www.elsevier.com / locate / bres
Short communication
Subcellular distribution of calponin and caldesmon in rat hippocampus
a a b a ,
*
Christopher Agassandian , Marc Plantier , Abdelatif Fattoum , Alfonso Represa
,
a
Elisabeth der Terrossian
a
INMED/INSERM U29, Parc Scientifique de Luminy, 13009 Marseille Cedex, France
b
CRBM du CNRS-UPR1086, Route de Mende, 34293 Montpellier Cedex 5, France
Accepted 19 September 2000
Abstract
Caldesmon and calponin are two actin- and calmodulin-binding proteins involved in the ‘actin-linked’ regulation of smooth muscle and 21
non-muscle Mg -actin-activated myosin II ATPase activity. In the present report we show that caldesmon and calponin are present in the post-synaptic side of symmetric synapses and accumulate in the post-synaptic densities of asymmetric synapses. Caldesmon- and calponin-immunoreactivities are also observed at the plasma membrane of the hippocampal neurones. Finally, while caldesmon seems strictly distributed to neurones, acidic calponin is present in both neurones and astrocytes. 2000 Elsevier Science B.V. All rights reserved.
Theme: Cellular and molecular biology
Topic: Cytoskeleton transport and membrane targeting
Keywords: Acidic calponin; Caldesmon; Dendritic spines; Astrocytes; Microfilaments
1. Introduction interaction of these proteins with calcium binding-proteins
which causes their release from the microfilaments.
Cal-F-actin-linked cytoskeleton is particularly enriched in desmon and Cp binding to F-actin may also be regulated
the synaptic domains of mature neurons [4] where it may by the phosphorylation of these proteins (for review see
contribute to the synaptic plasticity and motility by either Ref. [6]).
the polymerisation-depolymerisation of microfilaments or / According to recent data both Cd and Cp: (1) bind to
21
and to the contractile activity of the actomyosin system. In myosin, actin, and Ca -calmodulin, and they are good
vitro studies, essentially performed on smooth muscle substrates for protein-kinases [13]. Furthermore, they also
proteins, have demonstrated that the contractile activity of bind to microtubules in vitro and this association is
21
the actomyosin system is mainly regulated by the phos- regulated by Ca -calmodulin and phosphorylation [5,10];
phorylation of one of the myosin light chains (reviewed in (2) are present in neuronal growth cones and glial cells in
Ref. [9]). In brain as in smooth muscles, there is also culture [1,11,14], where they collocate with actin and
evidence for the presence of a secondary actin-linked myosin; (3) are also present in the somatodendritic
do-regulatory system, represented mainly by three actin-bind- mains of mature rat brain neurones in situ while apparently
ing proteins, inhibitors of the actomyosin activity: tropo- absent from axonal tracts [14].
myosin (TM), caldesmon (Cd) and calponin (Cp). While In an attempt to shed more light on the specific
21
the binding of TM to F-actin is Ca -independent, the functions of Cd and Cp, we investigate here their
subcellu-21
binding of Cd and Cp is modulated by Ca through the lar distribution in the hippocampus of adult rats.
*Corresponding author. Present address: INMED-INSERM U29, 163 2. Materials and methods
Route de Luminy, BP 13, 13273 Marseille Cedex 09, France. Tel.:
Polyclonal antibodies, affinity purified on their
respec-133-491-828-114; fax:133-491-828-101.
E-mail address: [email protected] (A. Represa). tive antigens covalently linked to Affigel (Bio-Rad), were:
pig platelet Cd (pCald) or turkey gizzard Cd (gCald), used
at 2.5 mg / ml [15]; turkey gizzard Cp (Gcp), used at 2.5
mg / ml; and C-terminal sequence of rat brain acidic
isoform Cp (Rcp), used at 0.5mg / ml [14]. Also used were
monoclonal antibody to b-actin (2 mg / ml; Sigma, clone
kj43a), biotinylated anti-mouse (1 / 2000; Sigma) and biotinylated anti-rabbit (1 / 1000; Vector).
Caldesmon was extracted at pH 6.9 from mature rat hippocampi and affinity purified as described before [16]. Triton X-100 insoluble cytoskeleton was extracted from rat hippocampi as described before [14], treated in a Dounce homogeniser with 2.5 ml of 1% Triton X-100, 70 mM dithiothreitol, 40 mM NaCl, 50 mM imidazole-HCl pH
6.9, 5 mM MgCl2 and EGTA, 1 mM azide, 0.5 mM
benzamidine, 0.2 mM diisopropyl fluorophosphate, and 20
mg of each the following antiproteases: leupeptin,
pep-statin, pefabloc, antipain. SDS–PAGE, Western blots and immunohistochemical procedures were performed as previ-ously described [14].
