www.elsevier.com / locate / bres
Short communication
Coupling of arterial pulse and 10-Hz rhythm in sympathetic nerve
discharge
*
Peter D. Larsen , Sheng Zhong, Craig D. Lewis, Gerard L. Gebber, Susan M. Barman
Department of Pharmacology and Toxicology, Michigan State University, East Lansing, MI 48824-1317, USA
Accepted 11 July 2000
Abstract
We used time series analysis to characterize the relationships among the arterial pulse (AP) and the cardiac-related and 10-Hz rhythms in sympathetic nerve discharge (SND) of urethane-anesthetized cats. We found that 10-Hz activity was more tightly coupled to the AP than to the cardiac-related rhythm. These data support the view that the dynamic coupling of AP and the 10-Hz rhythm in SND involves a direct influence of baroreceptor activity on the 10-Hz oscillator. 2000 Elsevier Science B.V. All rights reserved.
Theme: Endocrine and autonomic regulation
Topic: Cardiovascular regulation
Keywords: Baroreceptor; Forced oscillation; Phase-locking; Time series analysis
Biological oscillators are commonly entrained by per- via coupling of the 10-Hz oscillator to the oscillator iodic inputs from either the external or internal environ- responsible for the cardiac-related rhythm in SND [10]. ment [7,11–13]. Work from this laboratory supports the Because there is variation in the phase relationship be-view that the cardiac-related rhythm observed in sympa- tween AP and the cardiac-related rhythm, we examined thetic nerve discharge (SND) is the consequence of 1:1 these two alternatives by comparing the strength of cou-entrainment of centrally generated low frequency (2- to pling of the cardiac-related and 10-Hz rhythms in SND 6-Hz) oscillations by pulse-synchronous baroreceptor nerve with that of the AP and 10-Hz rhythm.
activity [8,14]. SND in the baroreceptor-intact, urethane- The protocols used in this study were approved by the anesthetized cat also contains a 10-Hz rhythm intermixed All-University Committee on Animal Use and Care of with the cardiac-related rhythm [4]. The 10-Hz rhythm is Michigan State University. Data were obtained from 12 generated by brainstem neurons distinct from those respon- cats (weight range, 2.75–4.35 kg), anesthetized with sible for the 2- to 6-Hz oscillations [2,3]. urethane (1.2–1.4 g / kg i.v.), following initial induction Several laboratories [5,6,10,15] have reported instances with isoflurane (2.5–3.5%) in 100% oxygen. The cats were of phase-locking of the arterial pulse (AP) and 10-Hz paralyzed (gallamine triethiodide; initial dose, 4 mg / kg rhythm in simple harmonic (e.g., 1:3, 1:4) or complex i.v.) and artificially ventilated with room air. A bilateral (e.g., 2:5, 2:7) ratios. The coupling of AP and 10-Hz pneumothoracotomy was performed. End-tidal CO2 was rhythm may reflect a direct action of elements in the maintained between 3.5 and 4.0% by adjusting the parame-afferent limb of the baroreceptor reflex arc on the brain- ters of ventilation. Rectal temperature was maintained near stem neurons responsible for the 10-Hz rhythm in SND. 388C by using a heat lamp.
Alternatively the 10-Hz rhythm may have been indirectly Blood pressure was measured from a catheter inserted entrained to pulse-synchronous baroreceptor nerve activity into the femoral artery, and the AP was considered to reflect the timing of pulse-synchronous baroreceptor nerve activity. As previously described [5], bipolar platinum
*Corresponding author. Tel.: 11-517-432-3154; fax: 1
1-517-353-electrodes were used to record monophasically from the
8915.
