Daftar Pustaka
DAFTAR PUSTAKA
[1] Report of the Third World Academy of Sciences. Safe Drinking Water: The
Need, The Problem, Solution and An Action Plan. www.twas.org:2002.
[2] Ratnaningsih, Enny, Hiskia Achmad. Kimia Organik, Ilmu Kimia dan
Kehidupan, dan Ilmu Kimia Lingkungan. Jurusan Kimia FMIPA-ITB.
Bandung:1997.
[3] Syafputri, Sri I. “Pembuatan Porous Membrane dari Pasir Aktif untuk Filter
Air”. Skripsi. Teknik Fisika, FTI-ITB: 2006.
[4] Dyalfikri, Ahmad. “Pembuatan Filter Air dari Karbon-Aktif dengan Metode
Semi-dry Press”. Skripsi. Teknik Fisika, FTI-ITB: 2006.
[5] Van der Toorn, J.D., A biological approach to water purification: I.
Theoretical aspects. 1987.
[6] Huisman, Prof. Ir. L., Rapid Filtration–Part I. Delf University of Technology, Department of Civil Engineering – Division of Sanitary Engineering. Delf: 1974.
[7] Doulton-Filter, Various Drinking Water Treatment Methods. Brochure (www.doultonfilters.com).
[8] Stokke, Jennifer, Bill White, dan Sara Charbonnet. Water, water,
everywhere: Filtering Filty Water-Filter Design Lab. (www.spice.centers.ufl.edu)
[9] Ye, Shufeng, at all. Preparation and Characterization of Ceramic Porous
Filter Using Pottery Raw Materials. Orlando: 2006.
[10] Baksi, S., Soumitra Biswas, dan S. Mahajan. Activated Carbon from
Bamboo–Technology Development towards Commercialisation. (www.tifac.org.in).
[11] Masschelein, Willy J. Unit Processes In Drinking Water Treatment. Marcel Dekker Inc. New York: 1992.
[12] Chesters, J. H. Refractories: Production and Properties. The Metal Society. London: 1983.
Program Studi Teknik Material Institut Teknologi Bandung
Ibnu Maulana Yusuf 137 02 051
Glass to Silica Sand for Dual Media Filtration. Journal of Environmental
Engineering and Science vol.1. 2002. Halifax–Kanada.
[14] Reynolds, Tom D. unit Operations and Processes In Environmental
Engineering. Brooks/ Cole Engineering Division – A Division of
Wodsworth Inc. California: 1982.
[15] Kwon. Processing of Ceramics and Cermets. ME477
[16] Reed, James E. Introduction to the Principles of Ceramic Processing. John Wiley and Sons. Singapore: 1989.
[17] Lee, Jong-Heun. Advanced Electronic Materials II. Korea University: 2004.
(http://mse.korea.ac.kr)
[18] Rezwan, Prof. Dr. Ing. Kurosch. Ceramic Nanotechnology: Shaping
Ceramic I – Bulk Materials. Universität Bremen. Bremen: 2007.
[19] Dinsdale, Allen. Pottery Science: Materials, Process, and Products. Ellis Horwood Limited. Chichester: 1986.
[20] ASTM C 819–77
[21] SK Menteri Kesehatan no.907/ MENKES/ SK/ VII/ 2002.
[22] Askeland, Donald R., Pradeep P. Phulé. The Science and Engineering of
Materials, 4th ed.
