www.elsevier.com / locate / bres
Short communication
Pentobarbital-induced modulation of flexor and H-reflexes in Spinal
rats
*
Marcus Duke, Claire Advokat
Department of Psychology236 Audubon Hall Louisiana State University, Baton Rouge, LA 70803, USA Accepted 8 August 2000
Abstract
Electrophysiological recordings of the H-reflex and nonnociceptive flexion reflex were obtained from pentobarbital-anesthetized Intact rats and from both, anesthetized and unanesthetized groups of Acute and Chronic Spinal rats. Results showed that the flexor, but not H-reflex, of Chronic Spinal rats was significantly larger than that of all other groups, which did not differ among themselves. The antispastic drug baclofen dose-dependently decreased the flexion response of Chronic Spinal rats (A 54.3 mg / kg62.1 and 9.0 mg / kg).
50
2000 Elsevier Science B.V. All rights reserved.
Theme: Motor systems and sensorimotor integration
Topic: Reflex function
Keywords: Spasticity; Spinal rat; Flexor reflex; H-reflex; Rate dependent suppression; Baclofen
Spinal cord injury (SCI) produces devastating functional used to evaluate the effects of centrally acting muscle
losses, including spastic paresis and chronic pain, for relaxants in nonhuman animal models, produced by
non-which present treatments are often unsatisfactory [3,20]. nociceptive stimulation [11,14,16,19]
Efforts to improve available therapies include the develop- Unfortunately, in animal models, these
electrophysio-ment of animal models that approximate at least some logical recordings are usually invasive and often require
aspects of the clinical conditions [15,18]. the use of anesthetics, which may mask or interact with the
Most of these studies use at least one of two electro- physiological condition or assessment of drug action. For
physiological measures to assess reflex function, i.e. the this reason, we have recently developed the Chronic Spinal
Hoffman (H)-reflex or the flexor reflex (FR). The H-reflex rat as an experimental model in which to assess
electro-is the electrophysiological expression of the monosynaptic physiological indices of reflex function in the awake,
(stretch) reflex [11,14,15,18,19] and exaggeration of this unanesthetized animal [1,2]. Experiment 1 extends our
response may occur in SCI as one manifestation of previous work, in two ways. First, parameters of the
H-spasticity [12,17]. When elicited at frequencies greater than and flexor-reflex, both of which were obtained from each
0.3 Hz, there is a decrease in H-reflex amplitude in normal rat, were compared in Intact, Acute Spinal and Chronic
humans, termed frequency, rate-dependent or post-activa- Spinal groups for the development of hyperreflexia after
tion depression, which is reportedly reduced in spastic spinal transection. Second, because the Intact rats had to be
human patients [4,8,12,17]. The FR is a polysynaptic anesthetized for these recordings, we included additional
response evoked in the flexor muscles, usually of the groups of Acute and Chronic Spinal rats, which also
hindlimb, by either nociceptive (e.g. $6.860.2 mA of received the anesthetic. Pentobarbital [14] was chosen
shock; 6) or nonnociceptive input. Because this reflex may because our preliminary studies indicated that volatile
also be hyperreactive in spasticity [13], the FR has been anesthetics (e.g. isoflurane; 5) abolished the
nonnocicep-tive FR (unpublished data); ketamine [11,15,18] had an excitatory effect on the FR in intact rats (unpublished
*Corresponding author. Tel.:11-22-538-88-500; fax: 1
1-22-538-84-data); and an a-chloralose / urethane combination
125.
E-mail address: [email protected] (C. Advokat). [11,14,19] can only be used if the preparation is terminal,
which would preclude using the Chronic Spinal rat for To examine rate-sensitive depression, the stimulus
in-evaluation of multiple agents or chronic drug administra- tensity that produced the maximum response was used to
tion. elicit 10 consecutive reflexes at each of four frequencies:
In Experiment 2 we assessed the effect of (6) Baclofen 0.3, 1.0, 3.0 and 5.0 Hz, with a 5-min inter-rate interval.