E-mail address: [email protected] (P.D. Larsen). central end of the cut inferior cardiac postganglionic
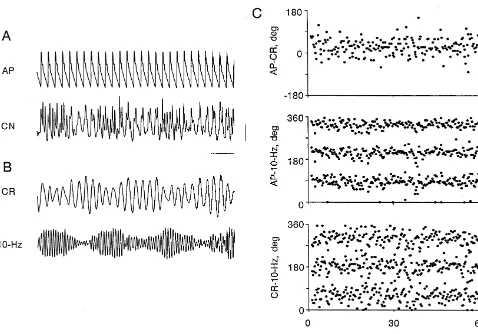
sympathetic nerve (CN) near its exit from the left stellate Following filtering we made cycle-by-cycle measurements ganglion. Nerve recordings were made with a bandpass of the interval (ms) between the peak of the AP (peak filter set at 1–1000 Hz (Grass Instruments 7P3 pream- systole) and the surrounding peaks of cardiac-related and plifier), so that envelopes of multiunit spikes appeared as 10-Hz slow waves in SND, and the intervals between the slow waves [5,6]. We analyzed epochs of data in which the peaks of the corresponding cardiac-related and 10-Hz slow CN activity contained a mixture of the 10-Hz and cardiac- waves in SND. These intervals were then converted to related rhythms. phase angles (f) in degrees [9], and were plotted as time We processed the original recording of SND (Fig. 1A, series (Fig. 1C). Positive and negative values of phase bottom trace) twice to produce two filtered records, one angle refer to lags and leads, respectively, of the second containing cardiac-related activity only (Fig. 1B, top signal relative to the first.
trace), the other 10-Hz activity only (Fig. 1B, bottom The example shown in Fig. 1 shows 1:1 phase-locking trace). A digital filter (symmetric, non-recursive type with of low frequency oscillations in SND to the arterial pulse a Lanczos smoothing function; RC Electronics, Santa (Fig. 1C, top), with 90% of the phase angles restricted Barbara CA) with a bandpass width of 4 Hz centered at the between 2308 and 758. In this experiment 1:3 phase-frequency of either the cardiac-related or the 10-Hz rhythm locking of the AP and the 10-Hz rhythm (Fig. 1C, middle) was used for this purpose [9]. The frequency of both the and the cardiac-related and the 10 Hz rhythms in SND cardiac-related and 10-Hz rhythms was determined from (Fig. 1C, bottom) is reflected by three horizontal bands the autospectra of SND [9]. There was minimal phase within the time series plots. In the plot showing the distortion and the filtered records were smooth and sinusoi- AP-10-Hz SND relationship, 90% of the values of phase dal in shape, thus allowing accurate detection of peaks. angles for the first band were restricted to a range of 608,
while in the plot of cardiac-related SND-10-Hz SND, 90% to 6-Hz slow waves in SND were spread over 3608 (Fig. of the values of phase angles for the first band were 2A, top panel). However, pulse-synchronous baroreceptor restricted to a range of 908. In 16 of 20 cases we observed nerve activity was strong enough to entrain the 10-Hz phase-locking of the AP and 10-Hz rhythms. In all of these rhythm in a 1:3 ratio (Fig. 2A, middle panel). As expected, cases the 10-Hz rhythm (which ranged in frequency from under these conditions no phase-locking of low frequency 7.6 to 12.4 Hz) was more tightly coupled to AP than to the oscillations and the 10-Hz activity was observed (Fig. 2A, cardiac-related rhythm (frequency range, 2.4–4.2 Hz) in bottom panel). Fig. 2B shows an example in which only SND (mean range containing 90% of AP-10-Hz SND low frequency oscillations in SND were coupled to AP. phase angles in the first band was 806258, versus 1106408 Mean blood pressure was 115 mmHg in this case. The for cardiac related SND-10-Hz SND, P50.002, paired band of phase angles between 270 and 3308 illustrates the
t-test). These results imply that the low frequency and 1:1 relationship (Fig. 2B, top panel). In contrast the phase 10-Hz components of SND are independently entrained to angles relating the 10-Hz rhythm to AP (Fig. 2B, middle pulse-synchronous baroreceptor activity. panel) or cardiac-related SND (Fig. 2B, bottom panel)
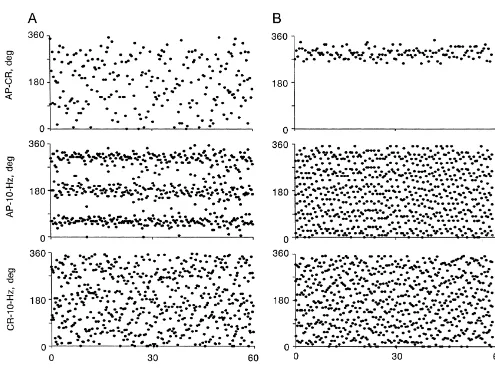
The results in Fig. 2 further support this view. The time were spread over 3608.
