© 2017 Fitra Yosi, Sofia Sandi and Miksusanti. This open access article is distributed under a Creative Commons Attribution Original Research Paper
The Visceral Organ, Gastrointestinal Tract and Blood
Characteristics in Pegagan Ducks Fed Ration Fermented by
Tape Yeast with Different Moisture Content
1Fitra Yosi, 1Sofia Sandi and 2Miksusanti
1Department of Animal Science, Faculty of Agriculture, University of Sriwijaya, South Sumatera, Indonesia 2Department of Chemistry, Faculty of Mathematics and Natural Sciences,
University of Sriwijaya, South Sumatera, Indonesia
Article history
Received: 10-02-2017 Revised: 19-05-2017 Accepted: 18-08-2017
Corresponding Author: Fitra Yosi
Department of Animal Science, Faculty of Agriculture, University of Sriwijaya, South Sumatera, Indonesia
E-mail:fitrayosi@unsri.ac.id
Abstract: The purpose of this study was to assess the effect of administration diet containing locally sourced materials fermented by tape yeast with different moisture content on the weight of the visceral organs, gastrointestinal tract and blood chemistry and profile of Pegagan ducks. As many as 200 Pegagan ducks aged 3 days were kept for 6 weeks in colony cages. This study used a completely randomized design that consisted of 5 treatments and 4 replicates. The treatments tested were symbolized as P1, P2, P3, P4 and P5 based on the level of moisture content used in the fermentation process of ration, namely control, 40, 50, 60 and 70%. Data were analyzed by analysis of variance continued by Duncan’s multiple range test at 5%. The weight of gizzard, pancreas, crop-oesophagus, proventriculus, the total of small intestine and ceca were significantly (p<0.05) affected by treatment, but did not significantly affect (p>0.05) on the weight of bile, liver, spleen, heart, duodenum, jejunum, ileum and rectum. Furthermore, blood cholesterol, triglyceride and LDL of Pegagan ducks were significantly (p>0.05) different. It can be concluded that the fermentation process of locally sourced ration using a moisture content of 50% gives the optimal result on the weight of visceral organ, gastrointestinal tract and also blood chemistry and profile of Pegagan ducks.
Keywords: Blood, Gastrointestinal Tract, Moisture Content, Tape Yeast,
Visceral Organ
Introduction
The use of local ingredients in reducing the cost of feed is widely utilized. However, their use for poultry is limited because of a high crude fiber content. This is due to poultry has limitation to digest crude fiber, such as cellulose and hemicelluloses (Yosi et al., 2016). Therefore, it needs a feed processing technology, such as fermentation, to reduce the fiber content and increase the nutrient digestibility. Yeast is a natural ingredient that is widely used as an inoculum in fermentation processes of poultry feedstuff (Khempaka et al., 2014; Sandi et al.,
2016; Yosi et al., 2016). In addition, the yeast is also used as a feed supplement in poultry rations that can stimulate the growth process (Yalçın et al., 2013). The use of yeast in the feed has been widely studied and have a significant effect on performance and physiological responses to poultry, such as increasing
the immune response (Asli et al., 2007; Yalçın et al., 2010; Haldar et al., 2011), lowering the blood cholesterol levels (Yalçın et al., 2010) and increasing the digestibility of nutrients (Gao et al., 2008).
One type of yeast that could potentially be used is the tape yeast (Yosi et al., 2016). The tape yeast contains
Consequently, aeration and mass transfer processes in the metabolism of microbes are inhibited.
By knowing the optimal moisture content in the fermentation process of locally sourced rations, it is expected that the growth of S. cerevisiae to be better so that the nutritional digestibility of the ration is increasing. It is known that the high nutrient digestibility will affect the activity of visceral organs and digestive tract, which can be measured by weight changes (Hetland et al., 2005). In addition, this will also affect the increase of erythrocyte numbers, hemoglobin and hematocrit (Huff et al., 2010), as well as stimulate the immune system to generate more antibodies (Shanmugasundaram et al., 2013). Based on this, further studies to find out the effect of feeding the local ration fermented by tape yeast with different moisture content on bird’s performances need to be implemented.
