160 Hatta Med J Indones
Evaluation
of anti-phenolic
glycolipid-I
IgM
and
CD4ICD8
T
cell
subsets
value
as high
risk
determination
indicator
for individuals
residing
in
a
leprosy endemic area in Indonesia
Mochammad Hatta
Abstrak
Penelirtan ini menilai antibodi IgM terhadap Phenolic Glycolipid-I (PGL-I) M. leprae dan jumlah subset sel T (CD4 dan CD8) untuk menentulcan derajat resiko mmdeita kusta di antaro individu melalui survei penduduk di daerah endemik Kusn. Di antara 2349
dari 3074 pendu.duk yang diperksa, angla seroposifif pado penduduk tersebut secara umum dan kontak senanah sangat tinggi dan ditemukan secara berturut-turut sebesar 40,27o dan 50,4Vo. Angka rata-rata CD4 dan CD8 dari subset sel T pada penduduk secara wnunt ditemal<an berturut-turut sebesar 849
t20
dan 646+
19. Angka ratct-rata CD4 dan CD8 dari subset sel T pada kontak serumah ditemulcan berturut-turut sebesar 785t
t3
den 572+
15. Tidak ada perbedaan bermalon dalatn jumlah subset sel T (CD4 dan CD8) antara penduduk secara wnum dan kontak serumah. Pada penduduk secara umum, angka rata- rata CD4 dan CD8 pada individu dengan antibodi IgM terhadap PGL-I positif berbeda secara bermakna dibandingkan dengan pada individu dengan antibodi IgM terhadapPGI-I
negatif. Pada penelitian ini antibodi IgM positif tidak selalu menggambarlan manifestasi klinis kusta, namun penemuanIgM
positifdkerni
rendahnya jumlah CD4 dart subset sel T masih mungkin menunjang teori tentang resiko tinggi untuk meniadi kusta tipe lepromatosa.Abstract
This study reports the evaluation of the presence of IgM antibody to Phenolic Glycolipid-I (PGL-I) of M. leprae andT cell subsets (CD4 and
CD|)
count to determine the high risk to contract leprosy among individuals through a total PoPulntion survey in an area where leprosy is endemic, in Indonesia- Among the 2349 individuals of the total population of 3074 people, the seropositive rates in general inhabitants and househald contacts were very high (40.2Vo and 50.4Vo, respectively). The mean CD4 and CD8 T cell subset caunt in general inhabitants was 849t20
and 646+
19, respectively. The mean of CD4 and CDB T cell subset count in household contacts was 785!
t3 and 572a
15, respectively. No statistically significant dffirences were found in peripheral blood CD4 and CD8 count between general inhabitants and household contacts. In general inhabitants the mean CD4 and CDB count showed significant diference between individuals with positive IgM antibodics to PGL-I compared to individuals with negative IgM antibodies to PGL-|. Although individuals with positive IgM antibodies toPGLI
might not sufering clinical manifestation of leprosy, the frnding of positive IgM and law number of CD4 count srtil support the theory of a high risk to contact lepromatous leprosy.Keywords: general inhabitant, household contact, lepromatous leprosy, tuberculoid leprosy
Leprosy
is
a
chronic infectious
disease caused by
Mycobacterium leprae.
The
disease
has
a
broad
spectrum
of
clinical
manifestation
from
the
tuber-culoid
form, with low
bacillary load,
to
the
lepromatous
form, with high bacillary load, and
may
lead toirreversible physical deformities
and extensivedamage
of
peripheral
nerves.
The
spectrum
of
this
disease
activity
is
best classified
to five
groups
asfollows:
full
tuberculoid (TT), Borderline
tuberculoid
(BT), mid
borderline
(BB),
borderline
lepromatous(BL), and
full lepromatous
(LL).t
Department of Medical Microbiology, Faculty of Medicine, Hasanuddin lJniversity, Ujung Pandang, Indonesia
In TT
and
BT
leprosy
the
T
lymphocytes
of
CD4+
subset arethe
most abundant
lymphocytes
which
aretypically
present
in the
central
region of
the
granulomas,
while
the CD8+
T
lymphocytes
are present as amantle
around the epitheloid structures.A
characteristic serologic feature
of TT and
BT
leprosy
is
low
antibody
response to M. Ieprae.demons-Vol 8, No 3, July - September 1999
trated
the
homogeneity
of
seropositivity
among
populations
at village
level
in
South Sulawesi,
In-donesia, and suggested
that seropositivity
in
apopula-tion
meant anendemic
exposure toM.
leprae.
