Brain Research 888 (2001) 263–266
www.elsevier.com / locate / bres
Research report
Dehydroepiandrosterone (DHEA) reduces neuronal injury in a rat
model of global cerebral ischemia
*
Hui Li, GaryM. Klein, Ping Sun, Alastair M. Buchan
Alberta Stroke Program, Department of Clinical Neurosciences, Foothills Hospital, University of Calgary, Calgary, AB T2N 2T9, Canada
Accepted 3 October 2000
Abstract
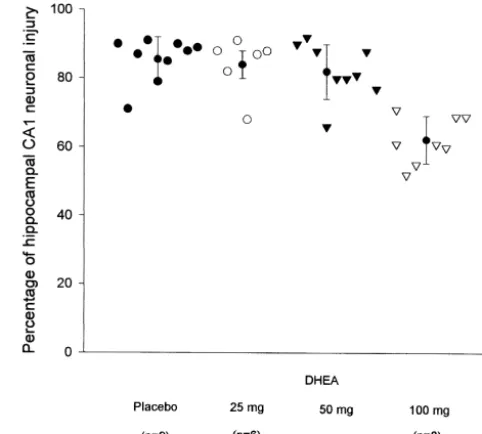
Introduction: Many studies report an inverse correlation between levels of DHEA and neurological diseases. Exogenous DHEA protects hippocampal neurons against excitatory amino acid induced neurotoxicity. The purpose of this experiment is to evaluate the effect of DHEA in an animal model of transient but severe forebrain ischemia. Methods: At thirteen days prior to induction of ischemia, male Wistar rats were implanted with various doses of DHEA–placebo, 25 mg, 50 mg or 100 mg. Forebrain ischemia was induced for 10 min using a modified four-vessel occlusion technique, with hippocampal neuronal injury assessed at 7 days post-ischemically and expressed as a percentage of total cells. Results: Both normal and necrotic hippocampal CA cells were counted. Percentages of hippocampal injury1
observed were 88613% in animals treated with placebo, 8468% in the 25 mg DHEA group, and 6067% in the 50 mg DHEA group. Animals treated with 100 mg DHEA displayed a significant (P,0.05) reduction of hippocampal CA cell injury at 601 67% Conclusion: Treatment with a high dose, but not a low or moderate dose, of DHEA implantation reduces hippocampal CA neuronal injury following1
severe but transient forebrain ischemia. 2001 Elsevier Science B.V. All rights reserved.
Theme: Disorders of the nervous system
Topic: Ischemia
Keywords: Cerebral Ischemia; Hippocampus; Rat; DHEA
1. Introduction able to delayed injury following ischemia [15,21]. Release
of glutamate and subsequent calcium entry contribute to
Dehydroepiandrosterone (DHEA) and its sulfate ischemic CA neuronal cell injury [7]. Blocking the effects1
(DHEAS) are the major secretory products of the adrenal of glutamate using NMDA receptor [13] or AMPA receptor
gland. The serum concentration of DHEAS is between 300 [13] antagonists can reduce neuronal injury.
and 500 times higher than that of DHEA, although DHEA Various studies show that DHEA(S) possess multiple
is probably more active at the tissue level. Brain de novo biological activities, such as neurotrophic effects [23,12],
can generate DHEA (termed neurosteroid), and its con- modulation of NMDA receptor and calcium channel [6],
centration is higher than that in the periphery [17,6]. up-regulation of neuronal excitability [18], and blocking
Decreases of DHEA have been linked to the development effects of glucocorticoid [3]. Both in vivo and in vitro
of several neurological diseases [4,6]. studies have demonstrated that treatment with DHEA can
Transient forebrain ischemia causes irreversible neuro- reduce NMDA-induced neurotoxic effects [14] and
oxida-nal degeneration to highly sensitive regions of the brain. In tive stress-induced damage [5] in the hippocampal CA1
particular, the hippocampal CA cell is exquisitely vulner-1 region.
Currently there are no animal experimental data avail-able to support the roles of DHEA and its sulfate (DHEAS) in cerebral ischemia. The purpose of this experiment was to determine whether DHEA offered
*Corresponding author. Tel.: 11-403-670-1581; fax: 1
1-403-670-neuroprotection in a rat model of transient but severe
1602.
