Influence of the growth stage of industrial hemp on the
yield formation in relation to certain fibre quality traits
V. Mediavilla
a,*, M. Leupin
a, A. Keller
baSwiss Federal Research Station for Agroecology and Agriculture,P.O.Box,CH-8046Zurich,Switzerland bSwiss Federal Research Station for Agricultural Economics and Engineering,CH-8356Tanikon,Switzerland
Received 6 January 2000; received in revised form 12 April 2000; accepted 14 April 2000
Abstract
To study the influence of the stage of growth of industrial hemp (Cannabis sati6aL.) on yield formation and fibre
morphology, a field trial was carried out in Switzerland in 1997. Different harvests took place at 7 – 14 day intervals, from the vegetative stage of growth to the senescence of the crop. Total yield and its components, fibre content and the frequency of primary and secondary fibres as well as the exact stage of growth were determined in male and female plants. Stem, bark and fibre yield reached their maximum at the time of flowering of the male plants (‘technical maturity’). Maximum stem yield amounted to 14.8 tons of dry matter (DM) per hectare. Bark yield showed a development similar to that of the stem yield and reached 5.8 tons DM/ha. Fibre yield was highly correlated with stem and bark development and also reached its maximum at the time of flowering of the male plants (yield: 4.1 tons DM/ha). During the vegetative phase, primary fibres were first created and then filled. The peak of the stem and fibre yield at the male plant flowering stage was probably caused by an increase in production and lead to a filling of secondary fibres. After that, and because of their characteristics, secondary fibres may cause a decrease in bark quality. With regard to fibre production, the upper third of the stem did not account for much fibre yield. © 2001 Elsevier Science B.V. All rights reserved.
Keywords:Cannabis sati6aL.; Fiber hemp; Bark; Growth; Harvest time; Fiber quality; Primary and secondary bark fibers www.elsevier.com/locate/indcrop
1. Introduction
Hemp (Cannabis sati6aL.) can be used in many
ways. The utilisation of fibres, seeds, oil, inflores-cences, essential oils, and secondary compounds
such as THC have been investigated. Similarly to other crops harvest time plays a considerable role in industrial hemp yield and quality (Bo`csa and Karus, 1997, pp. 104 – 130).
Different criteria for the determination of the optimal harvesting time for fibre hemp are applied in different countries: calendar date, stem colour, drop of the internodia-leaves and of course, crop growing stage (Fe´de´ration Nationale des Produc-teurs de Chanvre, 1984; Lohmeyer, 1997). With * Corresponding author. Tel.:+41-1-3777267; fax: +
41-1-3777201.
E-mail address: [email protected] (V. Medi-avilla).
monoecious varieties ‘technical maturity’ for fibre production is the peak of flowering, that is the time when most of the bracts are formed. For dioecious varieties, ‘technical maturity’ coincides with the time when most of the staminate flowers on male plants are open and occurs usually at the beginning of the bract formation on female plants (Bo`csa and Karus, 1997, p. 105).
Depending on end-use, fibre quality is defined differently: For the production of pulp and paper, no significant influence on bast fibre strength and thermo mechanical pulping handsheet strength properties could be observed until 2 months after flowering (van der Veen, 1995). However, late harvest may cause problems with the harvest ma-chinery (Lohmeyer, 1997). For the production of textiles and high-tech composites, fibre character-istics such as strength, length, diameter,
chem-istry, and homogeneity are influenced
considerably by harvest time. Schulz (1998) brought the influence of harvest time into rela-tionship with fibre yield, strength, elasticity, and the finesse of hemp fibres. Rowell and Stout (1998) reported changes in chemical composition (e.g. cellulose, sugars and lignin) and fibre charac-teristics (e.g. length and width) of jute and kenaf at different stages of plant growth. Morrison et al. (1999) describe the chemical characterisation and the retting of maturing kenaf stems.
Hemp contains three different kinds of fibres: core fibres are located inside the vascular cam-bium (0.5 – 0.6 mm long), primary bast fibres are situated outside the vascular cambium (about 20 mm long) and secondary bast fibres rise from the prodesmogen (about 2 mm long) (van der Werf et al., 1994). For long fibre uses (e.g. textiles and composites), the stem value depends primarily on the proportion of bark fibres. It is common knowledge that a high bark yield and bast fibre content as well as a low secondary bast fibre content are advantageous for the production of textiles.