Indeed,we
alsofound
evidenceof
cluster
of
leprosy in certain
villages.a'5The
subsetof
CD4+Tcell
becamecentral
toconsidera-tions
of
protection
andpathology
in
human
disease.Therefore
we
analyze
the
correlation
between
positivity of
IgM
antibodies
to
Phenolic
Glycolipid-I
(PGL-D
ofM.
leprae
andT cell
subset(CD4
andCD8)
count to determine
therisk
factors
amongindividuals
through
a total population survey
in
an
area
whereleprosy is endemic,
in
Indonesia.
METHODS
Field study
An
isolated
village
in
arural
areaof
South
Sulawesi,Indonesia,
with
3074 inhabitants was
selectedfor
thesurvey. The leprosy
prevalence
was
expected
to
be 0.6411,000on
the basisof
available
information from
the local
health service.
Before
the study
was under-taken, theindividuals of
thevillage
wereinformed
onthe purpose
of
the
study, and written
consent
wasobtained
from
all participants.
Only individuals living
in
thevillage for
at least threemonths
andgiving
their
consent rvereincluded
in
the
study.Clinical examination
was
carried out by
experiencedleprosy
worker
anddiagnosis
of
leprosy
was based onthe
classification
of
Ridley and Jopling.t
Slit
tkin
smears
were taken
from
earlobes and a
samplefrom
one lesion at
least,
from all
leprosy
patients
for the
determination
of
thebacterial index
(BI).
Blood collection
Blood
samples
were collected
by
venipuncture
(1.0
ml).
Samples werecentrifuged
3000rpm for
5minutes
to
separatethe
serum and transfèred
to 500
pl vials
(Sarstedt,
Assist Trading
Co.,
Ltd.)
on the
same day.All
serawere
storedat
-4oCfor further
analysis.The
samples
collected
from individuals were
coded,and serological tests were performed without prior
knowledge
of
theclassification of
the samples.MLPA
test
The
Serodia-leprae
microtitre particle
agglutination
testkit
for
thedetection
of anti-PGL-I
antibodies
was purchasedfrom Fujirebio
Inc.,
Japan.6Anti PGL-I IgM and CD4/CD8 as leprosy risk
indicator
161Serum samples were
diluted to
I : 16 and l:32in 96well
U-bottom
microplates.
Twenty five pl of
unsensitized
gelatin
particle
and25
pl
of
the
particles
sensitizedwith
synthetic trisacharide ofPGL-I (NT-P-BSA)
weremixed
with
25
pl of
l:16
and
25
1tl
l:32
diluted
samples, respectively.
After
being incubated
for
2hours
at
room
temperature,
the plate was read for
agglutination using a mirror
reader.
Serum
samplesshowing agglutination at
atitre
of
> 1:32 were
con-sideredpositive.
Positive
serawere
serially diluted
to
aconcentration
of
l:1024 for
quantitative testing.
ELISA
IgM
andIgG
antibodies toPGL-I
andLAM-B
in
serumwas
measured
by
indirect
ELISA.
An
ELISA
plate
(Nunc-Immunoplate Maxsorp
F96) was coatedwith
50pl
of PGL-I or
LAM-B
solution
(0.1
pg/ml in
carbo-natebuffer, pH 9.6),
andthen the
free
surface
of
thewell
wasblocked by incubation
with
17oBSA
at3Jo Cfor
60 minutes. The
testserum,
diluted
to
l:300 with
dilution buffer (PBS
with
20Vofetal
calf
serum
and0.5% Tween 20),
was addedto
thewell
andincubated
ar- 37o
C
for
60 minutes.
After rinsing
the
plate with
washing
buffer,
peroxidase-conjugated
anti-human
IgM
or IgG
antiserum
(Dako immunoglobulins A/S)
was
addedto
the
well
andincubated
at
37oC
for
60minutes.