Materials and Methods
Birds, Feed and Housing
A total of two hundred of Pegagan ducks aged 3 days was allocated into 5 treatments of experimental used only for illumination at night. Rations used is feed by using local raw materials, including refined corn meal, palm kernel cake, coconut pulp, cassava leaf meal, water hyacinth meal, lamtoro leaf meal, kale leaf meal, snail meal, egg shell meal and premix. The composition and nutrient content of the ration can be seen in Table 1.
Method of Fermentation
The treatments ration was symbolized as P1, P2, P3, P4 and P5 based on the level of moisture content used in the fermentation process of ration, namely control (without adding water), 40, 50, 60 and 70%. Determination of water content in the ration according to (AOAC, 1995). The fermentation process of ration refers to Bidura et al. (2009) modified. The first stage was the ration was inserted into the container, then added to warm water (50-60°C) to each ration treatment in accordance with a certain amount. The mixture of feed and water was stirred until uniform, then aerated for 5 min. After that, the tape was inserted into the yeast ration as much as 0.3% of the weight ration and stirred until blended. The ration was then covered with plastic and stored for 7 d for aerobic fermentation. After 7 d, the ration is put into the oven at 45°C for 6 h. Rations and drinking water are given ad libitum. In this study, tape yeast used is brand "Ragi Tape NKL", which is produced locally and is commonly used in the manufacture of Indonesian food called "Tape".
Table 1. Composition and nutrient content of the experimental ration
Feed ingredients % material (w/w)
Refined corn meal 57.00
Measurement of the Weights of Visceral Organ and
Gastrointestinal Tract
A total of 10 ducks on each treatment, at the end of the experiment, was randomly selected as many as 2 birds. Ducks were then individually weighed and slaughtered, feathered and eviscerated. The weights of the gizzard, spleen, liver, pancreas, bile and heart were calculated and expressed as a percentage of BW (Rahbar et al., 2011). The empty weight of crop-esophagus, proventriculus, intestinal, duodenum, jejunum, ileum, cecum and rectum parts were recorded. Duodenum was measured from gizzard outlet to the end of the pancreatic loop, jejunum was from the pancreatic loop to Meckel’s diverticulum and ileum was from Meckel’s diverticulum to the cecal junction. The weight of gastrointestinal tracts parts to slaughtering weight was calculated and expressed as a percentage (Yalçın et al., 2013)
Analysis of Blood Profile and Chemistry
was taken and stored at -20°C (Yalçın et al., 2013) for testing of triglyceride, cholesterol, Low-Density Lipoprotein (LDL) and High-Density Lipoprotein (HDL).
Statistical Analysis
The data were Analyzed with Analysis of Variance (ANOVA) using the SPSS software with version 17. The significance of mean differences among treatments was tested by Duncan’s multiple range test at 5% of a significance level.
Results
The Weight of Gastrointestinal Tract and Visceral
Organ
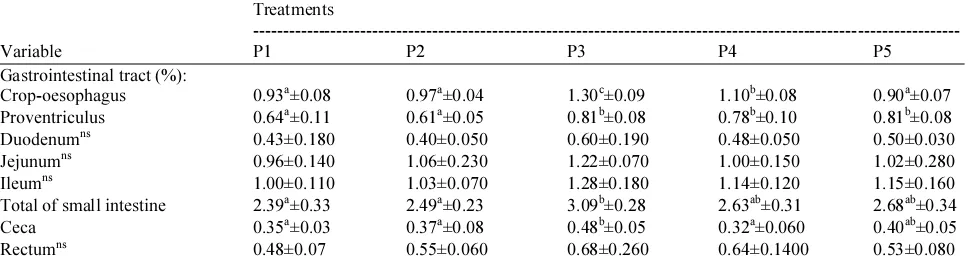
Based on Table 2, the treatment significantly (p<0.05) affected the weight of the crop-oesophagus, proventriculus, total of small intestine and ceca, but not significantly (p>0.05) affected the weight of duodenum, jejunum, ileum and rectum. The weight of crop-oesophagus, total of the small intestine and ceca of ducks fed rations fermented with a moisture content of 50% (P3) was significantly (p<0.05) the highest among the other treatment ration, namely 1.30, 3.09% and 0.48%, respectively. Meanwhile, the ducks consumed rations fermented with moisture content above 50% produced the weight of the proventriculus and small intestine that was significantly (p<0.05) higher than that under 50%. However, the weight of the proventriculus of ducks fed rations fermented with 50-70% of moisture content was not significantly different (p>0.05), which was 0.78 to 0.81%.