(BAC; a GABAB agonist) on the nonnociceptive FR of Both, H-reflex response amplitudes and latencies (ms) were
chronic spinal rats. Although baclofen is presently the obtained for all 10 responses at each stimulus rate and the
most efficacious, clinically available agent for the treat- means determined for each frequency.
ment of spasticity [7,10], most of the nonhuman behavioral Following H-reflex assessment, the FR was examined in
research has assessed its purported analgesic effects. To the contralateral leg, using the same procedures as
previ-our knowledge this is the first report describing the dose- ously reported [1,2], i.e. five square wave shocks, at 500
related effect of systemic baclofen on the nonnociceptive Hz, 0.2-ms duration. Stimulus intensity was set at 2.53
FR in a proposed animal model of SCI-induced spasticity. threshold and five responses were elicited at approximately
Experiment 1 included 33 male albino Sprague–Dawley 30-s intervals. Each response was rectified and integrated,
rats (Division of Laboratory Animal Medicine, Louisiana within a time window of 200 ms, providing an index of the
State University Veterinary School, Baton Rouge, LA), area under the curve (AUC) in mV3ms as the measure of
weighing an average of 348 (62) g; and Experiment 2 reflex magnitude. The FR response was defined as the
used 28 male albino Sprague–Dawley rats (Holtzman mean of the five elicited FR responses for each rat.
Laboratories, Madison, WI), weighing an average of 339 For comparisons among the groups, the M-wave,
H-(66) g. All rats were singly housed in plastic cages in a reflex and FR responses were analyzed by either one-way
colony room maintained on a 12:12 h light:dark cycle, ANOVAs or two-way repeated measure ANOVAs
(Sigma-with dark onset at 19:00 h, and had continuous access to Stat, Jandel, San Rafael, CA). If a significant overall effect
food and water. The procedures for spinalization and was indicated, post-hoc tests (Newman–Keuls) were
per-post-operative care have been described in detail [1,2]. formed to determine which groups differed.
Acute Spinal rats (Experiment 1) were tested 2 days after In Experiment 2, the effect of baclofen on the FR of
surgery and Chronic Spinal rats were tested an average of Chronic Spinal rats was tested by first obtaining five
41 (6,1) days (Experiment 1) or 35 (61) days (Experi- pre-drug baseline scores for each rat. Separate groups were
ment 2) after surgery. All stimulation and recording then injected with either saline (n59) or subcutaneously
procedures were performed with a Nicolet Viking IV D with 1.0 (n54), 3.0 (n55), 10.0 (n55), or 30.0 (n55)
system (Nicolet Instrument Corporation, Madison WI). At mg / kg (6) BAC (Research Biochemicals, Natick, MA).
the end of the experiment rats were euthanized by an The FR was again assessed 30, 60, 90 and 120 min later.
anesthetic overdose or administration of CO . All pro-2 At each post-drug test point, the effect of BAC was
cedures were reviewed and approved by the Institutional quantified as percent (%) of baseline with the formula:
Animal Care and Use Committee of Louisiana State post-drug score / pre-drug score3100. The area under each
University (Baton Rouge, LA). time-effect curve (AUC) was calculated for each rat with
In Experiment 1, five groups of rats were injected the computer program PHARM / PCS (MicroComputer
intraperitoneally with either 50 mg / kg pentobarbital or Specialists, Philadelphia, PA). With this transformation a
saline (1 ml / kg). There was one group of unoperated, dose–response curve was obtained and the A50 value
Intact rats (Intact / Pento: n57), two groups of Acute (695% Confidence Intervals; CI) was calculated using the
Spinal rats (Acute / Pento: n57; Acute Spinal: n57), and Litchfield–Wilcoxon method (PHARM / PCS,
MicroCom-two groups of Chronic Spinal rats (Chronic / Pento: n56; puter Specialists). This dose–response function was also
Chronic Spinal: n56). Thirty minutes after the respective analyzed by one-way ANOVA. For all statistics, results
injections, the tibial nerve was stimulated (single square- were considered significant at P,0.05.