series in Fig. 2A show one of three cases in which only the Fig. 3 shows episodes of phase-walk of the 10-Hz 10-Hz rhythm in SND was coupled to the AP. In this case rhythm in SND relative to the AP without a change in the mean blood pressure was 82 mmHg. At this level of blood strength of 1:1 phase-locking of the low frequency oscilla-pressure, pulse-synchronous baroreceptor nerve activity tions to the AP. While the relationship between AP and was not strong enough to entrain the 2- to 6-Hz oscillations cardiac-related activity remained relatively constant over in SND, and therefore the phase angles between AP and 2- the 60-s period shown, the relationship between AP and
cases of 1:3 AP-10-Hz phase-locking in the absence of entrainment of 2- to 6-Hz activity by AP (Fig. 2A) demonstrates that, at least in some circumstances, the influence of pulse-synchronous baroreceptor nerve activity on the 10-Hz oscillator was stronger than on the generator of 2- to 6-Hz activity.
Another feature of a nonlinear interaction between a forcing input and an oscillator is phase-walk, or relative coordination [12,16]. Phase-walk occurs when the strength of the input is not sufficient to result in phase-locking, but there is still an attraction of the oscillator to the input [7] resulting in an ordered cycle-by-cycle change in phase angle. In our experiments we observed episodes of walk of the 10-Hz rhythm relative to AP when 1:1 phase-locking of the low frequency oscillations to the AP was maintained.
The significance of dynamic coupling of the 10-Hz rhythm in SND to pulse-synchronous baroreceptor nerve activity is not clear. It has been suggested that such interactions are a basic feature of coordination of neural
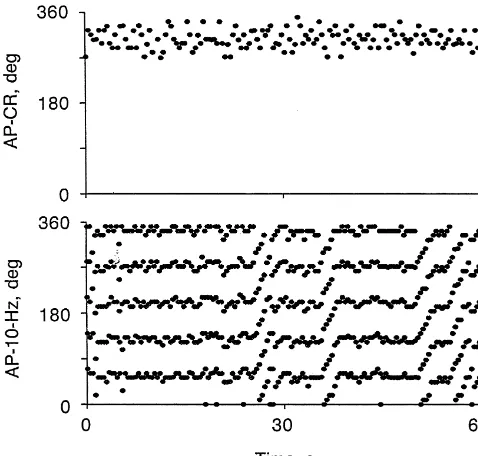
Fig. 3. Time series plots (top to bottom) of AP-CR and AP-10-Hz phase function [12]. In this regard, our laboratory has demon-angles. Phase-locking was present throughout the time series in the
strated that entrainment of the 10-Hz rhythm by medullary
AP-CR plot, but in the AP-10-Hz plot there were episodes of
phase-raphe stimulation leads to increases in the amplitude of the
walks. The frequency of the CR and 10-Hz rhythms were 2.5 and 12.4
10-Hz slow waves (resonance) [10]. It is therefore possible
Hz, respectively.
that entrainment of 10-Hz rhythm by pulse-synchronous baroreceptor activity may influence the level of sympa-10-Hz activity showed a number of transitions between 1:5 thetic outflow to peripheral targets.
phase-locking and phase-walk. The phase-walks consisted of cycle-by-cycle increases in the phase angle between AP and 10-Hz slow waves for five to six cycles until
phase-Acknowledgements
locking was reestablished. Note that the phase-walks did not alter the preferred phase angle during phase-locking. In
This study was supported by National Heart, Lung, and two cases where 1:3 phase-locking was the predominant
Blood Institute Grant HL-13187. mode of coordination between AP and 10-Hz rhythm we
also observed periods of phase-walk in the absence of any changes in the relationship between AP and low frequency
oscillations in SND. References
While we cannot exclude the possibility that some form
of coupling exists between the generators of the 2- to 6-Hz [1] V.I. Arnold, in: Geometrical Methods in the Theory of Ordinary oscillations and the 10-Hz rhythm, we have demonstrated Differential Equations, Springer, New York, 1988, p. 365.