E-mail address: [email protected] (A.M. Buchan). forebrain ischemia.
264 H. Li et al. / Brain Research 888 (2001) 263 –266
2. Material and methods
Male Wistar rats, weighing 150–175 g (Charles River, Montreal, Canada), were used in this study. Pellets of DHEA or placebo, purchased from Innovative Research of America (Sarasota, FL, USA), were implanted dorsally in the subcutaneous tissue of the neck 12 days before surgical preparation for ischemia, and remained in the body until the animals were sacrificed. Animals were placed into one
of the following four groups: placebo (n59), 25 mg
DHEA (n56), 50 mg DHEA (n59), or 100 mg DHEA
(n58).
The surgical method for preparation of 4-vessel occlu-sion (4-VO) has been previously described in detail [22]. In brief, the animals were anesthetized with a mixture of 2% halothane, 70% N , and 28% O . Reversible ligatures2 2
were placed around both common carotid arteries and both vertebral arteries were cauterized. The wounds were closed and animals were returned to their cages overnight.
Fig. 1. Percentage of hippocampal CA neuronal injury (mean6S.D.)
The following day, the animals were subjected to 10 min 1
measured after 7 days of survival following ten min of forebrain ischemia
of forebrain ischemia by tightening the ligatures around the
in rats receiving varying dosages of DHEA or placebo.
carotid arteries and the para-vertebral muscles in which the collateral arteries are located. Body temperature was
maintained at 37.58C. sented in Fig. 1. Hippocampal CA cell injuries were not1
Seven days following ischemia the animals were reanes- significantly different among placebo (88613%), DHEA
thetized and perfusion-fixed with a transcardiac infusion of 25 mg (8468%), and DHEA 50 mg (8266%) groups. In
heparinized saline, followed by 4% buffered formalin. the DHEA higher dosage group (100 mg), a mild but
Serial 7 mm sections were cut and stained with hemato- significant reduction of neuronal injury in the hippocampal
xylin and eosin. The number of necrotic and normal cells CA region was measured. In this group, neuronal injury1
in the CA region of the hippocampus were counted and1 was reduced to 6067% from 88613% seen in the placebo
expressed as mean6standard deviation of percent neuronal group (P,0.05).
injury. The percentages were compared with the Kruskal– Wallis test and the Mann–Whitney U test with Bonferroni
corrections. This sample size, through a priori calculation 4. Discussion
using an expected mean of 80620%, was adequate to
detect a 30% difference among groups (a50.05, b50.2). This is the first animal experiment to test the efficacy of
All data are expressed in percentage mean6S.D. DHEA as a neuroprotectant in cerebral ischemia. The
results from this experiment demonstrate that a high dose, but not a low or moderate dose, of DHEA reduces
3. Results neuronal injury following transient and severe forebrain
ischemia. Although the neuroprotection is mild and weak,
3.1. Mortality DHEA might be used as supplementary neuroprotective
agent for future stroke treatment.
Two animals from group 2 (DHEA, 25 mg) and one The molecular basis of selective CA1 neuronal injury
animal from group 4 (DHEA, 100 mg) died during the following ischemia remains speculative. Excessive
gluta-ischemic period and were thus excluded from the study. mate release and resulting calcium entry, as well as
Animals exhibited normal weight gain and did not show increased post-ischemic sensitivity to excitatory
neuro-any abnormal behaviour. All animals maintained a body transmitters, may be key factors in CA1 neuronal loss
temperature of 37.58C following the DHEA implantation [7,25,9]. Antagonists of the
amino-T-hydroxy-5-methyl-4-and ischemia. All animals met ischemic criteria amino-T-hydroxy-5-methyl-4-and DHEA isoxazole proprionic acid (AMPA) receptors have been
did not influence the behaviour of the animals during or found to reduce CA loss [16,24]. SNX-111, which blocks1
21
following 4-VO. N-type Ca channels and prevents glutamate release, has
been shown to reduce CA1 injury [25,9]. Moreover, a
3.2. Neuropathology previous study suggests that reducing the inflammation
incurred by ischemia can also reduce CA cell injury [19].1
H. Li et al. / Brain Research 888 (2001) 263 –266 265
[2] N.J. Alkayed, I. Harukuni, A.S. Kimes, E.D. London, R.J.