[23] MSDS PVA Sciencelab.com (www.sciencelab.com)
Program Studi Teknik Material Institut Teknologi Bandung
Ibnu Maulana Yusuf 137 02 051
Lampiran
LAMPIRAN 1
DAFTAR SENYAWA-SENYAWA
YANG DAPAT/ TIDAK DAPAT DIADSORPSI KARBON-AKTIF
Tabel L.1.1. Bahan kimia dengan tingkat probabilitas very-high
teradsorpsi oleh karbon-aktif
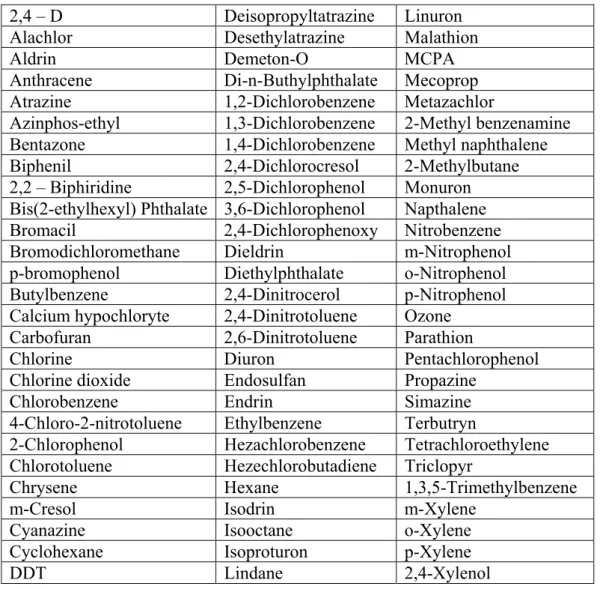
2,4 – D Deisopropyltatrazine Linuron
Alachlor Desethylatrazine Malathion
Aldrin Demeton-O MCPA
Anthracene Di-n-Buthylphthalate Mecoprop Atrazine 1,2-Dichlorobenzene Metazachlor Azinphos-ethyl 1,3-Dichlorobenzene 2-Methyl benzenamine Bentazone 1,4-Dichlorobenzene Methyl naphthalene Biphenil 2,4-Dichlorocresol 2-Methylbutane 2,2 – Biphiridine 2,5-Dichlorophenol Monuron
Bis(2-ethylhexyl) Phthalate 3,6-Dichlorophenol Napthalene
Bromacil 2,4-Dichlorophenoxy Nitrobenzene Bromodichloromethane Dieldrin m-Nitrophenol
p-bromophenol Diethylphthalate o-Nitrophenol Butylbenzene 2,4-Dinitrocerol p-Nitrophenol Calcium hypochloryte 2,4-Dinitrotoluene Ozone
Carbofuran 2,6-Dinitrotoluene Parathion
Chlorine Diuron Pentachlorophenol
Chlorine dioxide Endosulfan Propazine
Chlorobenzene Endrin Simazine
4-Chloro-2-nitrotoluene Ethylbenzene Terbutryn
2-Chlorophenol Hezachlorobenzene Tetrachloroethylene Chlorotoluene Hezechlorobutadiene Triclopyr
Chrysene Hexane 1,3,5-Trimethylbenzene
m-Cresol Isodrin m-Xylene
Cyanazine Isooctane o-Xylene
Cyclohexane Isoproturon p-Xylene
DDT Lindane 2,4-Xylenol
Program Studi Teknik Material Institut Teknologi Bandung
Ibnu Maulana Yusuf 137 02 051
teradsorpsi oleh karbon-aktif
Aniline Dibromo – 3 – chloropropane 1 – Pentanol
Benzene Dibromochloromethane Phenol Benzyl alcohol 1,1 – Dichloroethylene Phenylalanine Benzoic acid Cis-1,2-Dichloroethylene o-Phthalic acid
Bis(2-chloroethyl) ether Trans-1,2-Dichloroethylene Styrene
Bromodichloroethane 1,2 – Dichloropropane 1,1,2,2-Tetrachloroethane
Bromoform Ethylene Toluene
Carbon tetrachloride Hydroquinone 1,1,1-Trichloroethane 1 – Chloropropane Methyl Isobutyl Keton Trichloroethylene
Chlorotoluron 4 - Methylbenzenamine Vinyl acetate
Tabel L.1.3. Bahan kimia dengan tingkat probabilitas moderate
teradsorpsi oleh karbon-aktif[*]
Acetic acid Dimethoate Methionine
Acrylamide Ethyl acetate Methyl-tert-butyl ether
Chloroethane Ethyl ether Methyl ethyl ketone Chloroform Freon 11 Pyridine
1,1 - Dichloroethane Freon 113 1,1,2 - Thrichloroethane 1,2 - Dichloroethane Freon 12 Vinyl chloride
1,3 - Dichloropropee Glyphosate
Dikegulac Imazypur [*] Hanya efektif pada kasus tertentu.