wave pulses; 0.2 ms duration) using a pair of platinum Table 1 summarizes the stimulus intensities and
corre-subdermal needle electrodes (Astro-Med, Inc., Atlanta, sponding response magnitudes for the FR, and the
maxi-GA) inserted subcutaneously in the gastrocnemius lateralis. mum H- and M-waves. The mean shock intensity for
Electromyogram (EMG) recordings were made from the elicitation of the FR (2.53threshold) differed among the
ipsilateral hindpaw using a pair of stainless steel barbed groups [F(4,32)54.61, P,0.005]. Specifically, the mean
broaches (Patterson Dental, New Orleans, LA) inserted shock value for the Chronic Spinal rats was significantly
percutaneously into the plantar muscles. lower than that of all pentobarbital groups (i.e. all groups
Prior to making the experimental recordings, 32 succes- except the Acute Spinal group). The magnitude of the FR
sive stimulations of increasing intensity (0.8–26.7 mA) also differed among the groups [F(4, 32)511.91, P,
were administered to determine the stimulus required to 0.001]. Importantly, the Chronic Spinal rats had a
sig-elicit the maximum reflex amplitude for each rat. Mag- nificantly larger FR (in response to a lower threshold
nitudes of the evoked EMG potentials were obtained by stimulus) than all other groups.
measuring the peak-to-peak amplitude (mV) of each H- Although the stimulus (mA) necessary to produce the
reflex, as well as the preceding M-wave (produced by maximum H-reflex did not differ among the groups, there
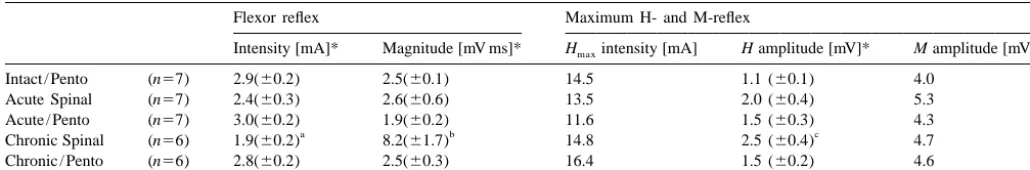
Table 1
Summary of spinal reflex results for each group presented as mean (6S.E.M.)
Flexor reflex Maximum H- and M-reflex
Intensity [mA]* Magnitude [mV ms]* Hmaxintensity [mA] H amplitude [mV]* M amplitude [mV]
Intact / Pento (n57) 2.9(60.2) 2.5(60.1) 14.5 1.1 (60.1) 4.0
Significantly different from all pentobarbital groups (i.e. not the Acute Spinal), P,0.05.
b
Significantly different from all other groups, P,0.05.
c
Significantly different from Intact / Pento group, P,0.05.
*
Significant group difference, P,0.05.
significantly larger in Chronic Spinal rats than Intact / Pento among themselves. The fact that the stimulus intensity
rats [F(4,32)54.19, P,0.01]. There was no significant under pentobarbital was the same for Intact, Acute and
difference across conditions in the stimulus necessary to Chronic Spinal conditions suggests that the drug (at the 50
produce the maximum M-reflex (data not shown), or its mg / kg dose) was more potent in Chronic than in Acute
amplitude. Spinal rats.