Two-dimensional gels were carried out as outlined in our previous report [14]. Purified Cd or Cp peptide was dissolved in 9.4 M urea, 2% Nonidet P-40, and 5% w / v dithiothreitol. Two-dimensional electrophoresis resolved acidic proteins (pI 3–10 for Cd) by isoelectric focusing in the first dimension for 15 h at 400 V. After washing the gel was equilibrated for 20 min in solubilisation buffer (10 mM Tris–HCl, 10% (v / v) 2-b-mercaptoethanol, 2% SDS, 15% glycerol and 0.001% bromophenol blue). Proteins were identified by Western blotting using the peroxidase technique.
For electron microscopy adult male Wistar rats (220– 250 g) were perfused transcardially with buffered 2% paraformaldehyde, 0.5% glutaraldehyde and brains kept in
the fixative for 6 h. Vibratome sections (60 mm) were
incubated overnight with polyclonal Cd or Cp antibodies. Sections were then washed and incubated in biotinylated anti-rabbit antibody (Amersham) and avidin-horseradish peroxidase (Vector). The reaction was visualised by treat-ment with 0.05% diaminobenzidine plus 0.01% hydrogen
peroxide. The sections were postfixed in 1% OsO , rinsed,4
cut into small pieces, dehydrated and embedded in a
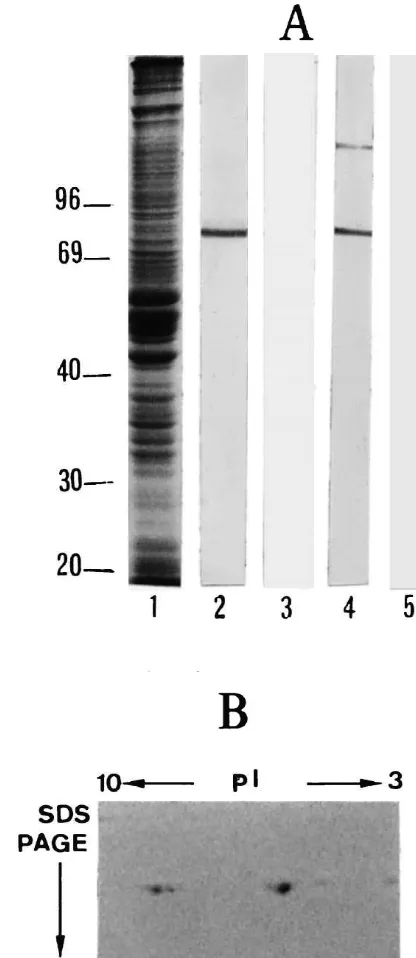
Fig. 1. Cd-immunoreactivity on Western blots. (A) Some 50-mg proteins
mixture of EPON-812 and Araldite-502. Areas of interest
from an homogenate of adult rat hippocampus were fractionated by
were then selected and their surfaces carefully sectioned on
SDS–12% gel electrophoresis and stained by Coomassie Blue R250 (lane
an ultramicrotome.
1). Corresponding blots were processed with 1mg / ml pig platelet Cd (lanes 2 and 3) and gizzard Cd (lanes 4 and 5) antibodies. Antibodies
25
were saturated with 10 M affinity purified brain Cd (lanes 3 and 5). (B) Separation by two-dimensional gel electrophoresis of the different isoforms of Cd (Triton X-100 insoluble cytoskeleton); caldesmon was
3. Results
labelled using the antibody to pCald at 1mg / ml.
The specificity of antibodies was monitored by
immuno-blotting (Fig. 1). One major band of |83 kDa was antibodies with Cd purified either from rat hippocampus or
recognised by both antibodies to Cd following SDS–PAGE chicken gizzard showed their high specificity (Fig. 1A).
and Western blotting. However, the antibody to gCald also Analysis of Cd purified from mature rat hippocampus
detected a faint band of|120 kDa, which may correspond (two-dimensional electrophoresis) revealed the presence of
to the Cd present in the blood vessels, where the protein is three isoforms of 83 kDa with a pI ranging from 5.6 to 7.
insoluble residue suggesting that these bands did not result Cp may help to protect the structure and the organisation
from a residual phosphorylation of a common isoform of the complicated short actin filament network of the
(phosphorylated Cd does not bind to F-actin). We conclude post-synaptic elements from severing, depolymerisation or
that in mature rat hippocampus there are three isoforms of proteolysis by proteins such as the actin depolymerising
Cd (Fig. 1B). The high specificity of the antibodies to Cp factor [2] or proteases.