primary hippocampal culture against NMDA induced
Traystman, P.D. Hurn, Gender-linked brain injury in experimental
toxicity [14] and enhance survival rates of cultured
neu-stroke, Stroke 29 (1998) 159–165.
rons [8]. In vivo, subcutaneous pellets of DHEA protected [3] B. Areneo, R. Daynes, Dehydroepiandrosterone functions as more hippocampal CA neurons against unilateral infusions of1 than an antiglucocorticoid in preserving immunocompetence after
thermal injury, Endocrinology 121 (1995) 135–143.
NMDA [14]. We have used the same pellet method of
[4] S. Bastianetto, C. Ramassamy, J. Poirier, R. Quirion,
Dehydro-DHEA administration and therefore infer that our plasma
eiandrosterone (DHEA) protects hippocampal calls from oxidative
levels were similar to Kimonides et al., who measured
stress-induced damage, Mol. Brain Res. 66 (1999) 35–41.
DHEA to be protective at 16.561.8 ng / ml. The results of [5] S. Bastianetto, R. Quirion, Any facts behind the DHEA hype?,
Kimonides et al. were felt to suggest that a decreased Trends Pharmacol. Sci. 18 (1997) 447–449.
[6] E.E. Baulieu, P. Robel, Dehydroepiandrosterone and
dehydroepian-DHEA level may contribute in part to the increased
drosterone sulfate as neuroactive neurosteroids, J. Endocrinol. 150
vulnerability of the aging or stressed human brain to such
(1996) S221–S239.
damage [2].
[7] H. Benveniste, J. Drejer, A. Schouboe, N.H. Diemer, Elevation of
In the current experiment, the mild reduction of is- the extracellular concentrations of glutamate and aspartate in rat
chemic neuronal injury demonstrated with treatment by hippocampus during transient cerebral ischemia monitored by
intracerebral microdialysis, J. Neurochem. 43 (1984) 374–396.
DHEA might suggest that NMDA is not a main contributor
[8] L. Bologa, J. Sharma, E. Roberts, Dehydroepiandrosterone and its
in the degenerative process involving hippocampal CA1
sulfated derivative reduce neuronal death and enhance astrocytic
cells following cerebral ischemia. Neuroprotective effects differentiation in brain cell cultures, J. Neurosci. Res. 17 (1987)
by NMDA glutamate antagonists have not been universally 225–234.
supported in animal studies. Experimental data has demon- [9] A.M. Buchan, S.Z. Gertler, H. Li, D. Xue, Z.G. Huang, K.E.
21
Chaundy, K. Barnes, H.J. Lesiuk, A selective N-type CA -channel
strated that NMDA antagonists (MK-801) were unable to
blocker prevents CA1 injury 24 h following severe forebrain
prevent selective neuronal death following brief but severe
ischemia and reduces infarction following focal ischemia, J. Cereb.
forebrain ischemia [10,20], although they might reduce Blood Flow Metab. 14 (1994) 903–910.
cortical infarction in focal ischemia [11]. This is possibly [10] A.M. Buchan, H. Li, W.A. Pulsinelli, The N-methyl-D-aspartate
as a result of altered regional cerebral blood flow, which is antagonist, MK-801, fails to protect against neuronal damage caused by transient severe forebrain ischemia in adult rats, J. Neurosci. 11
possible in focal but not global ischemia. In this
experi-(1991) 1049–1056.
ment, which used transient but severe forebrain ischemia,
[11] A.M. Buchan, A. Slivka, D. Xue, The effect of the NMDA receptor
blood flow is severely attenuated and not easily influenced. antagonist MK-801 on cerebral blood flow and infarct volume in
The behaviour of the DHEA-treated animals during is- experimental focal stroke, Brain Res. 574 (1992) 171–177.
chemia did not suggest any improvement in flow that [12] N.A. Compagnone, S.H. Melon, Dehydroepiandrosterone: a
po-tential signaling molecule for neocortical organization during
de-might account for possible neuroprotective effects.
velopment, Proc. Natl. Acad. Sci. USA 95 (1998) 4678–4683.