Within the scope of examining the production of hemp fibres for textiles and composites we
studied an enzymatic degumming process
(Leupin, 1998). Based on this process we analysed the influence of harvest time on fibre yield and quality, stem proportion and plant sex. In this
paper we will describe the results on above-ground dry matter production and distribution as well as filling degree and the cross-section area of primary and secondary fibres. The consequences for chemical and physical fibre properties have been laid down by Keller et al. (2001).
2. Methods
Fibre hemp of the ‘Kompolti’ variety, was sown on ando-eutric cambisol soil (International Soci-ety of Soil Science, 1986) at a rate of 60 kg/ha in Hettlingen (Switzerland) near the town of Win-terthur on the 2nd of May 1997 (460 m above sea level; 8°45% E, 47°35%N). The experiment involved a complete randomised block design with four replications. In consideration of the cultural precedent (beans), nitrogen fertilisation was
re-duced to 60 kg/ha N. Weed and pest control were
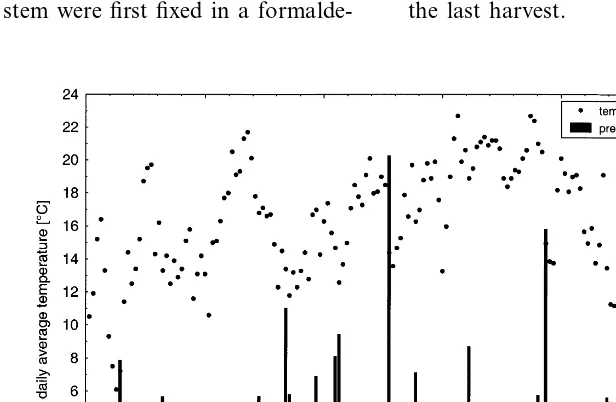
not carried out. Precipitation was well distributed over the season (beginning of May to the end of September) and reached a total level of 436 mm (Fig. 1).
With each harvest (Table 1), in every replicate,
those plant parts 1 m2
above-ground were har-vested and the number of plants was determined. For each replication, leaf and stem dry matter content was measured (sample of ten plants dried at 100°C for 24 h). For each replication, the stage of growth was determined by means of a ran-domised sample of 30 plants, according to Medi-avilla et al. (1998b) (Table 2). Crop stages were defined, assuming that 50% of plants had reached the same stage of growth.
For the determination of bark and fibre con-tent, a randomised sample of 25 plants per repli-cation was subdivided, if evident, into male and female plants — into three equal parts, namely top, middle, and bottom. The bark of all three parts was separated manually from the fresh stems and dried at 40°C. The unlignified apex was discarded. Taking into consideration plant den-sity, stem yield, the fibre content of each stem
and — if evident — the male/female ratio, fibre
The fibre content of the bark was determined in ground samples by means of a NIR Systems scanning monochromator, model 6500. Calibra-tion equaCalibra-tions were obtained with modified par-tial least squares regression (MPLS; Infrasoft International, Silver Spring, MD, USA). The cali-bration set of samples was determined gravimetri-cally by cutting 2.5 g of bark into 1 – 1.5 cm long pieces and autoclaving them in 25 ml alkaline solution (6% (w/v) KOH/0.4% )w/v) Na2SiO3/ 0.3% (w/v) Na4P2O7) at 121°C for 30 min. After cooling down, the fibres were washed thoroughly with hot and cold water, neutralised with 1% acetic acid and washed again with water, prior to being dried at 110°C.
For each replication microscopical analyses of fibres were made from a sample of five plants. Samples taken from a transverse section in the middle of the stem were first fixed in a
formalde-hyde (35%) – acetic acid – ethanol – water solution (10:5:50:35) for at least 24 h, then dehydrated with an ethanol tert-butyl alcohol series and finally embedded in paraffin (Gerlach, 1984).