has been described before [14]. It is interesting to note that Cd has been shown to
Adjacent hippocampal slices were labelled with either contribute to the capping of the receptors on the surface of
pig platelet or chicken gizzard Cd antibodies; both anti- T lymphocytes [17]. Caldesmon and Cp may therefore be
bodies gave a similar pattern of immunostaining. In the involved in the capping of the receptors in the
post-mature rat hippocampus, Cd antibodies exclusively stain synaptic neuronal membrane, thus maintaining a
favour-neurones, mainly pyramidal cells and interfavour-neurones, within able structure for receptor activity. The presence of Cd and
the Ammon’s horn. As previously reported [15], labelling Cp in the post-synaptic elements of asymmetric (essentially
of glial cells was never observed. Calponin-immuno- glutamatergic in the hippocampus) but also symmetrical
reactivity (Cp-IR) was also present in the hippocampus, (essentially GABAergic) synapses indicates that the
func-where neurons were slightly immunoreactive while as- tion of these two proteins is not dependent on the type of
trocytes were strongly immunoreactive. Neuronal staining receptors.
obtained with Cd and Cp antibodies was punctiform as In the present report we also show that Cd and Cp
described before [15] (not shown). distribute along dendritic microtubules, in agreement with
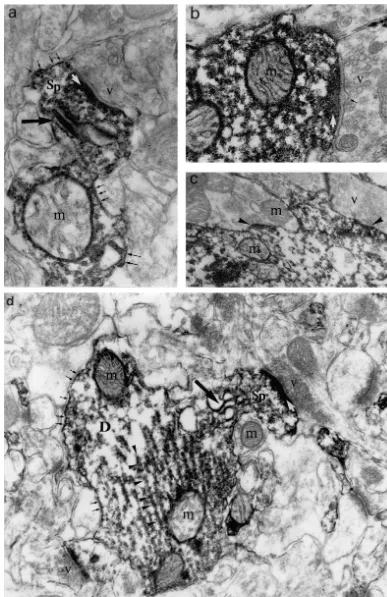
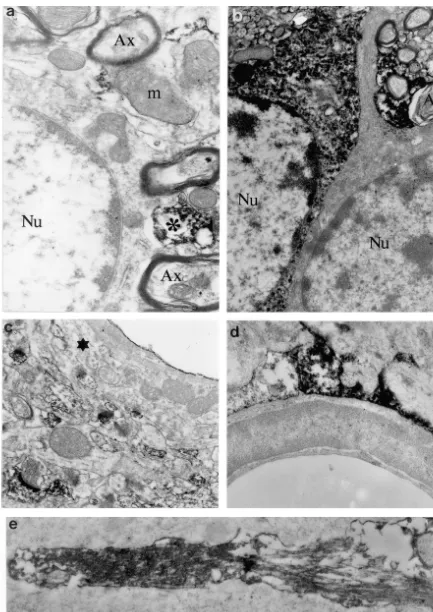
The immuno-electron-microscopy analysis of Cd and Cp previous biochemical data, suggesting that these proteins
revealed that some dendrites were stained with Cd or Cp bind to microtubules [5,10]. It is therefore possible that
antibodies while other dendrites in the same field were not they play a modulatory role in their assembly or transport
(Figs. 2 and 3) suggesting that only a subset of neurons to the dendrites.
was immunoreactive. The patches of Cd immunoreactivity Finally, our work has also confirmed the colocalisation
seemed to be associated with the microtubule network in of acidic brain Cp with glial intermediate filaments [14].
the dendritic shaft. Dendritic spines were intensely stained The effect of this association on the function of astrocytes
by Cd and Cp antibodies. Strong accumulations of Cd- or is unknown, but it may suggest that Cp regulates the
Cp-IR were seen in the post-synaptic density (white arrows motility and plasticity of glial extensions. In contrast to
in Figs. 2 and 3) and between, but never inside, the sacs of Cp, Cd immunoreactivity, which was reported to be
the spine apparatus (large black arrow in Figs. 2 and 3). present on cultured glial cells [15], is apparently absent
The labelling was present as scattered deposits on the from identified glial cells in situ. Different factors may
filamentous network in the subsynaptic web and in the explain this contradiction, such as the neuron-glia
interac-dendritic shaft from which the spines emerged. The pre- tions (absent from astroglial cultures) or the metabolic
synaptic domains characterised by the accumulation of the stress of cultured astrocytes.
synaptic vesicles were never labelled. Calponin but not In conclusion, in the mature hippocampus, the lack of
caldesmon antibodies also stained glial cell bodies and Cd and of Cp in the axons and terminal boutons provides
extensions (Fig. 3). Within these radial-like glial exten- strong evidence that Cd and Cp are not involved in the
sions, acidic Cp is particularly enriched along the bundles formation of pre-synaptic vesicle clusters nor in the
of intermediate filaments (Fig. 3e). molecular mechanism regulating the neurotransmitter
re-lease. Their presence in the post-synaptic elements sup-ports the hypothesis that they may be active in the
4. Discussion modulation and organisation of the morphological structure
of the cytoskeleton in these areas. This study constitutes the first analysis of the
localisa-tion of Cd and Cp at the ultrastructural level in mature
neurons. We show that Cd and Cp are restricted to the Acknowledgements
postsynaptic domains, where they likely collocate with
actin [7], myosin II [12] and TM4 [8], and are totally The authors are grateful to S. Guidasci and to J.-P.
