J.A. Carreira
a,*, R. Garcı´a-Ruiz
a, J. Lie´tor
a, A.F. Harrison
baArea de Ecologı´a, Universidad de Jae´n, Campus Las Lagunillas B-3, 23071 Jae´n, Spain bMerlewood Research Station, Institute of Terrestrial Ecology, Grange-over-Sands LA11 6JU, UK
Accepted 30 May 2000
Abstract
The indirect effects of acid-misting applied to tree canopies, on soil P availability, phosphatase activity and P mineralisation rates in a Sitka spruce (Picea sitchensis(Bong.) Carr) stand have been investigated. The treated trees, grouped according to five height classes, had for four years previously received acid mist consisting of a mixture of H2SO4and NH4NO3(pH 2.5) at concentrations of 3.2 mM H1and 1.6 mM each
of NH14;NO 2
3 and SO224 :These simulate cloudwater composition in uplands affected by acidic deposition. Another group of mixed
height-class trees, received a double dose of acid mist. Control — no acid-misted-trees received only the usual precipitation inputs. The acid mist treatments had previously been found to induce a significant reduction in stem diameter growth, but no canopy visible injury symptoms were observed. Root bioassays had demonstrated the induction of P nutritional stress in acid-misted versus no acid-misted trees. Soil chemical analysis showed that acidifying inputs induced changes in the inorganic P subcycle by: (i) increasing acidity and P sorption capacity of the soils; and (ii) decreasing the concentration of labile inorganic P. We report here on changes in the organic P subcycle (phosphatase activity and gross mineralisation rates). We used a method which applies independent treatments to soil samples (control, HgCl2-addition, HgCl2
-addition1autoclaving) to separate mineral solubilisation, organic mineralisation and immobilisation of solution P. Soil phosphatase activity was significantly lower under acid-misted trees (263.9mg pNP h21g21) than under control trees (382.0mg pNP h21g21). Soils beneath double-dose acid-misted trees showed a 42% reduction in the labile inorganic P size, and a 33% increase in the labile organic P pool-size, compared to soils under control trees. A pattern of decreasing net P solubilisation and gross P mineralisation rates with increasing acid-mist dose-level was also found. Thus, the combined effects of acid-acid-misting on the inorganic and organic P subcycles, in addition to the suggested effect of N saturation enhancing demand for P by the trees, may have induced P deficiency and reduced tree growth. The effects described are considered to be important factors in the forest decline syndrome.q2000 Elsevier Science Ltd. All rights reserved.
Keywords: Picea sitchensis; Acidic deposition; Soil acidification; Phosphorus availability; Gross P mineralisation; Net P solubilisation; Immobilisation; Phosphatase activity
1. Introduction
Much research has concentrated on the acidifying effects of atmospheric pollutant inputs on soils and their roles in forest decline (Schulze, 1989), as this has become a signifi-cant economic and environmental problem in Europe. Forest damage both in Europe and in North America is frequently associated with acute nutritional unbalances (Hu¨ttl, 1988; Tomlinson, 1990), often caused by base cations (Mg and K) leaching from the soil (Ulrich et al., 1980; Van Breemen et al., 1983) and induced cation defi-ciencies (Van Dick and Roelofs, 1988; Kazda, 1990). The induction of phosphorus deficiency has also been proposed
as a possible effect of acidic deposition (Foy et al., 1978), but there is a lack of studies specifically addressing the question (Binkley et al., 1989). A limited number of reports have indicated that pollutant inputs are inducing phosphorus deficiency in forest trees, at least in some sensitive areas both in North America (Pare´ and Bernier, 1989a) and in Europe (Mohren et al., 1986; Van Dick and Roelofs, 1988; Houdijk and Roelofs, 1993). Phosphorus deficiency is known to be a key limiting factor in forest production across Europe (Harrison, 1989). However, little research has been carried out on changes in soil P cycling induced by pollutant inputs and in the balance between N and P nutrition of trees.