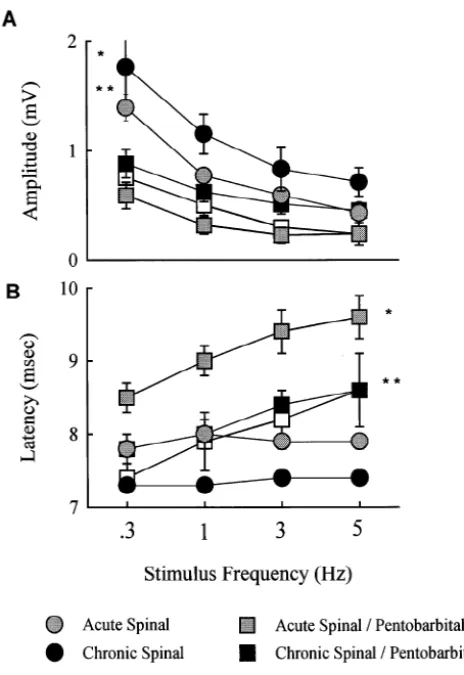
As seen in Fig. 1A, rate-dependent depression was This differential effect is more evident in regard to
obtained, in that the amplitude of the H-reflex, across all response magnitude, which was substantially greater in the
frequencies, differed among the groups [F(4,84)58.91, Chronic Spinal rats than in all other groups, which did not
P,0.001]. Overall, the H-reflex of Chronic Spinal rats was differ among themselves. Again, the size of the FR in
significantly larger, i.e. rate-sensitive depression was less, Acute Spinal rats under pentobarbital was the same as that
than the response of all pentobarbital groups (i.e. all except of Acute rats without the anesthetic. In contrast, the
the Acute Spinal group). In addition, rate-sensitive depres- response of Chronic Spinal rats was larger without the
sion in Acute Spinal rats was less than in Intact / Pento and anesthetic. This outcome is consistent with an earlier
Acute / Pento rats. Taken together, these results suggest that report of increased FR ‘sensitivity’ of Chronic (2 months)
pentobarbital increases rate sensitive depression of the Spinal compared with Acute (40–48 h) Spinal rats, to
spinally transected rat. intraperitoneal administration of a variety of drugs [9].
As seen in Fig. 1B, there were also significant differ- Taken together, the decrease in stimulus intensity required
ences in H-reflex latencies. First, collapsed across fre- to elicit the flexor, and the hyperresponsiveness of the
quency, there was a main effect of condition [F(4,84)5 Chronic Spinal rats, support the development of spasticity
9.97, P,0.001], in that, the latency of the Acute / Pento in this preparation, although, admittedly, this conclusion
group was longer than all other groups and the latency of must be tentative because comparisons could not be made
the Chronic / Pento group was longer than that of the with an unanesthetized, Intact group.
Chronic Spinal group. These results show that pentobarbi- Unlike the flexor, there was no difference between
tal increases the H-reflex latency of the spinally transected Acute and Chronic Spinal rats in either intensity or
rat. Second, there was a main effect of frequency, i.e. amplitude of the maximum H-reflex, or M-wave. Nor did
H-reflex latencies became longer at each of the stimulus pentobarbital differentially affect these measures in Spinal
frequencies [F(3, 84)527. 83, P,0.001]. Third, there was rats. However, pentobarbital did significantly influence
a significant interaction between condition and frequency rate-dependent suppression of the H-reflex. It decreased
[F(12, 84)54.05, P,0.001]. These results suggest that, amplitudes and increased latencies compared to the
re-regardless of condition (i.e. intact or spinally transected) sponse of the respective, unanesthetized, spinal groups.
pentobarbital increased the latency of the H-reflex. But Acute and Chronic Spinal rats did not differ in the
Fig. 2 shows the effects of BAC on the FR of Chronic absence of pentobarbital. Furthermore, the latencies of
Spinal rats. As expected, BAC produced a dose-dependant unanesthetized Spinal rats did not differ across stimulus
decrease in the magnitude of the FR [F(4,27)510.32, frequency, whereas the latencies of pentobarbital-treated
P,0.001] with an A of 4.3 mg / kg (95% CI: 2.1 and rats increased as a function of frequency. Finally, the
50
9.1). Though they did not differ from one another, the two pentobarbital-induced increase in latency was significantly
highest doses (10 and 30 mg / kg) significantly reduced the greater in Acute Spinal than Chronic Spinal or Intact rats,
FR in comparison to all other groups. suggesting that the Acute Spinal rats were more vulnerable
These results (Table 1) show that both, spinalization and to the anesthetic.
pentobarbital, significantly and differentially affected re- The absence of a hyperactive H-reflex, or impaired
flex function. The stimulus intensity necessary to evoke the rate-dependent inhibition in Chronic, relative to Acute
FR in Chronic Spinal rats was lower than that required for Spinal rats, does not support the use of this measure as a
Fig. 2. The effect of (6) BAC on the flexor reflex of Chronic Spinal rats.