The different water content in fermentation process also significantly affected (p<0.05) the weight of some organs, such as the gizzard and pancreas (Table 3). Moreover, the gizzard and pancreatic weights on ducks fed ration fermented with moisture content
above 50% were significantly (p<0.05) higher than that under 50%. However, the weight of the bile, liver, spleen and heart in this study was not significantly (p>0.05) affected by treatment.
Blood Profile and Chemistry
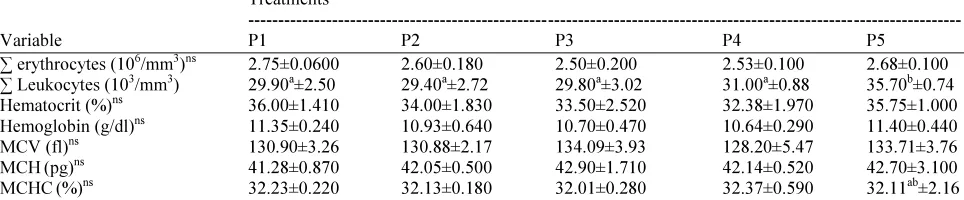
Based on Table 4, it can be observed that the treatment significantly (p<0.05) effected on the number of leukocytes, but the number of erythrocytes, hematocrit, hemoglobin, MCV, MCH and MCHC was not significantly (p>0.05) affected by the treatment. The number of leukocytes in ducks consumed rations fermented with a moisture content of 70% was significantly (p<0.05) the highest among the other treatment ration, i.e., 35,700 per mm3. Furthermore, the ducks fed rations of fermented with a moisture content of 40 to 60% had a number of leukocytes, which was not significant (p>0.05).
The administration of a ration fermented by tape yeast with a different water content had a significant difference (p<0.05) on the level of cholesterol, triglycerides and LDL in the blood of Pegagan ducks, but had not a significant difference (p>0.05) on the HDL levels in the blood (Table 5). Moreover, levels of blood cholesterol and LDL of ducks fed rations fermented with moisture content above 50% were significantly (p<0.05) lower than that under 50%. If the water level used in fermentation exceeds 50%, the levels of blood cholesterol produced did not differ significantly. The triglyceride level in the blood of ducks fed rations fermented with a water content of 70% was significantly (p<0.05) the lowest among other treatments, which is 133.75 mg dL−1. The HDL
in the duck's blood that was consumed rations fermented with 40-70% moisture content was relatively equal, i.e. 35.50 to 38 mg/dl.