Our working hypothesis is that acidifying inputs, and the induced changes in soil acidity, decrease the availability of P in soil by reducing both soil solution P (due to increased P
0038-0717/00/$ - see front matterq2000 Elsevier Science Ltd. All rights reserved.
PII: S 0 0 3 8 - 0 7 1 7 ( 0 0 ) 0 0 1 5 9 - 0
* Corresponding author. Tel.:134-9-53-212551; fax:134-9-53-212141.
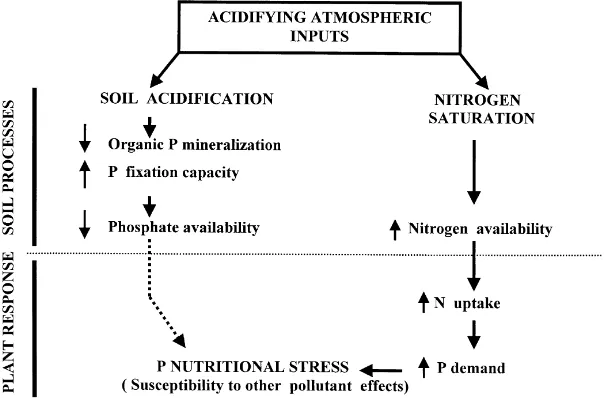
adsorption capacity) and the rate of organic phosphorus mineralisation. In addition, associated N saturation creates enhanced demand for P by the trees relative to the supply from the soil. The combination of these two factors will induce P deficiency and reduce tree growth (Fig. 1).
To test this hypothesis, the effects of acid-mist applied to tree canopies, on tree P nutrition, and soil P cycling and availability, were assessed in a field manipulation experi-ment in Scotland. Acid-mist treatexperi-ments had been applied for four consecutive years to a Sitka spruce plantation and had caused a significant reduction in stem diameter growth compared to non-acid-misted trees (Sheppard et al., 1995). Foliar analysis were carried out in an attempt to relate such reduction in stem area to the induction of nutrient deficien-cies (including P), but results were inconclusive (Sheppard et al., 1995). However, Carreira et al. (1997) applied a more sensitive root bioassay technique (based on the metabolic uptake of32P by excised roots) and demonstrated that acid-mist treated trees were under P nutritional stress relative to non-acid-misted trees. Additionally, acid-mist treatments were found to affect soil acidity (lower pH and marked changes in the cation exchange complex composition) and processes involved in the inorganic P subcycle (higher soil P adsorption capacity) (Carreira et al., 1997). Here, we report on changes in processes involved in the soil organic P cycling (phosphatase activity and organic P mineralisation).
2. Materials and methods
2.1. Study site and experimental design
The study site was a 12×12 block (2.5 m spacing), Sitka spruce (Picea sitchensis (Bong.) Carr), single clone, 19-year-old plantation, growing on a brown earth soil at
Glencorse Mains, south of Edinburgh (Scotland). The site had been previously used to evaluate the whole tree response of mature Sitka to acid-mist (Sheppard et al., 1994, 1995). Trees were grouped into five height classes, from the shortest (H1, mean 3.8 m) to the tallest (H5, mean 5.5 m), along a 68diagonal slope across the plot. Two blocks of four trees per height class were enclosed in open-top chambers equipped with side roller blinds that were pulled down only during period of treatment application. Twice a week from July to December 1990, and from April to Octo-ber 1991–92–93, chamOcto-bers received the equivalent of 2 mm precipitation as acid-mist consisting of an equimolar mixture of H2SO4and NH4NO3at pH 2.5 (3.2 mM H1, and
1.6 mM each of NH14; NO
2
3 and SO242; simulating
stan-dard, acidic, cloud water composition (Crossley et al., 1991). An extra chamber, enclosing a mix of tree height classes, was used to apply an unreplicated, double dose, acid-mist treatment (8 mm of mist/week). Eight individual trees for each height class, not enclosed within chambers, were selected as control (non-acid-misted) trees. Control trees received non-acid (pH 5.0) mist at 2 mm per spraying in 1993. Detailed information on the experimental layout, treatment applications, and previous results of the Glencorse study can be found elsewhere (Crossley et al., 1991; Shep-pard et al., 1995; Carreira et al., 1997).