The data represent the AUC / 10 (6S.E.M.) of the values obtained at 30,
60, 90, and 120 min after SC injection of 1.0, 3.0, 10.0, 30.0 mg / kg BAC, or saline administration. There was a significant difference among the groups. * At doses of 10.0 and 30.0 mg / kg BAC significantly reduced the flexor reflex compared to all other doses and saline (P,0.05). The
A50was 4.3 mg / kg (95% CI: 2.1 and 9.1).
Although this H-reflex analysis did not provide evidence of spasticity, there was a significant influence of pen-tobarbital, which was consistent with recent results show-ing that halothane anesthesia also increased the stimulus threshold of the H-reflex in Acute Spinal rats, relative to an unanesthetized group [5]. Because Spinal animals do not require anesthesia, they may provide useful comparisons with results from clinical investigations of awake, human
Fig. 1. (A) The effect of pentobarbital on the amplitude of the H-reflex at subjects. In fact, one recent study [13] reported that 88% 0.3, 1, 3 and 5 Hz in Intact, Acute, and Chronic Spinal rats. The data of spastic patients had ‘pathologically enhanced flexor represent the mean (6S.E.M.) amplitude (in mV) at each frequency
reflexes in the lower limbs,’ . . . ‘accompanied in 47% of
following injection of either pentobarbital (50 mg / kg, i.p.) or saline.
cases by abnormal decrease of reflex threshold.’
Adminis-* The amplitude of Chronic Spinal rats (filled circles) was significantly
tration of intrathecal baclofen reduced amplitudes and
greater than that of all groups (P,0.05) except Acute Spinal rats (shaded
circles). ** The amplitude of Acute Spinal rats was significantly greater increased thresholds in all patients. It was concluded that than that of Intact and Acute Spinal / Pentobarbital groups (P,0.05; open
data from FR recordings added to that provided by clinical
and shaded squares, respectively). (B) The effect of pentobarbital on the
measures, and that the FR was ‘a useful tool’ for
‘quantify-latency of the H-reflex at 0.3, 1, 3 and 5 Hz in the same groups of Intact,
ing the benefit of antispastic treatment.’
Acute, and Chronic Spinal rats shown in (A). The data represent the mean (6S.E.M.) latency (in ms) at each frequency. * The latency of the Acute
Spinal / Pentobarbital group (shaded squares) is significantly longer than that of all other groups (P,0.05). ** The latency of the Chronic Spinal /
Acknowledgements
Pentobarbital group (filled squares) is significantly longer than that of the Chronic Spinal group (P,0.05; filled circles). When collapsed across
groups, the latency of the H-reflex is significantly longer at each The authors would like to thank Michal Maranto for her successive stimulus frequency.
assistance in performing the surgical procedures and data entry.
be premature because hyperreflexia might have been seen
at longer post-transection intervals [9,15], or at other References
frequencies, intensities or patterns of stimulation [5,15,18].
In fact, H-reflex hyperreactivity in patients with spasticity [1] C. Advokat, M. Duke, Comparison of morphine-induced effects on
thermal nociception, mechanoreception, and hind limb flexion in
is often quite variable and may not always be evident in
chronic spinal rats, Exp. Clin. Psychopharmacol. 7 (1999) 219–225.
comparison with nonspastic control subjects [12]. Even
[2] C. Advokat, M. Duke, R. Zeringue, Dissociation of (2)
baclofen-rate-dependent modulation of the H-reflex may not be induced effects on the tail withdrawal and hindlimb flexor reflexes
correlated with clinical measures of hyperreflexia in the of chronic spinal rats, Pharmacol. Biochem. Behav. 63 (1999)
[3] M.D. Christensen, C.E. Hulsebosch, Chronic central pain after spinal rigidity, and cerebellar hypotonia, J. Neurol. Neurosurg. Psychiatry cord injury, J. Neurotrauma 14 (1997) 517–537. 30 (1967) 325–331.