After
the
excess
conjugate
was
removed
using washing
buffer,
substratesolution (0.4 mg
of
o-phenylenediamine
per
ml
and
0.4
pl
of hydrogen
peroxide
perml in
0.1M
citrate-phosphate
buffer,
pH
5.0) was added and the plate was developedin
the darkfor
15to
30 minutes. The reaction was stopped
with
1.25M
sulfuric
acid. The
absorbance was
then
measuredwith
amicroplate photometer
at490
nm.Counting of CD4/CD8 T cell
subset
The counting
of
CD4|CD8
T
cell
subsetwas
estab-lished
in
Dynabeads
T4-T8 Quant (Dynal
kir,
Oslo,
Norway).
Briefly,
0.5
ml of
blood was collected
by
venipuncture
and countsof CD4/CD8
wereperformed
atthe
same day.The
DynabeadsT4-T8
kit uses
mag-netic particles
coatedwith
antibodies
to
theCD4
andCD8
antigen
to
capture and
isolate T4
and
T8
lym-phocytes
from
whole blood. The isolated
cells are
lysed
and counted
directly
in
haemocytometer after
staining
with
Sternheimer-Malbin solution.
First
250pl
blood
in EDTA
wasrotated
for
2minutes
at
room
temperature
and225
pl
washing/dilution
buffer
was
added.
For
monocytes
depletion
25
1tlDynabeads
anti
CDl4
was
addedand incubated
in
aplaced
in the Dynal MPC-Q for
2minutes and
carefully
200pl
of monocyte-depleted blood was
transferedinto
eachof two
supplied microtubes.
The tubecontaining
the rosettedmonocytes
was disposed.For
the
isolation
of CD4/CD8
T cell
subset,into
oneof
the two
microtubes containing 200
pl monocyte
deplesedblood,
200pl washing/dilution buffer
and 25pl
Dynabead
CD4
was added,
andto
the other
25 pl
Dynabead
CD8. The tubes were
capped and mixed
carefully
by
turning
and incubatedfor
10 minutes. Thetubes
wereplaced
in
theDynal MPC-Q for
2 minutes andthe
supernatantwas discarded. The isolated cells
were washedby adding
500pl
washing/dilution
buffer
and placedin
theDynal
MPC-Q for
afew minutes
andthe washing
step was repeated once.
The cells
wereresuspended
in 80
pl
lysis buffer
and
vortexed,
and leavedfor 5
minutes.Again
the cells were vortexed andthe tubes
were
leaved
in
the
MPC-Q
for
I
minute
to
separate
the Dynabeads from nuclei. Twenty pl
Sternheimer-Malbin staining solution
was
added and thenuclei were counted
in
a haemo.cytometer.Data analysis
All
datawere recorded on
specialforms
and analysedusing a statistical software
package(Epi-Info,
version
6.02)
in
appropriate hardware. Statistical
analysis wasapplied
asindicated
in
theresults.
All
P presented aretwo-tailed P
value.
RESULTS
Sample
sizeand incidence rate
At this
survey,
from
3047people that were
registered a totalof
2349 inhabitants
(7 6Voof
registeredpopula-tion)
met the
inclusion criteria
and wasclinically
ex-aminedfor
signsof
leprosy. Twenty nine new leprosy
patients were detectedduring
the survey and 2 patientshad been
identified
years
before by the local
health
service,giving
a totalof
31leprosy patients
(20 malesand
1l
females),
ranging in
agefrom
12to
67
years. Based onthe
slit skin
smearresults
andclinical
diag-nosis,
27 patients
were classified
astuberculoid
andfour as lepromatous (bacterial indices
of slit skin
smears
ranging
from 1.0 to 4.0). There was a
sig-nificant
difference
in leprosy
patients between
males andfemales (P < 0.05). The indicende
rate among thepopulation was thus
10.0/1000population.