2.2. Soil sampling and general analyses
One year after the end of treatment applications, four soil samples (litter plus 0–5 cm depth) were collected using a 11.5 cm diameter core sampler from each treatment block (5 tree height classes×2 acid-mist doses — no acid-mist and standard acid-mist dose —, and 1 double acid-mist dose with mixed height classes). Within each acid-misting cham-ber, soil samples were taken at random but, to avoid edge
effects, collection was restricted to the inner “quadrat” between the stems of the four trees enclosed in each cham-ber. Similarly, soil samples were collected from within a 1.0 m radius in a direction selected at random beneath each of four, individual, non-acid-misted trees in each height class.
Litter and underlying mineral soil from each core sample were separated. Each bulk soil sample was weighed, sieved (2 mm), and the volume of the.2 mm fraction recorded to calculate bulk density. Soil subsamples were oven-dried (1058C, 24 h) to calculate moisture content, and then ignited in a muffle (5508C, 2 h) to calculate loss-on-ignition. Water holding capacity (WHC) was estimated from bulk density (BD) using the equation: WHC61.89p (BD)21.099
r2
0:96; n100; P,0:001; developed for forest soils
(Harrison and Bocock, 1981; Harrison, unpublished data). There were no significant differences between acid-mist dose-level treatments for soil moisture at the field 23:2^
1:2% gravimetric water content), for soil bulk density
(0.76 g cm23), and for soil WHC (83.7% gravimetric water content). Effective cation exchange capacity (CECe)
was measured on air-dried soil samples as the sum of bases plus exchangeable acidity displaced by 0.1 M BaCl2
(Hendershot and Duquette, 1986). pH and P fractionation analyses were carried out on fresh soil samples. Soil pH was measured in 1:1 H2O (McLean, 1982). Labile P fractions
were obtained by sequentially extracting soil samples with anion-exchange resins (Dowex 1-X8) and 0.5 M NaHCO3
(Hedley et al., 1982). Results are expressed as g P m22of 0– 5 cm depth soil using bulk density values of individual soil samples.
2.3. Phosphatase activity and net P transformation rates
Air-dried soil samples were wetted (25% gravimetric water content; 30% WHC) and pre-incubated at 208C for 21 days prior to the assessment of phosphatase activity. Phosphomonoesterase activity was determined following the method of Tabatabai (1994). Since the activity of phos-phatase enzymes is strongly affected by pH, standard assays are usually carried out at pH 6.5 or pH 11.0 (the average optimal pH for acid and alkaline phosphatase activity in a wide range of soils) to allow for inter-studies comparisons in terms of potential phosphatase activity. However, we were interested on effects induced by soil acidification under
acid-mist application. On this basis, rather than assaying all soils at a fixed pH in a buffered solution, we performed non-buffered assays in order to make pH conditions resem-ble the observed pattern of soil pH variation between acid-mist treatments (Table 1). The possibility that further pH changes occurred during the 1 h assay incubation cannot be fully discounted, although we expect that relative pH differ-ences between acid-mist treatments were maintained. These considerations should be taken into account when compar-ing the results reported here with those from other studies on potential activity at optimal pH. In any case, the results from brief, laboratory enzyme assays must not be interpreted beyond their scope as qualitative indicators of the changes induced by chronic, acid-mist inputs.