Cross-sections were cut at 20 – 50 mm from fixed
paraffin embedded material at 5 mm, using a
Sorvall JB-4 hand-microtome. All sections were stained with Toluidine blue. For each harvesting date microscopical pictures of the plant cross-sec-tions were taken in triplicate. Using the photo-graphs, the amount of primary and secondary fibre cells was counted, the state of ripeness evalu-ated, their surface measured and average cell size calculated. The stage of fibre maturity was differ-entiated, depending on the degree of ripeness in low (non-filled to low-filled), middle-filled and full-filled (well filled to ripe) cells. No fibre matu-rity or cross-section areas could be determined at the last harvest.
Fig. 1. Daily average temperature and precipitation in the trial location during the period of growth (DOY).
Table 1 Harvest times
4 3
2 8 9
1 7
Harvest c 5 6
Date 14 July 28 July 4 August 11 August 18 August 25 August 3 September 17 September 1 October
Table 2
Definitions and codes of growth stages ofC.sati6aL. dioecious plants according to Mediavilla et al. (1998b)
Code Definition Remarks
Vegetati6e stage refers to main stem;lea6es are considered as unfolded when leaflets are at least1cm long 1 leaflet
1010 5th leaf pair
¡ ¡
nth leaf pair
10xx xx=2(nth leaf pair)
Flowering and seed formation refers to the main stem including branches
Change of phyllotaxis on the main stem from opposite to alternate; distance between petioles of GV point
2000
alternate leaves at least 0.5 cm Flower
2001 Sex nearly indistinguishable primordia
Male plant Flower
2100 First closed staminate flowers formation
Beginning of
2101 First opened staminate flowers flowering
2102 Flowering 50% opened staminate flowers End of
2103 95% of staminate flowers open or withered flowering
Female plant Flower
2200 First female flowers; bract with no pistils formation
Beginning of
2201 Styles on first female flowers flowering
2202 Flowering 50% of bracts formed 2203 Beginning of First seeds hard
seed maturity
2204 Seed maturity 50% of seeds hard
End of seed 95% of seeds hard or shattered 2205
3. Results and discussion
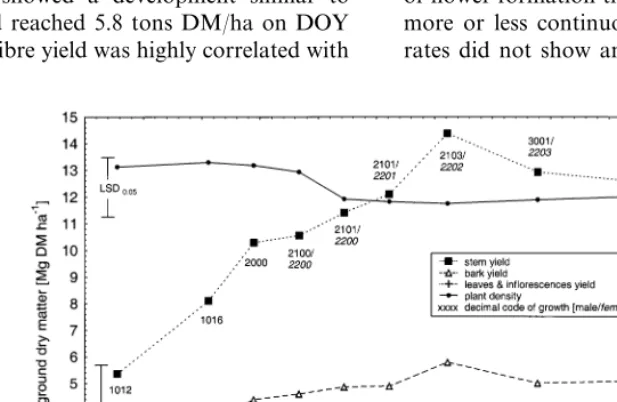
Average plant density of the different harvests was approximately 170 plants/m2(Fig. 2). This is a typical density for fibre production after day of the year (DOY) 200 for Central Europe (Meijer et al., 1995). We assume that a decrease of plant density took place before the first harvest, that is during the first phases of the vegetative stages.
Stem yield increased rapidly during the vegeta-tive growing stages (growth codes 1012 – 1016, according to Mediavilla et al. (1998b)) until the ‘GV point’ (change of the phyllotaxis) was reached on DOY 216 (code 2000). Maximum stem yield was 14.8 tons of dry matter (DM) per hectare at DOY 246, at the end of male flowering (code 2103) and the peak of female flowering (code 2202), which is described as ‘technical maturity’ (Fig. 2). After that the yield declined due to the senescence: first of the male, then of the female plants (Fig. 2). For different varieties a similar growth pattern with a maximum of stem yield at flowering was described (Meijer et al., 1995; Mediavilla et al., 1998a).
Bark yield showed a development similar to
stem yield and reached 5.8 tons DM/ha on DOY
246 (Fig. 2). Fibre yield was highly correlated with
stem and bark development and reached the
max-imum at the same time (4.1 tons DM/ha) (Fig. 3).
