Pharm 202
“Digitally Enabled Genomic
Medicine” and Its Role in Cancer
Treatment
Phil Bourne
Take Home Message
• We are undergoing a revolution in our approach to treating disease
• This has been driven by the human genome project and the technologies that go with it
• A key element is the integration of information derived from genotype to phenotype
• Much of this information is now digital rather than analog • This is much more than faster ways to develop drugs – it
has to do with diagnostic treatments, preventive medicine, personalized medicine
Today -
• Overview of the revolution • Drug discovery specifically
• The much more part as it relates to cancer
– Improve the outcomes of radiotherapy in treatment of breast and prostate cancer
Approach Today
• Rather than discuss specific papers of work
completed we will take a broader perspective on proposed work on large scale projects that have the potential to impact people’s lives through digitally enabled genomic medicine
Cell Biology
Cell Biology
Anatomy
Atoms & Molecules
Atoms & Molecules
SCIENTIFIC RESEARCH & DISCOVERY REPRESENTATIVE
DISCIPLINE
EXAMPLE UNITS
Migratory
Migratory
Sensors
REPRESENTATIVE TECHNOLOGY
Technologies
Digital vs Analog
• The lower levels of biological complexity have always been digital – the higher levels analog
• This made it very hard to correlate across biological scales
Lower Levels – Digital (sort of)
This digital image of cAMP dependant protein kinase
(PKA) depicts years of collective
knowledge.
We can only
interpret it in this form and the
Higher Levels – The Patient Record
• 8% of patient records are lost • They are mostly paper (analog)
• They can only be interpreted by humans • Errors are rampant
• There are exceptions – tumor registries,
Drug Discovery as an Example of
this Revolution
• Requires a higher level of digital enablement
Discovery and Development
• Discovery includes: Concept, mechanism, assay, screening, hit identification, lead
demonstration, lead optimization
• Discovery also includes in vivo proof of concept in animals and concomitant
demonstration of a therapeutic index
Discovery and Development
• The time from conception to approval of a new drug is typically 10-15 years
• The vast majority of molecules fail along the way
Drug Discovery - Status Today
• Somewhat digitally enabled (FDA still requires paper submission)
• Will benefit from emergent technologies • Human targets are relatively well defined • Process for finding appropriate targets in
other organisms is evolving
Drug Discovery Processes Today
Primary Assays Biochemical Cellular
Pharmacological Physiological
Sources of Molecules Natural Products Synthetic Chemicals Combichem
Biologicals
+
Initial Hit Compounds
Drug Discovery Processes - II
Initial Hit Compounds
Secondary Evaluation - Mechanism Of Action
- Dose Response
Initial Synthetic Evaluation - analytics - first analogs
Hit to Lead Chemistry - physical properties -in vitro metabolism
First In Vivo Tests
Drug Discovery Processes - III
Lead Optimization
Potency Selectivity
Physical Properties PK
Metabolism
Oral Bioavailability Synthetic Ease Scalability
Pharmacology Multiple In Vivo Models
Chronic Dosing Preliminary Tox
Development Candidate
Remains Serendipity
• Often molecules are discovered/synthesized for one indication and then turn out to be
useful for others
– Tamoxifen (birth control and cancer)
– Viagra (hypertension and erectile dysfunction) – Salvarsan (Sleeping sickness and syphilis)
Issues in Drug Discovery
• Hits and Leads - Is it a “Druggable” target? • Resistance
• Pharmacodynamics and kinetics • Delivery - oral and otherwise
• Metabolism
What has changed in
What has changed in
identifying targets?
identifying targets?
In principle we know all the
In principle we know all the
human targets -
human targets -
human genome
polysaccharides lipids nucleic acids proteins
Problems with toxicity, specificity, and difficulty in creating potent inhibitors
human genome
polysaccharides lipids nucleic acids proteins
proteins with binding site
Relating druggable targets
Relating druggable targets
to disease...
to disease...
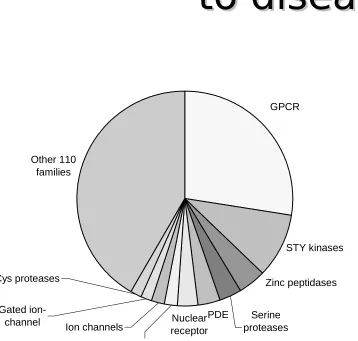
GPCR
STY kinases
Zinc peptidases
Serine proteases PDE
Other 110 families
Cys proteases
Gated
ion-channel Ion channels Nuclear receptor P450 enzymes
Analysis of pharm industry reveals:
• Over 400 proteins used as drug targets
• Sequence analysis of these proteins shows that most targets fall within a few major gene families
(GPCRs, kinases, proteases and peptidases)
Remaining issues
• Characterization of human proteins is on-going (see each revision from Ensembl) • Our ability to locate coding regions is
improving
• Our ability to annotate putative proteins is improving
The Structural Genomics Pipeline
(X-ray Crystallography)
Basic Steps
Target Selection
Crystallomics
• Isolation, • Expression, • Purification, • Crystallization
Data
Functional
Annotation Publish
Anticipated Developments
Bioinformatics
• Distant homologs • Domain recognition
Automation Bioinformatics
• Empirical rules
Automation Better sources
Software integration Decision Support
MAD Phasing Automated fitting
Bioinformatics
• Alignments • Protein-protein interactions • Protein-ligand interactions
• Motif recognition
From Structural Genomix
• FAST™ is a proprietary lead generation technology developed by SGX for identification of novel, potent and selective small molecule
inhibitors of drug targets within a rapid six-month timeframe. The
FAST™ process involves crystallographic screening of lead-like drug fragments followed by structure-guided elaboration of the fragments by parallel chemical synthesis, guided by proprietary computational tools. Iterative determination of crystal structures for multiple
target/compound complexes in parallel with assays, computational design and synthesis results in optimized leads with high binding affinities and low molecular weights. The combinatorial nature of
FAST™ provides access to expansive chemical diversity in the order of 160 million compounds, while requiring only a small number of compounds to be synthesized and screened. Thus the FAST™
Summary
• Need information flow from genotype to phenotype and back
• Digital enablement provides that
• The human genome and the associated technologies has accelerated this process dramatically
• Example – human genome provides more targets • Example – structural genomics leads to faster
identification of leads