The effects of acid-misting tree canopies on potential soil P transformation rates was evaluated using a modification of the radiation–autoclaving–incubation procedure of Zou et al. (1992). Three subsets of two replicated, air-dried, height class-composite soil samples corresponding to the non-acid-mist, standard acid-mist dose and double acid-mist dose treatments were pre-incubated aerobically at 208C and 15% water content (approximately 20% WHC) for a period of three weeks to equilibrate microbial activity to each sample potential under these temperature and water poten-tial conditions. This pre-incubation period aimed to over-come transient effects of environmental conditions at the sampling date and sample pre-processing, and to ensure that differences in microbial activity were due to the ‘permanent’ effects of chronic, acid-mist treatments through the acidification of soils. Respiration rates were measured at the end of the pre-incubation period using an aqueous alkali trapping method (Zibilske, 1994). Then, one subset of samples, used as control, was brought to 30% gravimetric moisture content (approximately 35% of WHC) with deio-nised water. The other two of the three subsets of soils were sterilised. To sterilise soils, we used HgCl2-addition at a rate
of 1500 mg kg21 of dry soil (Wolf and Skipper, 1994), instead of G-radiation (Zou et al., 1992). To apply the HgCl2-treatment, the corresponding samples were evenly
sprinkled with the appropriate volume of a concentrated HgCl2solution to bring soils to 35% of WHC. All samples
were then incubated for an additional 7 days, and respiration rates were measured again to check for the degree of micro-bial sterilisation achieved by the HgCl2treatment. Then, one
of the two HgCl2-treated subsets of samples was autoclaved
Non-acid-misted 9.26Aa 5.09Aa 7.02Aa 89.8Aa 9.47Aa
Standard-dose 8.68Aa 4.76Bb 7.15Aa 76.3Bb 3.53Bb
to denature soil enzymes. Thus, three independent treat-ments were applied to soils beneath non-acid-misted, stan-dard-dose acid-misted, and double-dose acid-misted trees: (i) control; (ii) HgCl2-addition; and (iii) HgCl2-addition plus
autoclaving.
Immediately after treatment, all soils were examined for resin-extractable P during a 48 h incubation by shaking samples, 2 g, with 30-ml deionised water and one resin-bag (1-g of oven-dry-equivalent Dowex 1-X8 resins satu-rated with Cl2) in 50-ml centrifuge tubes in a reciprocal shaker (150 strokes min21). To ensure complete sterilisation of soils by the HgCl2 and the HgCl21autoclaving
treat-ments, and to mantain sterile conditions during incubation with resins, additional HgCl2 (2500 mg kg21 of dry soil)
was added along with the 30-ml of water to the correspond-ing centrifuge tubes. After incubation, resins were extracted with 30 ml 0.5 M HCl on a reciprocal shaker for 1 h. Phos-phate concentration in the solutions was analysed using the ascorbic acid–molybdenum blue method of John (1970).
Phosphorus extracted by resins from the control soils reveals the net balance between solubilisation of inorganic P, mineralisation of organic P, and immobilisation of solu-tion P. Resin-extractable P from the HgCl2-treated samples
results from the sum of solubilised P and mineralised P (HgCl2-sterilisation avoids microbial immobilisation of
solution P). Phosphorus from the HgCl2-treated plus
auto-claved soils comes from the solubilisation of inorganic P only (autoclaving of HgCl2-treated soils additionally avoids
P mineralisation by phosphatase enzymes), and gives an estimate of net P solubilisation rate. The difference in resin-extractable P between the HgCl2-addition and the
HgCl2-addition plus autoclaved treatments is an estimate
of gross P mineralisation rate, whereas the difference between the HgCl2-addition and control treatments is an
estimate of P immobilisation rate.
To check for the direct effect of HgCl2-addition and
auto-claving on resin-extractable P, three subsamples of 2 g moist soil from each of the control, HgCl2-addition, and
HgCl2-addition plus autoclaving treatments were extracted
with anion exchange resin bags as above except that extrac-tion time was only 1 h. If there were differences in resin-P concentrations before and after HgCl2-addition and HgCl2
-addition plus autoclaving, correction factors were applied in the calculation of P transformation rates to allow for direct treatment effects (Zou et al., 1992). Effects of treatments on phosphatase activity were also tested.