Approximate 54% of the fibres were located in the ‘bottom’ part, 34% in the ‘middle’, and only 12% in the ‘top’ part of the stem. These results corre-spond to those recorded by van der Werf et al. (1994) who pointed out that under comparable growth and climatic conditions, 11 – 12% of bark fibres are located in the upper 30% of the stem. During flower formation, fibre yield increase was very pronounced at the bottom of the stem (Fig. 3). After flowering (DOY 246) fibre yield decreased in the whole stem. Female plants showed a higher fibre yield than male plants, not because of a higher fibre content, but due to the higher female/
male ratio in the crop (53:47%).
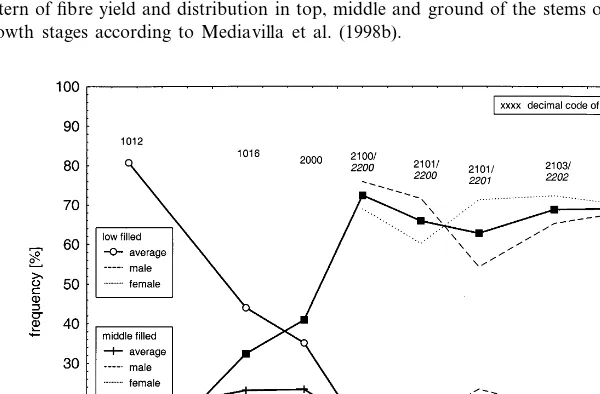
During the season of growth, primary fibres in the middle of the stem showed different degrees of ripeness. At the 6th leaf pair stage (DOY 195), 81% of all fibres were almost empty. Two weeks later, at the 8th leaf pair stage, the beginning of the fibre filling process could be recorded (Fig. 4). This was when the filling of 44% of the fibres was low and that of 33% was medium. From the beginning of flower formation the total fibres filling rate was more or less continuous. Male and female filling rates did not show any distinct pattern.
Fig. 3. Growth pattern of fibre yield and distribution in top, middle and ground of the stems of male and female plants during the growth season. Growth stages according to Mediavilla et al. (1998b).
Fig. 4. Filling degree of fibres distributed in top, middle and ground of the stems of male and female plants during the season of growth. Growth stages according to Mediavilla et al. (1998b).
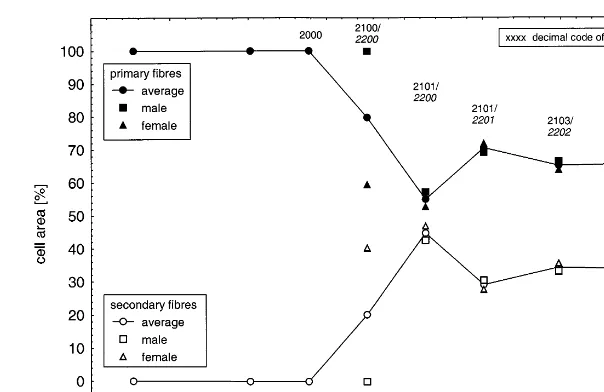
During the vegetative growth of the plants, in the middle of the stems, only primary fibres could be identified (Fig. 5). With the flowering induc-tion, secondary fibres appeared in the bark and commanded up to 45% of the fibre area at DOY 230. In contrast to male plants, after the ‘GV point’, the female plants showed an abrupt
cli-Fig. 5. Cross-section of primary and secondary fibre cells distributed in top, middle and ground of the stems of male and female plants during the season of growth. Growth stages according to Mediavilla et al. (1998b).
matic conditions — a similar primary fibre con-tent of 57 – 68% (weight) could be determined in the middle of the stem.
4. Conclusions
Stem, bark and fibre yield are clearly correlated with the growth stage of the crop. Harvesting dioecious hemp at the time of its ‘technical matu-rity’ maximises stem and fibre yield.
The process of bark fibre formation is complex and also strongly related with the growth pattern of the plant. During vegetative growth and until flower formation, primary fibres are first created and then filled. With the induction of the genera-tive phase, secondary fibres, especially at the bot-tom of the stem, are built. Peaks of stem yield and fibre yield at ‘technical maturity’ are probably caused by an increase in the production of sec-ondary fibres. Because of their characteristics (shorter, thicker walls and high lignin content (Hoffmann, 1961)) bark quality may decrease. In female plants — and probably also in monoe-cious plants — the formation of secondary fibres begins earlier and could contribute to a faster decrease of bark quality.