[2] S.M. Barman, G.L. Gebber, Lateral tegmental field neurons of cat
for the first time that the dynamic coupling of AP and
medulla: a source of basal activity of ventrolateral medullospinal
10-Hz rhythm in SND is primarily due to a direct influence
sympathoexcitatory neurons, J. Neurophysiol. 57 (1987) 1410–
of baroreceptor activity on the 10-Hz oscillator. This is
1424.
demonstrated by the observations that the 10-Hz activity [3] S.M. Barman, H.S. Orer, G.L. Gebber, Caudal ventrolateral medul-was always more tightly coupled to AP than it medul-was to lary raphe neurons are elements of the network responsible for the
cardiac-related SND, and that AP-10-Hz phase-locking 10-Hz rhythm in sympathetic nerve discharge, J. Neurophysiol. 72 (1994) 106–120.
occurred, on occasion, in the absence of entrainment of
2-[4] S.M. Barman, G.L. Gebber, Subgroups of rostral ventrolateral
to 6-Hz activity to the AP.
medullary and caudal medullary raphe neurons based on patterns of
Theoretically 1:1 entrainment is the most stable form of relationship to sympathetic nerve discharge and axonal projections, phase-locking between a periodic input and an oscillator J. Neurophysiol. 77 (1997) 65–75.
[1]. This means that the strength of the forcing input [5] S.M. Barman, G.L. Gebber, S. Zhong, The 10-Hz rhythm in sympathetic nerve discharge, Am. J. Physiol. 262 (1992) R1006–
required to achieve 1:1 coupling is lower than that required
R1014.
for 1:3 coupling, and therefore we expected to see cases
[6] M.I. Cohen, P.M. Gootman, Periodicities in efferent discharges of
(Fig. 2B) in which there was cardiac-related activity (i.e., splanchnic nerve of the cat, Am. J. Physiol. 218 (1970) 1092–1101. 1:1 entrainment of 2- to 6-Hz oscillations) in the absence [7] G.B. Ermentrout, J. Rinzel, Beyond a pacemaker’s entrainment
[8] G.L. Gebber, Basis for phase relations between baroreceptor and [13] G.A. Petrillo, L. Glass, A theory for phase-locking of respiration in sympathetic nerve discharge, Am. J. Physiol. 230 (1976) 263–270. cats to a mechanical ventilator, Am. J. Physiol. 246 (1984) R311– [9] G.L. Gebber, S. Zhong, C. Lewis, S.M. Barman, Differential R320.
patterns of spinal sympathetic outflow involving a 10-Hz rhythm, J. [14] D.G. Taylor, G.L. Gebber, Baroreceptor mechanisms controlling Neurophysiol. 82 (1999) 841–854. sympathetic nervous rhythms of central origin, Am. J. Physiol. 228 [10] G.L. Gebber, S. Zhong, S.Y. Zhou, S.M. Barman, Nonlinear (1975) 1002–1013.
dynamics of the frequency locking of baroreceptor and sympathetic [15] I. Ninomiya, T. Akiyama, N. Nishiura, Mechanisms of cardiac-rhythms, Am. J. Physiol. 273 (1997) R1932–R1945. related synchronized cardiac sympathetic nerve activity in awake [11] M.R. Guevara, A. Shrier, L. Glass, Phase-locked rhythms in cats, Am. J. Physiol. 259 (1990) R499–R506.
periodically stimulated heart cell aggregates, Am. J. Physiol. 254 [16] E. von Holst, Relative coordination as a phenomenon and as a (1988) H1–H10. method of analysis of central nervous functions, in: R. Martin (Ed.), [12] J.A.S. Kelso, in: Dynamic Patterns. The Self-Organization of Brain The Collected Papers of Erich von Holst, Univ. Miami Press, Coral