2.4. Statistical analysis
A 2-way ANOVA was performed to determine for the effects of acid-misting (non-acid-misting, and standard-dose acid-misting) and tree height class (H1–H5) on soil and litter P fractions. A 1-way ANOVA was performed pooling data from different height classes to determine the dose-effect of acid-mist application (non-acid-misting, stan-dard acid-mist dose, and double acid-mist dose). When no significant effect of height class was found in the previous analysis, unweighted means for all height classes combined were calculated for the non-mist and the standard acid-mist dose treatments, and compared to the double acid-acid-mist dose treatment. Otherwise, we used in the analysis non-acid-misting and standard acid-mist dose weighted means with the same proportion of height classes as in the double acid-mist dose chamber (2 H3:1 H4:1 H5).
To analyse data resulting from the application of the Zou et al. (1992) method, an ANOVA was first applied to test for significant effects of treatments on resin-extractable P (comparing the amount of P extracted by resins during 1 h in the control, HgCl2-treated, and HgCl2-treated plus
auto-claved soils). When significant differences appeared, correc-tion factors were applied to estimate net P solubilisacorrec-tion rate, gross P mineralisation rate, and P immobilisation rate (Zou et al., 1992).
ANOVA requirements of normality (Kolmogorov–Smir-nov test) and homogeneity (Bartlett–Box F test) were checked at a0:01:For variables that did not meet such
requirements, log (x11) transformation was enough to achieve normality and homogeneity.
3. Results
3.1. Soil acidity
There were no differences in loss-on-ignition between
Table 2
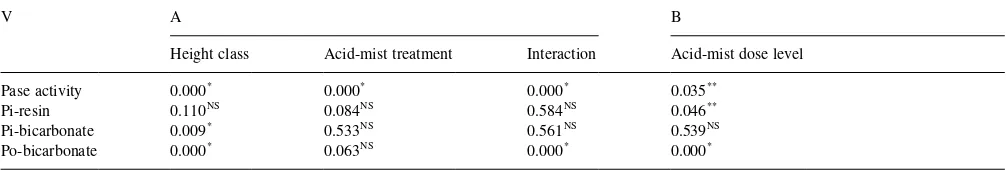
ANOVA results on effects of acid-mist treatment and tree height class (A), and on acid-mist dose level applied to the tree canopy (non-acid-misting, standard-dose, and double-dose acid-misting treatments) (B) for soil phosphatase activity and soil labile P pools of the Glencorse experiment (see Section 2.4 for further details)
V A B
Height class Acid-mist treatment Interaction Acid-mist dose level Pase activity 0.000* 0.000* 0.000* 0.035**
Pi-resin 0.110NS 0.084NS 0.584NS 0.046** Pi-bicarbonate 0.009* 0.533NS 0.561NS 0.539NS Po-bicarbonate 0.000* 0.063NS 0.000* 0.000*
*P
#0:01; 2-way ANOVAF-tests. **P
dose-level acid-mist treatments (Table 1). Soil pH (1:1 H2O)
decreased significantly P#0:001 with increasing
acid-mist dose, from 5.09 in soils under non-acid-acid-misted trees to 3.93 in soils under double-dose acid-mist treated trees. Acid-mist treatments induced little change in soil CEC, which was relatively low in all soils (low buffering capa-city). Consequently, marked changes in the proportion of individual exchangeable cations were observed. Base satura-tion decreased from near 90% to less than 30%, and the (Ca1
Mg):Al ratio decreased from 9.5 to 0.4, among the non-acid-misting and the double-dose acid-non-acid-misting treatments.
3.2. Phosphatase activity and respiration rates
The effects of tree height-class, acid-mist treatment, and interaction between them, on the potential rates of phospha-tase activity at the soil pH were all highly significant (Tables 2 and 3). Mean phosphatase activity (all height classes combined) was lower in soils under acid-misted trees (255.1mg pNP g21h21) than in soils under non-acid-misted trees (400.6mg pNP g21h21), although this trend was only consistent for the smallest height classes (H1–H3). Soil phosphatase activity in the double-dose acid-mist treatment was also significantly lower than in the non-acid-mist treatment.