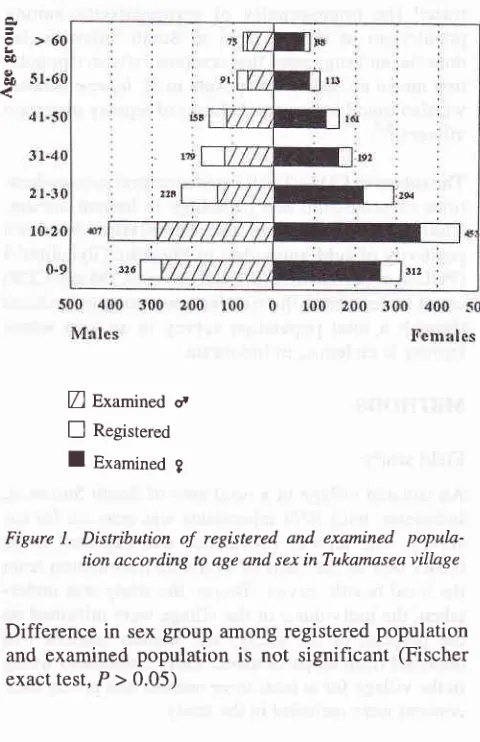
The male : female
ratioin
the examinedpopulation
was0.82 compared
with
0.90
in
the registered population
(Figure
l).
Med J Indones
Z
Examineda
E
Registered [image:3.595.342.582.89.460.2]I
Examined gFigure 1. Distribution
of
registered. and examined popula-tion according to age and sex in Tukamasea villageDifference in
sexgroup among registered population
and examined
population
is not significant
(Fischer
exacttest,P>0.05)
Seropositivity rate to M. leprae
related
antigens
in
general
inhabitants
and
household
contacs
As it
wasnot feasible to test
all individual
samplesin
this
survey
by Mycobacterium leprae particle
agglu-tination
MLPA test and ELISA, we examine
1483 generalinhabitants (NC
group)
and73
householdcon-tacts
(HC
group).
As
shown
in Table
1, usingELISA
to
detect
anti PGL-I IgG antibody, the seropositivity
rate
in male
and
female in NC group was
7.8Vo and6.4Vo,rcspectively. However,
the rateof IgM
positive
in
male and female inNC
group was3l
.1Vo and46.5Vo,respectively.
The
rate
of IgIrI positive in female
wassignificantly higher than
in
male
in
the
NC
group
(Fischer
exact test,P < 0.01).
Using
ELISA to
detect anti
LAM-B IgG
antibody,
nodifference
wasfound in seropositivity
rate
of LAM-B
IgG
betweenmale
andfemale
in
both
theNC
and theHC group (Fischer exact test,
P > 0.01), while
theseropositivity
rate
of PGL-I IgM in female was
sig-nificantly
higher thanin
malein the HC group (Fischer
exacttest,P<0.01).
Vol 8, No 3, July - September 1999 Anti PGL-I IgM and CD4/CD8 as leprosy risk indicator r63
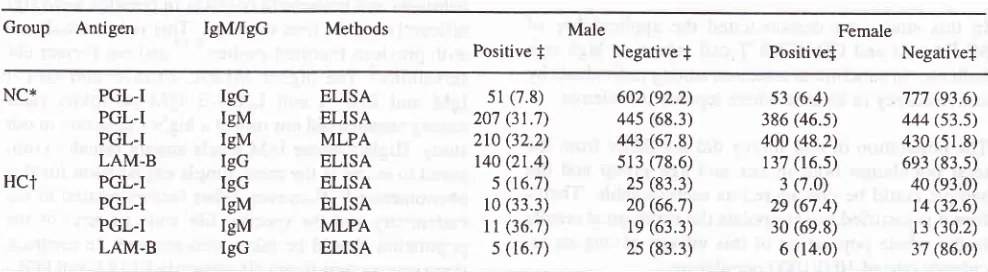
Table
l. MLPA
and ELISA results using M. Ieprae antigen in general inhabitants and household contactsGroup Antigen IgM/IgG Methods Male
Positive
f
Negativef
Female
Positivef
NegativefNC* HCT PGL-I PGL-I PGL-I LAM-B PGL-I PGL-I PGL-I LAM-B IgG IgM IgM IgG IgG IgM IgM IgG ELISA ELISA MLPA ELISA ELISA ELISA MLPA ELISA 51(7.8) 207 (31.7) 210 (32.2)
t40 (21.4) 5 (16.7)
l0
(33.3)l1
(36.7) s (16.7)602 (92.2) 445 (68.3)
443 (67.8) s13 (78.6) 2s (83.3) 20 (66.'t)
le
(63.3) 2s (83.3)s3 (6.4) 386 (46.5) 400 (48.2)
137 (r6.s)
3 (7.0) 2e (67.4) 30 (69.8) 6 (14.0) 777 (93.6) 444 (s3.s) 430 (51.8) 6e3 (83.s) 40 (e3.0)
t4 (32.6) r3 (30.2)
37 (86.0)
* NC = General Inhabitants, HCt
-
Household contacts, fAbsolute number (7o), Cut off for positive to PGL-I IgG (ELISA) = 80, PGL-I IgM(ELISA) = 380, PGL-I IgM (MLPA) = 32, LAMB IgG (ELISA) = 250
Using MLPA
test todetect anti
PGL-I
IgM
antibody,
the
seropositivity rate
in
female was significantly
higher
thanin
male
in
both
the HC and the NCgroup
(Fischer exact
test,P < 0.01).