´ `
[4] M. Faist, D. Mazevet, V. Dietz, E. Pierrot-Deseilligny, A quantitative [13] M. Parise, L. Garcıa-Larrea, P. Mertens, M. Sindou, F. Mauguiere, assessment of presynaptic inhibition of Ia afferents in spastics. Clinical use of polysynaptic flexion reflexes in the management of Differences in hemiplegics and paraplegics, Brain 117 (1994) 1449– spasticity with intrathecal baclofen, Electroencephalogr. Clin.
1455. Neurophysiol. 105 (1997) 141–148.
¨
[5] M. Gozariu, V. Roth, F. Keime, D. Le Bars, J.C. Willer, An [14] M. Schwarz, T. Klockgether, U. Wullner, L. Turski, K.-H. Sontag, electrophysiological investigation into the monosynaptic H-reflex in d-Aminovaleric acid antagonizes the pharmacological actions of
the rat, Brain Res. 782 (1998) 343–347. baclofen in the central nervous system, Exp. Brain Res. 70 (1988) [7] M. Malcangio, N.G. Bowery, GABA and its receptors in the spinal 618–626.
cord, Trends Pharmacol. Sci. 17 (1996) 457–462. [15] R.D. Skinner, J.D. Houle, N.B. Reese, C.L. Berry, E. Garcia-Rill, [8] J. Nielsen, N. Petersen, M. Ballegaard, F. Biering-Sorensen, O. Effects of exercise and fetal spinal cord implants on the H-reflex in Kiehn, H-reflexes are less depressed following muscle stretch in chronically spinalized adult rats, Brain Res. 729 (1996) 127–131. spastic spinal cord injured patients than in healthy subjects, Exp. [16] M. Sypniewska, The effect of baclofen on the hind limb flexor reflex Brain Res. 97 (1993) 173–176. of the spinal rat, Pol. J. Pharmacol. Pharm. 31 (1979) 493–501. [9] M. Nozaki, J.A. Bell, D.B. Vaupel, W.R. Martin, Responses of the [17] S. Taylor, P. Ashby, M. Verrier, Neurophysiolgical changes
follow-flexor reflex to LSD, tryptamine, 5-hydroxytryptophan, methox- ing traumatic spinal lesions in man, J. Neurol. Neurosurg. Psychiatry amine, and d-amphetamine in acute and chronic spinal rats, Psycho- 47 (1984) 1102–1108.
pharmacology 55 (1977) 13–18. [18] F.J. Thompson, P.J. Reier, C.C. Lucas, R. Parmer, Altered patterns [10] G. Ochs, A. Struppler, B.A. Meyerson, B. Linderoth, J. Gybels, B.P. of reflex excitability subsequent to contusion injury of the rat spinal
Gardner, R. Teddy, A. Jamous, P. Weimann, Intrathecal baclofen for cord, J. Neurophysiol. 68 (1992) 1673–1686.
long-term treatment of spasticity: a multicenter study, J. Neurol. [19] L. Turski, P. Jacobsen, T. Honore, D.N. Stephens, Relief of Neurosurg. Psychiatry 52 (1989) 933–939. experimental spasticity and anxiolytic / anticonvulsant actions of the
´
[11] H.-R. Olpe, H. Demieville, V. Baltzer, W. L Bencze, W.P. Koella, P. alpha-amino-3-hydroxy-5-methyl-4-isoxazolepropionate antagonist Wolf, H.L. Haas, The biological activity of D- and L-baclofen 2,3-dihydroxy-6-nitro-7-sulfamoyl-benzo(F)quinoxaline, J.
Phar-
(Lioresal ), Eur. J. Pharmacol. 52 (1978) 133–136. macol. Exp. Ther. 260 (1992) 742–747.