absent from the presynaptic bouton, reinforcing previous Pagnac for photographic assistance, and to I. Jorquera and
data depicting differences between pre- and post-synaptic H. Trabelsi-Terzidis for expert technical assistance.
cytoskeleton. We conclude that Cd and Cp are not required for the normal function of mature axon terminals in vivo
but that they may contribute in contrast to the plasticity of References
the postsynaptic specialisations. In fact, microfilaments in
the postsynaptic domain are apparently involved in the [1] E.M. Abd-el-Basset, I. Ahmet, S. Fedoroff, Actin and actin-binding
shape and size changes occurring on dendritic spines [4]. proteins in differentiating astroglia in tissue culture, J. Neurosci.
[2] B.W. Bernstein, J.R. Bamburg, Tropomyosin binding to F-actin tropomyosin are localized in neuronal growth cones, J. Neurosci. protects the F-actin from disassembly by brain actin-depolymerizing Res. 127 (1995) 441–451.
factor (ADF), Cell. Motil. 2 (1982) 1–8. [12] M. Morales, E. Fifkova, In situ localization of myosin and actin in [3] E. Ehler, G. Karlhuber, H.C. Bauer, A. Draeger, Heterogeneity of dendritic spines with the immunogold technique, J. Comp. Neurol.
smooth muscle-associated proteins in mammalian microvasculature, 279 (1989) 666–674.
Cell Tissue Res. 279 (1995) 393–403. [13] F. Nakamura, T. Mino, J. Yamamoto, M. Naka, T. Tanaka, Identifi-[4] E. Fifkova, M. Morales, Actin matrix of dendritic spines, synaptic cation of the regulatory site in smooth muscle calponin that is plasticity and long-term potentiation, Int. Rev. Cytol. 139 (1992) phosphorylated by protein kinase, J. Biol. Chem. 268 (1993) 6194–
267–307. 6201.
[5] T. Fujii, T. Hiromori, M. Hamamoto, T. Suzuki, Interaction of [14] M. Plantier, A. Fattoum, B. Menn, Y. Ben-Ari, E. Der Terrossian, A. chicken gizzard smooth muscle calponin with brain microtubules, J. Represa, Acidic calponin immunoreactivity in postnatal rat brain Biochem. 122 (1997) 344–351. and cultures: subcellular localization in growth cones, under the [6] M. Gimona, J.V. Small, Calponin, in: M. Barany (Ed.), Biochemistry plasma membrane and along actin and glial filaments, Eur. J.
of Smooth Muscle Contraction, Academic Press, San Diego, 1996, Neurosci. 11 (1999) 2801–2812.
pp. 91–103. [15] A. Represa, H. Trabelsi-Terzidis, M. Plantier, A. Fattoum, I. [7] P. Greengard, Interactions of synaptic vesicles with F-actin, Science Jorquera, C. Agassandian, Y. Ben-Ari, E. der Terrossian, Dis-259 (1993) 780–785. tribution of caldesmon and of the acidic isoform of calponin in [8] L. Had, C. Faivre-Sarraihl, C. Legrand, J. Mery, J. Brougidou, A. cultured cerebellar neurons and in different regions of the rat brain: Rabie, Tropomyosin isoforms in rat neurons: the different de- an immunofluorescence and confocal microscopy study, Exp. Cell velopmental profiles and distributions of TM-4 and TMBr-3 are Res. 221 (1995) 333–343.
consistent with different functions, J. Cell Sci. 107 (1994) 2961– [16] K. Sobue, T. Tanaka, K. Kanda, N. Ashino, S. Kakiuchi,
Purifica-2973. tion and characterization of caldesmon77: a calmodulin-binding
[9] A. Horowitz, C.B. Menice, R. Laporte, K.G. Morgan, Mechanisms protein that interacts with actin filaments from bovine adrenal of smooth muscle contraction, Physiol. Rev. 76 (1996) 967–1003. medulla, Proc. Natl. Acad. Sci. USA 82 (1985) 5025–5029. [10] R. Ishikawa, O. Kagami, C. Hayashi, K. Kohama, The binding of [17] G. Walker, W.G.L. Kerrick, Y.W. Bourguignon, The role of
caldes-non-muscle caldesmon from brain to microtubules: regulation by mon in the regulation of receptor capping in mouse T-lymphoma
21