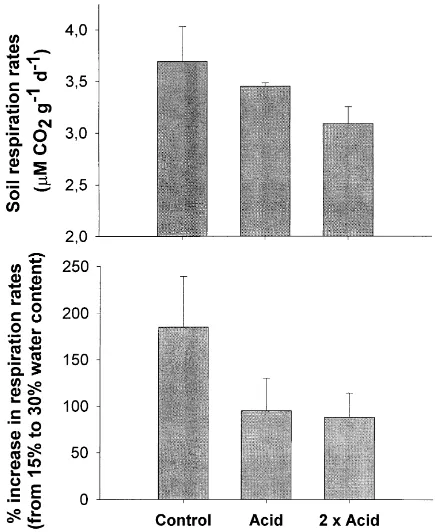
Soil respiration rates showed a decreasing trend (although not significant) with the dose of applied acid-mist (from 3.69 in the non-acid-mist to 3.09mmol CO2g21day21 in
the double-dose treatments, at 30% moisture content)
Acid-mist treatment Height classes
H1 H2 H3 H4 H5 All combined
(unweighted means)
H3:H4:H5
(2:1:1, weighted means)
Phosphatase activity
Non-acid-misting 496.8 (42.8) 361.6 (46.4) 383.0 (48.1) 379.9 (8.3) 382.0 (37.6) 400.6 382.0A(17.5) Standard-dose 137.4 (28.8) 198.6 (19.0) 116.4 (25.8) 366.8 (50.1) 456.1 (57.4) 255.1 263.9B(43.3)
Double-dose – – – – – – 279.9AB(8.1)
Resin-extractable P
Non-acid-misting 0.68 (0.16) 0.51 (0.06) 0.54 (0.17) 0.35 (0.07) 0.42 (0.13) 0.50A(0.06) –
Standard-dose 0.41 (0.05) 0.53 (0.10) 0.36 (0.05) 0.23 (0.03) 0.40 (0.03) 0.39AB(0.03) –
Double-dose – – – – – – 0.29B(0.04)
Bicarbonate-extractable inorganic P
Non-acid-misting 1.49 (0.20) 1.32 (0.09) 1.36 (0.21) 1.04 (0.09) 0.99 (0.09) – 1.19A(0.09)
Standard-dose 1.29 (0.10) 1.34 (0.10) 1.22 (0.06) 0.94 (0.09) 1.17 (0.04) – 1.14A(0.04)
Double-dose – – – – – – 1.30A(0.03)
Bicarbonate-extractable organic P
Non-acid-misting 2.59 (0.06) 2.58 (0.06) 2.43 (0.13) 2.09 (0.03) 1.89 (0.12) – 2.21A(0.08) Standard-dose 3.12 (0.21) 2.28 (0.07) 2.14 (0.13) 2.23 (0.07) 2.47 (0.08) – 2.25A(0.06)
Double-dose – – – – – – 2.94B(0.13)
Fig. 2. CO2release (mmol CO2g21day21) at 30% moisture content (a), and
increment in CO2release (expressed as % of initial) after increasing
(Fig. 2a). The increment in soil microbial activity (measured as respiration rate) in response to changing the sample moisture from 20 to 35% of water holding capacity was significantly higher in soils under non-acid-misted-trees (185% increment from initial respiration rate) than in soils under acid-misted soils (about 90% increment) (Fig. 2b).