In
addition,
no
significant difference was found in
seropositivity
rate between
HC group and
NC
group
by
ELISA
or
MLPA
(Fischer
exact test,P
>0.01).
Number of CD4 and CD8 T cell
subset
in general
inhabitants and household
contacts
Among
the 1483 generalinhabitant (NC group) and73
household contact
(HC
group) samples tested
by
Dynabead
method,
the mean ofCD4
and CD8 countin
the
NC group
was 849+20
and646
+
19,respectively;
while CD4/CD8 ratio
in
the
NC
group was
1.45
+0.058.
The
mean
of
CD4
and
CD8 count in
the
HC
group
was785
+ 13
and5721
15,respectively;
while
CD4|CD8 ratio in the HC
group was
I.44
+
0.074.No
statistically significant
difference
in the mean of
CD4
and
CD8 count was found
between
the two groups
(Student's t
test, P> 0.01) (Table
2).Table 2. CD4 and CD8 count in general inhabitans and
house-hold contacts
Group
Correlation
between
seropositivity and CD4
and
CD8 count
The coirelation
between
seropositivity
and
CD4
andCDS
count is
shown onTable
3. There was significantdifference
in the meanof CD4
andCD8 count
betweenanti PGL-I
IgM
seropositive and
seronegative
(Student's t
test, P<
0.01).However,
there
was no significant difference
in
themean of
CD4
andCD8 count
betweenanti
PGL-I IgG
seropositive and seronegative (Student's test,
P
>0.0r).
There was no
significant difference in
the mean ofCD4
and
CD8
count between antiLAM-B
IgG
seropositive
and seronegative
(Student's
t test,
P
>0.01).
Table 3. Corelation between seropositivity and CD4 and CD8 count
Antibodies Positive/Negative
cD4
count*
CD8
CD4/CD8count*
ratio*CD4 count* cD8 count* CD4/CD8 ratio*
Anti PGL-I IgM (+) Anti PGL-I IgM (-)
P valuel
Anti PGL-I IgG (+)
Anti
PGLI
IgG (-) P value Anti LAMB IgG (+) Anti LAMB Iec G)P value
949 +
lo*
735 +16
1.28 + 0.06635+12 528+9
l.2l +0.09
sign
sign
ns893
+26
683 +14
1.35 +0.10 855 +14
648 +15
1.46 + 0.08ns
ns
ns821 +
l7
644 +23 L35 + 0.05867 +
28
658 +27
1.47 + 0.08ns
ns
nsGeneral Inhabitans Household contacts
849
t2O
646t19
785 +
13
572 + 151.45
t
0.058 1.44 + 0.074* Mean
t
SDNo statistically significant difference in CD4 and CD8 count between
the wo groups
x
MeanlSD (of the number or ratioof
T
cell subset, usingDynabeads method)
t
The difference between individuals with positive and negative antibodies, in the number of CD4/CD8 count and CD4/CD8 ratio was measured by student's t test. sign = significant, [image:4.595.76.570.100.236.2]164 Hatta
DISCUSSION
In
this
study, we
demonstrated
the applicability
of
MLPA
test
and
CD4/CD8
T
cell
subset
ashigh risk
indicator
in
subclinical
infection
amongindividuals by
active survey
in
an areawhere leprosy is
endemic.The
population in this
survey
did not differ from
thetotal population both
in
sex and
age
group
and
thesample
could be
considered
as representable.There-fore
it
isjustified
to extrapolate the serological
resultsto
the
whole population
of
this village, giving
anin-cidence rate
of
10.0/1000population.