Tabel L.1.4. Bahan kimia dengan tingkat probabilitas unlikely to be effective
teradsorpsi oleh karbon-aktif
Acetone Methylene chloride Acetonitrile 1 – Propanol
Acrylonitrile Propionitrile Dimethylformaldehyde Propylene
1,4 – Dioxane Tetrahydrofuran Isopropyl alcohol Urea
Methyl chloride
Program Studi Teknik Material Institut Teknologi Bandung
Ibnu Maulana Yusuf 137 02 051
Lampiran
LAMPIRAN 2
DATA HASIL PENGUJIAN KEKUATAN LENTUR FILTER
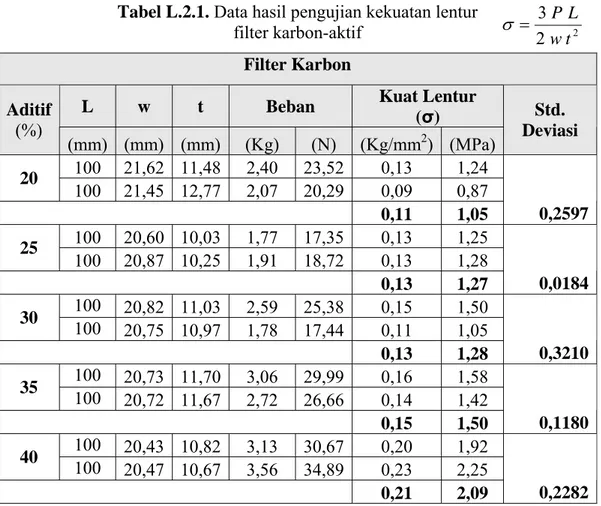
Tabel L.2.1. Data hasil pengujian kekuatan lentur
filter karbon-aktif 2 2 3 t w L P = σ Filter Karbon
L w t Beban Kuat Lentur
(σ) Aditif (%) (mm) (mm) (mm) (Kg) (N) (Kg/mm2) (MPa) Std. Deviasi 100 21,62 11,48 2,40 23,52 0,13 1,24 20 100 21,45 12,77 2,07 20,29 0,09 0,87 0,11 1,05 0,2597 100 20,60 10,03 1,77 17,35 0,13 1,25 25 100 20,87 10,25 1,91 18,72 0,13 1,28 0,13 1,27 0,0184 100 20,82 11,03 2,59 25,38 0,15 1,50 30 100 20,75 10,97 1,78 17,44 0,11 1,05 0,13 1,28 0,3210 100 20,73 11,70 3,06 29,99 0,16 1,58 35 100 20,72 11,67 2,72 26,66 0,14 1,42 0,15 1,50 0,1180 100 20,43 10,82 3,13 30,67 0,20 1,92 40 100 20,47 10,67 3,56 34,89 0,23 2,25 0,21 2,09 0,2282
Program Studi Teknik Material Institut Teknologi Bandung
Ibnu Maulana Yusuf 137 02 051
filter silika-aktif 2 2 t w = σ Filter Silika
L w t Beban Kekuatan Lentur (σ) Aditif (%) (mm) (mm) (mm) (Kg) (N) (Kg/mm2) (MPa) Std. Deviasi 100 20,70 11,78 0,55 5,39 0,03 0,28 40 100 20,73 12,65 0,42 4,14 0,02 0,19 0,02 0,23 0,0667 100 20,82 10,50 0,73 7,11 0,05 0,47 35 100 20,85 10,95 0,71 6,95 0,04 0,42 0,04 0,44 0,0340 100 20,55 14,10 1,66 16,27 0,06 0,60 30 100 20,40 13,83 1,55 15,19 0,06 0,58 0,06 0,59 0,0096 100 19,63 13,93 2,73 26,75 0,11 1,05 25 100 20,42 14,85 2,12 20,78 0,07 0,69 0,09 0,87 0,2551 100 20,52 11,47 3,11 30,52 0,17 1,70 20 100 20,38 11,28 2,91 28,52 0,17 1,65 0,17 1,67 0,0343
