra, Brain Res. 660
(1994) 216–224.
allylnitrile-treated animals continued to display behavioral
[9] B.L. Jacobs, E.C. Azmitia, Structure and function of the brain
abnormalities over a period of 1 to 14 days postdosing. As
serotonin system, Physiol. Rev. 72 (1992) 165–229.
with the GABA immunolabelling in the SNc, it is not clear [10] M.M. Mata, B.K. Schrier, R.Y. Moore, Interpeduncular nucleus:
at present why the amount of TH immunolabelling in the differential effects of habenula lesions on chronic acetyltransferase
SNc(probably due to changes in DA the neurons) increased and glutamic acid decarboxylase, Exp. Neurol. 57 (1977) 913–921.
[11] E.J. Neafsey, C.D. Hull, N.A. Buchwald, Preparation for movement
by 14 days postdosing, but this increase might be related to
in the cat: II. Unit activity in the basal ganglia and thalamus,
changes in the striatum, since the DA neurons in the SNc
Electroenceph. Clin. Neurophysiol. 44 (1978) 714–723.
have a role in maintaining stable levels of DA in the [12] L.F.B. Nicholson, R.L.M. Faull, H.J. Waldvogel, M. Dragunow,
striatum [7,15]. The decreased levels of TH immuno- GABA and GABAA receptor changes in the substantia nigra of the
rat following quinolinic acid lesions in the striatum closely resemble
labelling in the locus coeruleus and caudoventrolateral
Huntington’s disease, Neuroscience 66 (1995) 507–521.
reticular nucleus (probably due to decreased levels of NA)
[13] T. Nishikawa, D. Fage, B. Scatton, Evidence for, and nature of, the
at 2 days postdosing support previous finding where tonic inhibitory influence of habenulointerpeduncular pathways upon
allylnitrile produces a decrease in NA content in the cerebral dopaminergic transmission in the rat, Brain Res. 373 (1986)
324–336.
hippocampus, cerebral cortex, hypothalamus and midbrain
[14] T. Nishikawa, B. Scatton, Inhibitory influence of GABA on central
around 2 days after treatment [21].
serotonergic transmission. Raphe nuclei as the neuroanatomical site
In conclusion, among 5 neuronal markers investigated, of the GABAergic inhibition of cerebral serotonergic neurons, Brain
GABA immunolabelling specifically changed in associa- Res. 331 (1985) 91–103.
tion with observed behavioral abnormalities. The [15] P.G. Overton, D. Clark, Burst firing in midbrain dopaminergic
neurons, Brain Res. Rev. 25 (1997) 312–334.
GABAergic systems through the medial habenular
nu-[16] G. Paxinos, C. Watson, The Rat Brain in Stereotaxic Co-ordinates,
cleus-interpeduncular nucleus-ascending raphe nuclei relay
Academic Press, New York, 1986.
and through the substantia nigra may be involved in the [17] M. Sakaue, K. Kumoi, N. Saito, C. Tanaka, Immunohistochemical
mechanism underlying allylnitrile-induced behavioral ab- demonstration of GABA and aspartate-containing neurons in rat
normalities. Further studies will be needed to explore the inferior olive, Biomed. Res. 10 (1989) 287–295.
[18] H. Selye, Lathyrism, Rev. Can. Biol. 16 (1957) 1–82.
mechanism by which allylnitrile specifically affects GABA
[19] M.M. Shapi, A. Hess, Gas chromatographic-mass spectrometric
neurons.
analysis of some potential toxicants among volatile compounds emitted during large-scale thermal degradation of poly(acrylonitrile-butadiene-styrene)plastic, J. Chromatogr. 562 (1991) 681–696. [20] H. Tanii, M. Hayashi, K. Hashimoto, Behavioral syndrome induced Acknowledgements
by allylnitrile, crotononitrile or 2-pentenenitrile in rats, Neuro-pharmacology 30 (1991) 887–892.
This work was supported by a Grant-in-Aid for
Sci-[21] H. Tanii, J. Huang, K. Hashimoto, Involvement of noradrenergic
entific Research (C11670340; HT) from the Ministry of and 5-hydroxytryptaminergic systems in allylnitrile-induced head
Education, Science, Sports and Culture, Japan. twitching, Brain Res. 626 (1993) 265–271.
[22] H. Tanii, Y. Kurosaka, M. Hayashi, K. Hashimoto, Allylnitrile: a compound which induces long-term dyskinesia in mice following 13 a single administration, Exp. Neurol. 103 (1989) 64–67.
References [23] H. Tanii, A. Okayama, A. Yamatodani, M. Hayashi, K. Hashimoto,
Alterations in the metabolism of serotonin and dopamine in the [1] M. Bentivoglio, D. Van der Kooy, H.G.J.M. Kuypers, The organiza- mouse brain following a single administration of allylnitrile, which tion of the efferent projections of the substantia nigra in the rat. A induces long-term dyskinesia, Toxicol. Lett. 58 (1991) 323–328. retrograde fluorescent double labeling study, Brain Res. 174 (1979) [24] J.M. Tepper, L.P. Martin, D.R. Anderson, GABAA
receptor-me-1–17. diated inhibition of rat substantia nigra dopaminergic neurons by
[2] G. Chevalier, J.M. Deniau, Disinhibition as a basic process in the pars reticulata projection neurons, J. Neurosci. 15 (1995) 3092– expression of striatal functions, Trends Neurosci. 13 (1990) 277– 3103.
280. [25] H. Yamamoto, M. Fujimiya, Y. Shirai, M. Nakashita, M. Oyasu, N. [3] T.M. De Lorey, R.W. Olsen, GABA and glycine, in: G.J. Siegel, Saito, Immunohistochemical localization of serotonin transporter in B.W. Agranoff, R.W. Albers, P.B. Molinoff (Eds.), Basic Neuro- normal and colchicine treated rat brain, Neurosci. Res. 32 (1998) chemistry, Raven Press, New York, 1994, pp. 389–400. 305–312.