In this
survey, we
found
that
theseropositivity
rateto
PGL-I
IgM
antibody
by MLPA
test among
generalinhabitants
was4O.2Vo.Inaprevious
survey,inthe
areaof
South Sulawesi, Indonesia,
we found that
theseropositivity rate
by
the-same
test among
generalpopulation
was
31.9Vo.2
The results
of
other
epidemiological
studiesperformed
in North
Sulawesi
also showed that
patient
topatient
contact is themayor
determinant
in
leprosy incident, with implication for
future control. The
above study revealed the contact
with a
leprosy patient
is
the major determinant in
leprosy
incident,
whereby the contact
wasnot
limited
only to
household relationships,
but
also
includes
neighbour and social
relationships.'
Thus, many
nor-mal
persons
in
the
community
in
an
area
of
high
leprosy
prevalence
have
increased
anti PGL-I IgM,
presumably
as a consequenceof
previous subclinical
infection. Our
resultshowever
revealed that manysub-jects with positive anti PGL-I
IgM
did not
show
anyclinical
sign. Therefore,
onecan hypothesize that
themajority
of
individual
exposed toM.
Ieprae can defend themselveseffectively
against thedevelopment
of
thedisease.
However,
it
remains unclear
why
in
someindividual
the
diseasecould
develop progressively to
lepromatous.
It
might be
suggestedthat the clinical
manifestation of this
diseasemight
be determinedby
acomplex
of
immune
mechanisms,
involving
the
mechanism
by which
this bacteria
evadedor triggered
the
cell
mediated orhumoral immune
response.o Inthis
study, we found that
the meanof CD4
andCD8 count
in
anti
PGL-I IgM
seropositive was significantly
higher
thanin
the
seronegativeindividuals. While
theimmunological
significance
of thiscorrelation
remains obscure,this
study revealed thatin
mostof tuberculoid
type
of
leprosy that
was
found by
active survey they
have high cellular immunity
to
M.
leprae. Thus,
it
seems
that
cell
mediated
mechanisms
of
immunity
rather
thanhumoral
are associatedwith
theprotection
and
clinical
statusofdisease.
Med J Indones
In
this
study,
the
seropositivity
rate
in
general
in-habitants
andhousehold
contactsin
females were
sig-nificantly
higher than
in
males.
This is in
with previoui
reported
studies,9'10 andour
servations.-'
The higher
MLPA,
ELISA,
IgM
and
ELISA
anti
LAM-B IgM
positivity
rates among femalesdid not reflect
ahigher
case ratein
our
study.
Higher
innate
IgM
levels
amongfemales
com-pared
to
males_is themost simple explanation
for
this
phenomenon.t'
Ho*eu".,
other
factors related
to
theendemicity
and
the specific
life
style
(if
any)
of
thepopulation
should be taken
into
account.
In contrast,
there was no
significant
difference
inELISA
antiPGL-I
IgG
or
ELISA
anti LAM-
B IgG
between males
and females.Our
previously
described
MLPA
testwas suitable
for
large
population in
clustering
of
MLPA
ng
thepopulation.a
Several reports indicated that the predominant
anti
PGL-I
responsein
most humanleprosy patients
wasof
the IgM type,"
andthat anti PGL-I
IgM
levels
werecorrelated
with
diseaseclassification, increasing
from
the tuberculoid toward the lepromatous pole
of
thedisease
spectrum.l3
To
better understand thecorrelation of
IgM
antibodies
and
CD4 and
CD8
count
with
the
maintenance
of
subinfection,
we
applied
MLPA
test
and Dynabead
method
through a
survey and
we
will
reexamine
the sameindividuals
living in
an areain which
leprosy
is endemic.CONCLUSION
In
generalinhabitants
the meanof CD4
andCD8 count
showed
significant difference between individuals
with
positive
IgM
antibodies
to
PGL-I
compared to
individuals
with
negative
IgM
antibodies
to
PGL-I.