Table 2. The mean of the weight of the gastrointestinal tract of Pegagan ducks aged 6 weeks Treatments
---
Variable P1 P2 P3 P4 P5
Gastrointestinal tract (%):
Crop-oesophagus 0.93a±0.08 0.97a±0.04 1.30c±0.09 1.10b±0.08 0.90a±0.07 Proventriculus 0.64a±0.11 0.61a±0.05 0.81b±0.08 0.78b±0.10 0.81b±0.08
Duodenumns 0.43±0.180 0.40±0.050 0.60±0.190 0.48±0.050 0.50±0.030
Jejunumns 0.96±0.140 1.06±0.230 1.22±0.070 1.00±0.150 1.02±0.280
Ileumns 1.00±0.110 1.03±0.070 1.28±0.180 1.14±0.120 1.15±0.160
Total of small intestine 2.39a±0.33 2.49a±0.23 3.09b±0.28 2.63ab±0.31 2.68ab±0.34 Ceca 0.35a±0.03 0.37a±0.08 0.48b±0.05 0.32a±0.060 0.40ab±0.05
Rectumns 0.48±0.07 0.55±0.060 0.68±0.260 0.64±0.1400 0.53±0.080
Table 3. The mean of the weight of visceral organs of Pegagan ducks aged 6 weeks Treatments
---
Variable P1 P2 P3 P4 P5
Visceral organs (%):
Gizzard 4.69a±0.220 5.13a±0.330 5.78b±0.35 5.77b±0.63 5.74b±0.19
Bilens 0.34±0.120 0.35±0.040 0.34±0.130 0.33±0.060 0.351±0.09
Liverns 3.66±0.750 3.26±0.670 3.78±0.350 3.63±0.810 3.61±0.550
Pancreas 0.42a
±0.04 0.43a
±0.04 0.57b
±0.07 0.50ab
±0.09 0.56b
±0.04
Spleenns 0.07±0.04 0.09±0.02 0.13±0.07 0.14±0.070 0.16±0.040
Heartns 0.75±0.06 0.70±0.09 0.82±0.12 0.80±0.110 0.84±0.050
Description: The same superscripts in the same column indicate significantly different (p<0.05). ns = non significant. P1 = Locally sourced ration fermented without adding water (control). P2, P3, P4 and P5 = locally sourced ration with a moisture content of 40, 50, 60 and 70%, respectively
Table 4. Mean values of blood profiles of Pegagan ducks aged 6 weeks Treatments
---
Variable P1 P2 P3 P4 P5
∑ erythrocytes (106/mm3)ns 2.75±0.0600 2.60±0.180 2.50±0.200 2.53±0.100 2.68±0.100 ∑ Leukocytes (103
/mm3
) 29.90a
±2.50 29.40a
±2.72 29.80a
±3.02 31.00a
±0.88 35.70b
±0.74 Hematocrit (%)ns 36.00±1.410 34.00±1.830 33.50±2.520 32.38±1.970 35.75±1.000 Hemoglobin (g/dl)ns 11.35±0.240 10.93±0.640 10.70±0.470 10.64±0.290 11.40±0.440 MCV (fl)ns 130.90±3.26 130.88±2.17 134.09±3.93 128.20±5.47 133.71±3.76 MCH(pg)ns
41.28±0.870 42.05±0.500 42.90±1.710 42.14±0.520 42.70±3.100 MCHC(%)ns 32.23±0.220 32.13±0.180 32.01±0.280 32.37±0.590 32.11ab±2.16 Description: The same superscripts in the same column indicate significantly different (p<0.05). ns = non significant. P1 = Locally sourced ration fermented without adding water (control). P2, P3, P4 and P5 = Locally sourced ration with a moisture content of 40, 50, 60 and 70%, respectively
Table 5. Mean values of blood chemistry of Pegagan ducks aged 6 weeks Treatments
---
Variable P1 P2 P3 P4 P5
Cholesterol (mg/dl) 146.25a
±2.87 145.25a
±2.87 143.00ab
±2.16 140.50b
±3.42 139.75b
±0.74 Triglyceride (mg/dl) 153.25a±1.89 152.00a±6.48 152.25a±5.12 152.75a±6.95 133.75b±4.11 LDL (mg/dl) 84.75a±2.870 83.25a±5.910 76.75b±2.990 77.25b±4.350 77.00b±2.160 HDL (mg/dl)ns 38.00±3.160 36.50±1.290 35.50±4.2000 36.75±2.9900 36.00±2.1500 Description: The same superscripts in the same column indicate significantly different (p<0.05). ns = non significant. P1 = Locally sourced ration fermented without adding water (control). P2, P3, P4 and P5 = locally sourced ration with a moisture content of 40, 50, 60 and 70%, respectively
Discussion
In principle, S. cerevisiae can affect the development of mucosa in the small intestine. S. cerevisiae can also promote the development of the small intestine by stimulating the growth of villi (de los Santos et al., 2007), so that ultimately can improve the function of the small intestine (Kidd et al., 2013) and improve the digestibility of nutrients (Akhavan-Salamat et al., 2011). Bradley et al. (1994) reported that supplementation of S. cerevisiae in ration caused a change in the morphology and the number of goblet cells in the chicken intestine. In line with this, Santin et al. (2001) also reported that administration of 0.2% S. cerevisiae in the ration could increase the villus height, located in the small intestine mucosa, which is thought to affect the weight of the small intestine. Furthermore, Rahbar et al. (2011)
reported that the use of S. cerevisiae into the basal diet containing Peganum harmala seed powder significantly influenced the villus height in the jejunum, but no significant effected on the duodenum and ileum.