Program Studi Teknik Material Institut Teknologi Bandung
Ibnu Maulana Yusuf 137 02 051
Lampiran
LAMPIRAN 3
SK MENTERI KESEHATAN No. 907/ MENKES/ SK/ VII/ 2002 Tentang Syarat-syarat dan Pengawasan Kualitas Air Minum
Tabel L.3.1. Daftar bahan anorganik Parameter Satuan Kadar Maksimum
yang Diperbolehkan Keterangan
1 2 3 4 Ammonia mg/ L 1,5 Alumunium mg/ L 0,2 Klorida mg/ L 250 Tembaga mg/ L 1 Kesadahan mg/ L 500 Hidrogen Sulfida mg/ L 0,05 Besi mg/ L 0,3 Mangan mg/ L 0,1 pH - 6,5 – 8,5 Sodium mg/ L 200 Sulfat mg/ L 250
Total Zat Padat Terlarut mg/ L 1000
Seng mg/ L 3
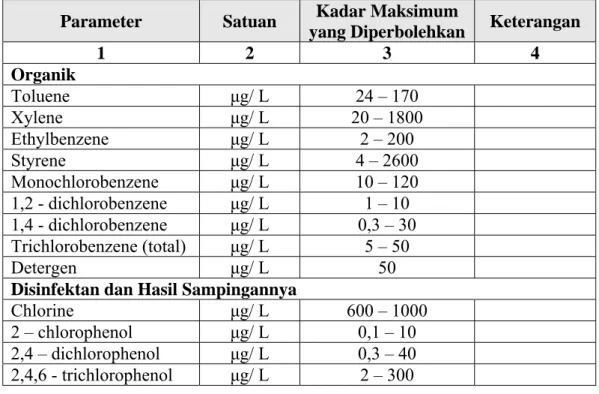
Tabel L.3.2. Daftar bahan organik, disinfektan dan hasil sampingannya Parameter Satuan Kadar Maksimum
yang Diperbolehkan Keterangan
1 2 3 4 Organik Toluene μg/ L 24 – 170 Xylene μg/ L 20 – 1800 Ethylbenzene μg/ L 2 – 200 Styrene μg/ L 4 – 2600 Monochlorobenzene μg/ L 10 – 120 1,2 - dichlorobenzene μg/ L 1 – 10 1,4 - dichlorobenzene μg/ L 0,3 – 30 Trichlorobenzene (total) μg/ L 5 – 50 Detergen μg/ L 50
Disinfektan dan Hasil Sampingannya
Chlorine μg/ L 600 – 1000 2 – chlorophenol μg/ L 0,1 – 10 2,4 – dichlorophenol μg/ L 0,3 – 40 2,4,6 - trichlorophenol μg/ L 2 – 300
Program Studi Teknik Material Institut Teknologi Bandung
Ibnu Maulana Yusuf 137 02 051
![Tabel L.1.3. Bahan kimia dengan tingkat probabilitas moderate teradsorpsi oleh karbon-aktif [*]](https://thumb-ap.123doks.com/thumbv2/123dok/4228462.2867873/4.892.158.806.218.439/tabel-bahan-kimia-tingkat-probabilitas-moderate-teradsorpsi-karbon.webp)