In a clinical setting, this may be a more complex issue.
[13] R. Gill, A. Foster, G. Woodruff, Systemic administration of MK-801
In men, DHEA acts like an estrogen and may protect
protects against ischemia-induced hippocampal neurodegeneration in
against cardiovascular disease [1]. In post-menopausal the gerbil, J. Neurosci. 7 (1987) 3345–3349.
women, metabolism of DHEA to testosterone may actually [14] V.G. Kimonides, N.H. Khatibi, C.N. Svendsen, M.V. Sofroniew, J.
increase the risk of cardiovascular disease. In pre- Herbert, Dehydrocpiandrosterone (DHEA) and DHEA-sulfate
(DHEAS) protect hippocampal neurons against excitatory amino
menopausal women, higher estrogen concentrations may
acid-induced neurotoxicity, Proc. Natl. Acad. Sci. USA 95 (1998)
counter-balance the androgenic effects of DHEA [17].
1852–1857.
In experimental models of cerebral infarction, there [15] T. Kirino, Delayed neuronal death in the gerbil hippocampus appear to be major sex-linked differences in brain damage following ischemia, Brain Res. 237 (1982) 57–69.
[7]. Estrogens have been shown to reduce tissue damage [16] H. Li, A.M. Buchan, Treatment with an AMPA antagonist 12 hours following severe normothermic forebrain ischemia prevents CA
after stroke in both male and female animals [13,26]. This 1
neuronal injury, J. Cereb. Blood Flow Metab. 13 (1993) 933–939.
may explain why the estrogen-like effects of DHEA are
[17] M.D. Majewska, Neurosteroids: endogenous bimodal modulators of
protective in men, while the opposite may be true in the GABAa receptor: mechanism of action and physiological
post-menopausal women. significance, Prog. Neurobiol. 38 (1992) 379–395.
DHEA is now widely available from a variety of [18] M.D. Majewska, S. Demoirgoren, C.E. Spivak, E.D. London, The
neurosteroid dehydroepiandrosterone sulfate is an allosteric
antago-sources. It has been extensively promoted as an
‘alter-nist of the GABA receptor, Brain Res. 526 (1990) 143–146.A
native’ therapy that prevents the effects of aging, boosts
[19] M. Nakayama, K. Uchimura, R.L. Zhu, T. Nagayama, M.E. Rose,
immunity to cardiovascular disease, offers natural weight R.A. Stetler, P.C. Isakson, J. Chen, S.H. Graham, Cyclooxygenase-2
control, prevents cancer, enhances brain function, and inhibition prevents delayed death of CA1 hippocampal neurons
extends life. following global ischemia, Proc. Natl. Acad. Sci. USA 95 (1998)
10954–10959.
[20] B. Nellgard, I. Gustafson, T. Wieloch, Lack of protection by the N-methyl-D-aspartate receptor mitigates neuronal damage in the rat
References brain following transient severe cerebral ischemia in the rat,
Anesthesiology 75 (1991) 279–287.
266 H. Li et al. / Brain Research 888 (2001) 263 –266
[22] W.A. Pulsinelli, A.M. Buchan, The four-vessel occlusion rat model: [25] J.K. Toung, R.J. Traystman, P.D. Hurn, Estrogen-mediated neuro-method for complete occlusion of vertebral arteries and control of protection after experimental stroke in male rats, Stroke 29 (1998) colateral circulation, Stroke 19 (1988) 913–914. 1666–1670.
[23] E. Roberts, L. Bologa, J.F. Flood, G.E. Smith, Effects of dehydro- [26] K. Valentino, R. Newcomb, T. Gadbois, T. Singh, S. Bowersox, S. epiandrosterone and its sulfate on brain tissue in culture and on Bitner, J. Ramachandran, A selective N-type calcium channel memory in mice, Brain Res. 406 (1987) 357–362. antagonist protects against neuronal loss after global ischemia, Proc. [24] M.J. Sheardown, E. Nielsen, A.J. Hansen, P. Jacobsen, T. Honore, Natl. Acad. Sci. USA 90 (1993) 7894–7897.