pancreatic weight. During the digestion process, S. cerevisiae extract might have stimulated the secretion of the pancreatic digestive enzyme, such as α-amylase, which is effective in the digestion of dietary starch. This is as reported by Matur et al. (2010) that influence of S. cerevisiae extract on pancreatic amylase could be useful in the digestion of dietary starch. Moreover, β-glucan, known as a component of S. cerevisiae, could stimulate cholecystokinin from enteroendocrine cells, where cholecytokinin was effective to stimulate the pancreatic secretion (Chandra and Liddle, 2009; Matur et al., 2010). However, the weight of the bile, liver, spleen and heart in this study was not affected by treatments. The non-significant results were also reported by Rahbar et al. (2011) that the weight of the spleen and liver of broilers added to S. cerevisiae in the rations were not significantly different.
The high number of leukocytes can be used as an indicator to determine the immune status of poultry. The higher number of immune cells in the blood means that the better the immune system of poultry. Related to the use of S. cerevisiae in feed, Savage and Zakrzewska (1996) reported that S. cerevisiae had a significant role to stimulate the immune system in the body, which would increase the levels of IgG and IgA. This has an impact on the improvement of the immune system and can increase poultry antigenic response in the intestinal mucosa (Santin et al., 2001). In line with this, Gao et al. (2008; 2009) also stated that chickens consume product fermented by S. cerevisiae could improve the lymphocyte cell so that the body immunity was increasing. The same results also reported by Al-Homidan and Fahmy (2007) and El-Husseiny et al., 2008) that a product fermented by S. cerevisiae might modulate the immune system so that the birds consuming the product could handle the stress better.
The level of cholesterol, triglycerides and LDL in the blood of Pegagan ducks were affected by treatment. The mechanism associated with the cholesterol-lowering effect of yeast could be linked with an inhibitor of 3-hydroxyl-3-methylglutaryl- coenzyme reductase (Saleh et al., 2013). In line with this, Paryad and Mahmoudi (2008) reported that dietary supplementation of 1% S. cerevisiae
significantly lowered plasma cholesterol and triglyceride concentrations in broiler chickens, while HDL was increased. The results obtained slightly different to that reported by Yalçın et al. (2008) that administration of S. cerevisiae in rations added oilseed meal had no effect on cholesterol and blood triglycerides.
Conclusion
It was concluded that the feeding of locally sourced ration fermented by yeast tape with a moisture content of 50% provides optimal results to the weight of visceral organs and gastrointestinal tract and also on blood profile and chemistry of Pegagan ducks.
Acknowledgment
Authors thank to Ministry of Research, Technology and Higher Education who have helped the research funding through the Competence Grant in 2016.
Author’s Contributions
and contains unpublished materials and has no ethical issues involved.References
Ahmad, T., T. Khalid, T. Mushtaq, M.A. Mirza and A. Nadeem et al., 2008. Effect of potassium chloride supplementation in drinking water on Broiler performance under heat stress conditions. Poult. Sci., 87: 1276-1280. DOI: 10.3382/ps.2007-00299 Akhavan-Salamat, H., H. Ghasemi, A. Khaltabadi-Farahani
and M. Kazemi-Bonchenari, 2011. The effects of
Saccharomyces cerevisiae on performance and nutrients digestibility in broilers fed with diet containing different levels of phosphorous. African J. Biotechnol., 10: 7526-7533.