In terms of fibre yield, the top third of the stem did not account for much and can, therefore, be used for other purposes. In contrast to the top, the bottom of the stem is very rich in fibres and should be cut as low as possible, given the fact that it has no influence on fibre quality.
Acknowledgements
The authors wish to thank Franca Ciocco and
Nicolas Mo-Costabella for their technical
support.
References
Bo`csa, I., Karus, M., 1997. Der Hanfanbau — Botanik, Sorten, Anbau und Ernte, 1st edn. C.F. Mu¨ller, Ko¨ln, Germany.
Fe´de´ration Nationale des Producteurs de Chanvre, 1984. La Culture non Battue du Chanvre Monoı¨que. Fe´de´ration Nationale des Producteurs de Chanvre, Le Mans, France. Gerlach, D., 1984. Botanische Mikrotechnik: Eine Einfu¨hrung.
Georg Thieme, Stuttgart.
Hoffmann, W., 1961. Hanf, Cannabis sativa L. Handbuch der Pflanzenzu¨chtung, vol. 5. Paul Parey, Berlin.
Keller, A., Mediavilla, V., Leupin, M., Wintermantel, E., 2001. Influence of the growth stage of industrial hemp on chemical and physical properties of the fibres. Ind. Crops Prod. 13 (1), 35 – 48.
Leupin, M., 1998. Enzymatic degumming through alkalophilic microorganisms — a new approach for bast fibre process-ing. In: Institute of Natural Fibres (Ed.), Natural Fibres, Hemp, Flax and Other Bast Fibrous Plant — Production, Technology and Ecology, Vol. 1, Symposium, 24 – 25 Sep-tember 1998, Poznan, Poland, 119-120.
Lohmeyer, D., 1997. Die Hanfernte. Nova-Institut, Hu¨rth, Germany.
Mediavilla, V., Bassetti, P., Konermann, M., Schmid-Slem-brouck, I., 1998a. Optimierung der Stickstoffdu¨ngung und Saatmenge im Hanfanbau. Agrarforschung 5 (5), 241 – 244. Mediavilla, V., Jonquera, M, Schmid-Slembrouck, I., Soldati, A., 1998b. A decimal code for growth stages of hemp (Cannabis sati6aL.). J. Int. Hemp Assoc. 5 (2), 65, 68 – 74. Meijer, W.J.M., van der Werf, H.M.G., Mathijssen, E.W.J.M., van den Brink, P.W.M., 1995. Constraints to dry matter production in fibre hemp (Cannabis sati6aL). Eur. J. Agron. 4 (1), 109 – 117.
Morrison, W.H., III, Akin, D.E., Archibald, D.D., Dodd, R.B., Raymer, P.L., 1999. Chemical and instrumental characterization of maturing kenaf core and bast. Ind. Crops Prod. 10, 21 – 34.
Rowell, R.M., Stout, H.P., 1998. In: Lewin, M., Pearce E.M. (Eds.), Handbook of Fiber Chemistry. Malcel Dekker, New York, 465 – 504.
Schulz, J., 1998. Entwicklung ausgewa¨hlter Qualita¨tsparame-ter der Hanffaser im Wachstumsverlauf. 5. InQualita¨tsparame-ternationale Tagung Stoffliche Nutzung Nachwachsender Roh-stoffe 28 – 29 Oktober 1998, Chemnitz, Germany, pp. 29 – 38.
van der Veen, J.E.H., 1995. The effect of age on quality of hemp bast fibres as raw material for pulp and papermak-ing. In: Nova Institute (Ed.), Bioresource Hemp, Proc. of the 1st Symposium, 2 – 5 March 1995, Frankfurt, Ger-many.
van der Werf, H.M.G., Harsveld van der Veen, J.E., Bouma, A.T.M., ten Cate, M., 1994. Quality of hemp (Cannabis sati6aL.) stems as a raw material for paper. Ind. Crops Prod. 2, 219 – 227.