Although individuals
with positive
IgM antibodies to
PGL-I might
not
suffering
clinical manifestation
of
leprosy, the
finding of positive
IgM
andlow
number
of CD4
count still support
thetheory
of
ahigh risk
tocontract lepromatous leprosy.
Acknowlegments
We thank
Dr.
Paul
R
Klatser,
Dr.
Stella M van
Beersand
Dr.
Shinzo
Izumi for critical
reading
of
themanuscript.
We would like to
expressour
gratitute
to
Vol 8, No 3, July - September 1999
Leprosy
Unit
of
the
Ministry
of
Health, Republic
of
lndonesia
for their
cooperation. The
financial
support
of
TheNetherlands Leprosy Relief Association
(NSL)
and
Pfeizer Health
ResearchFoundation
of
Japan isgreatly
appreciated.REFERENCES
l.
Ridley DS, Jopling WH. Classification of leprosy accordingto immunity: a fir,e-group system. Int J lepr 1966;34:255-73. 2. Hatta M, Izumi S, Klatser PR. Evaluation of Mycobacterium leprae particle agglutination (MLPA) test as a tool
in
theepidemiology of leprosy in high prevalence village in South
Sulawesi, Indonesia. Southeast Asia J Trop Med Pub Health 1995;4:631-5.
3.
Izumi
S,
Hatta
M,
Kawatsu
K,
Matsuoka
M.
Seroepidemiological study
of M.
Ieprae infectionin
theinhabitants
of
endemic villagesin
South Sulawesi,In-donesia. Int J Lepr 1995;63:650.
4. Hatta M, van Beers SM, Madjid B, Achmad D, de Wit MYL,
Klatser PR. Distribution and persistence of Mycobacterium
Leprae nasal carriage among a population in which leprosy
endemic
in
Indonesia.Trans
R
SocTrop
Med
Hyg 1995;89:38 1-5.5. Hatta M, Izumi S. Present situation of leprosy in Indonesia
and
future
prospect:Molecular epidemiological
andSeroepidemiological study
of
leprosyin
South Sulawesi, Indonesia. Int Sci Meeting of International Epidemiological Association; 1996 August 27-30; Nagoya, Japan.6. Izumi S, Fujiwara T, Ikeda M, Nishimura
Y,
Sugiyama K, KawatsuK.
Novel gelatin particle agglutination tests forAnti PGL-I [gM and CD4/CD8 as leprosy risk
indicator
165serodiagnosis
of
leprosyin
thefield.
J Clin
Microbiol1990;28:525-9.
7. van Beers SM, Hatta M, Klatser PR. Patients contact is the
major determinant in incident leprosy: Implications for
fu-ture control. Int J leprosy. I 998 submitted.
8. Ir{uthukkarupan VR, Chakkalath HR, Malarkannan S. The classical and alternative pathways of
T
cell activation areimpaired in leprosy. Immunol Lett 1982;19:55-8.
9. Fine PE, Ponnighaus JM, Burgess P, Clarkson JA, Draper
CCM. Seroepidemiological studies
of
leprosyin
norhernMalawi base
on
an enzym-linked immuno-sorbent assayusing synthetic glycoconjugate antigen.
Int
J
Lepr1988;56:243-54.
10. Ulrich
M,
Smith PG, Sampson C,ZunigaM,
Centeno M, GarciaV,
et al.IgM
antibodiesto
native glycolipid-l in contacts of leprosy patients in Venezuela: epidemiological observation and a prospective study of the risk of leprosy. Int J Lepr l99l;59:405- 15.11. Maddison SE, Stewart CC, Farshy CE, Reimer CB. The relationship of race, sex and age to concentrations of serum
immunoglobulins expressed in intemational units in healthy
adufts in the USA. Bull WHO 1975:52:l'79-85.
12.
Levis WR,
MeekerHC,
Schuller-LevisG,
Sersen E,Schwerer B. IgM and IgG antibodies to phenolic glycolipid-l
lrom Mycobacterium leprae in leprosy: insight into patient monitoring, erythema nodosum leprosum and bacillary per-sistence. J Invest Dermatol 1986;86:529-34.
13. Brett SJ, Payne SN, Gigg J, Burgess PJ, Gigg R. Use
of
syntheti
c
glycoconj u gates containing
Mycobacteium
Ieprae specific and immunodominant epitope
of
phenolic