Al-Homidan, A. and M.O. Fahmy, 2007. The effect of dried yeast (Saccharomyces cerevisiae) supplementation on growth, performance, carcass chemical analysis, immunity, ileum villi heights and bacterial counts of broiler chickens. Egypt. Poult. Sci. J., 27: 613-623.
AOAC, 1995. Official methods of analysis of AOAC international. AOAC Int., Gaithersburg, MD. Asli, M.M., S.A. Hosseini, H. Lotfollahian and F.
Shariatmadari, 2007. Effect of Probiotics, yeast, vitamin E and vitamin C supplements on performance and immune response of laying hen during high environmental temperature. Int. J. Poult. Sci., 6: 895-900. DOI: 10.3923/ijps.2007.895.900 Bidura, I., I. Mahardika, B. Suyadnya, I. Partama and
D. Oka et al., 2012. The implementation of
Saccharomyces spp.n-2 isolate culture (isolation from traditional yeast culture) for improving feed quality and performance of male Bali ducking. J. Agric. Sci. Res., 2: 486-492.
Bradley, G.L., T.F. Savage and K.I. Timm, 1994. The effects of supplementing diets with Saccharomyces cerevisiae var. boulardii on male poult performance and ileal morphology. Poult. Sci., 73: 1766-1770. DOI: 10.3382/ps.0731766
Chandra, R. and R.A. Liddle, 2009. Neural and hormonal regulation of pancreatic secretion. Curr. Opin. Gastroenterol., 25: 441-446.
DOI: 10.1097/MOG.0b013e32832e9c41
de los Santos, F.S., A.M. Donoghue, M.B. Farnell, G.R. Huff and W.E. Huff et al., 2007. Gastrointestinal maturation is accelerated in turkey poults supplemented with a mannan-oligosaccharide yeast extract (Alphamune). Poult. Sci., 86: 921-930. DOI: 10.1093/ps/86.5.921
El-Husseiny, O.M., A.G. Abdallah and K.O. Abdel-Latif, 2008. The Influence of biological feed additives on broiler performance. Int. J. Poult. Sci., 7: 862-871. DOI: 10.3923/ijps.2008.862.871
Gao, J., H.J. Zhang, S.G. Wu, S.H. Yu and I. Yoon et al., 2009. Effect of Saccharomyces cerevisiae
fermentation product on immune functions of broilers challenged with Eimeria tenella. Poult. Sci., 88: 2141-2151. DOI: 10.3382/ps.2009-00151
2011. Effects of yeast (Saccharomycescerevisiae) and yeast protein concentrate on production performance of broiler chickens exposed to heat stress and challenged with Salmonella enteritidis. Anim. Feed Sci. Technol., 168: 61-71. DOI: 10.1016/j.anifeedsci.2011.03.007 Hetland, H., B. Svihus and M. Choct, 2005. Role of
insoluble fiber on gizzard activity in layers. J. Applied Poult. Res., 14: 38-46. DOI: 10.1093/japr/14.1.38 Huff, G.R., W.E. Huff, M.B. Farnell, N.C. Rath and
F. Solis de los Santos et al., 2010. Bacterial clearance, heterophil function and hematological parameters of transport-stressed turkey poults supplemented with dietary yeast extract. Poult. Sci., 89: 447-456. DOI:10.3382/ps.2009-00328
Khempaka, S., R. Thongkratok, S. Okrathok and W. Molee, 2014. An evaluation of cassava pulp feedstuff fermented with A. oryzae, on growth performance, nutrient digestibility and carcass quality of broilers. J. Poult. Sci., 51: 71-79. DOI: 10.2141/jpsa.0130022 Kidd, M.T., L. Araujo, C. Araujo, C. D. McDaniel and
D. McIntyre, 2013. A study assessing hen and progeny performance through dam diet fortification with a Saccharomycescerevisiae fermentation product. J. Applied Poult. Res., 22: 872-877.
DOI: 10.3382/japr.2013-00774
Matur, E., E. Ergul, I. Akyazi, E. Eraslan and Z.T. Cirakli, 2010. The effects of Saccharomyces cerevisiae extract on the weight of some organs, liver and pancreatic digestive enzyme activity in breeder hens fed diets contaminated with aflatoxins. Poult. Sci., 89: 2213-2220. DOI: 10.3382/ps.2010-00821
Nishii, M., M. Yasutomi and Y. Sone, 2015. Inhibitory effect of whole grain paddy rice feeding on the colonization of Campylobacter jejuni in the cecum of broiler chicks. J. Poult. Sci., 52: 312-317. DOI: 10.2141/jpsa.0140193
Paryad, A. and M. Mahmoudi, 2008. Effect of different levels of supplemental yeast (Saccharomyces cerevisiae) on performance, blood constituents and carcass characteristics of broiler chicks. African J. Agric. Res., 3: 835-842.
Rahbar, M.G., P. Farhoomand and A. Kamyab, 2011. The effect of different concentrations of Peganum harmala seeds with or without a yeast cell wall product on the live performance, intestinal histomorphology and weights of visceral organs of broiler chickens. J. Applied Poult. Res., 20: 454-462. DOI: 10.3382/japr.2010-00261
Rohmawati, D., I. Djunaidi and E. Widodo, 2015. Nutritional values of soybean husk with tape yeast at different levels and incubation times. J. Ternak Trop., 16: 30-33.
Saleh, A.A., K. Hayashi and A. Ohtsuka, 2013. Synergistic effect of feeding Aspergillus awamori
and Saccharomyces cerevisiae on growth performance in broiler chickens; promotion of protein metabolism and modification of fatty acid profile in the muscle. J. Poult. Sci., 50: 242-250. Sandi, S., F. Yosi and M. Miksusanti, 2016. Performance
and protein efficiency ratio of starter phase pegagan ducks fed fermented rations made from locally sourced materials. Pakistan J. Nutr., 15: 268-273. DOI: 10.3923/pjn.2016.268.273
Santin, E., A. Maiorka, M. Macari, M. Grecco and J.C. Sanchez et al., 2001. Performance and intestinal mucosa development of broiler chickens fed diets containing Saccharomyces cerevisiae
cell wall. J. Applied Poult. Res., 10: 236-244. DOI: 10.1093/japr/10.3.236
Savage, T. and E. Zakrzewska, 1996. The performance of male turkeys fed a starter diet containing a mannanoligosaccharide (Bio-Mos) from day old to eight weeks of age. Proceedings of the Alltech’s 12th Annual Symposium Biotechnology in the Feed Industry, (BFI’ 96), pp: 47-54.
Yalçın, S., H. Eser, S. Yalçın, S. Cengiz and O. Eltan, 2013. Effects of dietary yeast autolysate (Saccharomyces cerevisiae) on performance, carcass and gut characteristics, blood profile and antibody production to sheep red blood cells in broilers. J. Applied Poult. Res., 22: 55-61. DOI: 10.3382/japr.2012-00577 Yalçın, S., B. Ozsoy, H. Erol and S. Yalçın, 2008. Yeast
culture supplementation to laying hen diets containing soybean meal or sunflower seed meal and its effect on performance, egg quality traits and blood chemistry. J. Applied Poult. Res., 17: 229-236. DOI: 10.3382/japr.2007-00064
Yalçın, S., S. Yalçın, K. Çakın, Ö. Eltan and L. Dağaşan, 2010. Effects of dietary yeast autolysate (Saccharomyces cerevisiae) on performance, egg traits, egg cholesterol content, egg yolk fatty acid composition and humoral immune response of laying hens. J. Sci. Food Agric., 90: 1695-1701. DOI: 10.1002/jsfa.4004