3.3. Labile P fractions
The concentration of the most easily available inorganic P fractions (resin-extractable P, expressed asmg P g21dw soil; data not shown) decreased significantly from soils under the smallest to soils under the tallest trees P
0:001;and was a 30% lower in soils under standard-dose
acid-misted trees than in soils under non-acid-misted trees
P0:04:No significant effects of tree height-class were
observed if data are expressed on an area basis (Tables 2 and 3). However, the size of the resin-extractable P pool in 0– 5 cm depth soils significantly P0:046 and linearly
decreased with increasing acid-mist dose levels (from 0.50 g m22 in soils under non-acid-misted trees to 0.29 g m22 in soils under double-dose acid-misted trees)
(Fig. 3). In contrast, the amount of labile organic P (extracted with bicarbonate) significantly increased in soils under acid-mist treated trees (from 2.21 g m22 in soils under non-acid-misted trees to 2.94 g m22 in soils under double-dose acid-misted trees, representing a 33% increment in the pool size; Fig. 3). The amount of bicarbo-nate-extractable inorganic P was significantly higher in soils under the smallest than under the tallest trees. Acid-misting did not affect the amount of bicarbonate-extractable inor-ganic P.
3.4. P transformation rates
3.4.1. Direct effects of HgCl2addition and autoclaving
treatments
The effects of HgCl2addition and autoclaving on
resin-extractable P in soils under non-acid-misted, standard-dose and double-dose acid-misted trees were not significant (1.9 and 1.3mg of increment in resin-extractable P g21 in the HgCl2-addition and HgCl2-addition plus autoclaving
treat-ments compared to the control treatment, respectively). Autoclaving was effective in denaturing soil phosphatase
Fig. 3. Pool sizes of soil labile P fractions as a function of tree-height class (a) and acid-mist dose level (b). Data are expressed as the difference % from the non-acid-misting treatment.
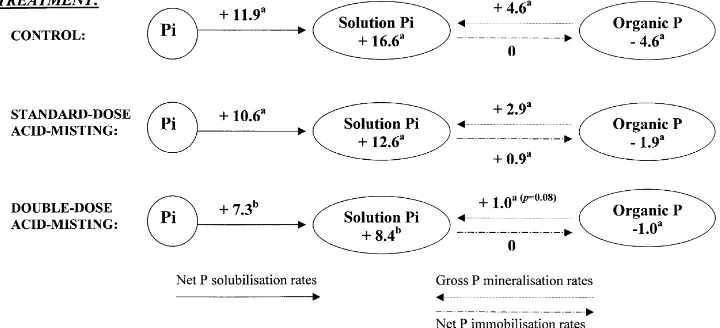
Fig. 4. Net solubilisation, gross mineralisation, and immobilisation rates of P in soils under non-misted, standard-dose misted, and double-dose acid-misted trees of the Glencorse field experiment. Circles are P pools and arrows indicate processes. Numbers in circles are the net changes in P pool sizes (mg P g21soil). Numbers by arrows are P transformation rates inmg P g21soil days21. Different letters for the same process indicate significant differences
enzymes as indicated by a reduction of soil phosphatase activity to zero in autoclaved samples. The addition of HgCl2did not affect soil phosphatase activity (mean
phos-phatase activities of non-treated and HgCl2-treated soils
were 420.8 and 431.5mg pNP g21h21, respectively). Addi-tion of HgCl2 followed by a one-week incubation at 35%
WHC markedly inhibited respiration rates in all soils (from 60 to 90% inhibition respect to the corresponding non-HgCl2-treated soils), although did not reduced it to zero.
Thus, in order to ensure that microbial immobilisation of solution P was precluded in the HgCl2-treated soils during
the 48 h incubation with resins, additional HgCl2
(2500 mg kg21of dry soil) was added to the corresponding distilled water–resin bag–soil slurries (see Section 2).
3.4.2. Net solubilisation, gross mineralisation and immobilisation rates
Net P solubilisation rates were higher than gross P miner-alisation and net P immobilisation rates (Fig. 4). Net P solubilisation rates were significantly lower a0:05 in
soils under double-dose acid-misted trees (7.3mg P g21day21) than in soils under non-acid-misted trees (11.9mg P g21day21). Gross P mineralisation rates were more than four times greater in the non-acid-misting treat-ment than in the double-dose acid-misting treattreat-ment P
0:08:Net P immobilisation rates were very low in all cases.
Net changes in soil solution P were all positive and
decreased significantly with increasing dose of acid-mist application. Net changes in organic P pools were all nega-tive. Soils under non-acid-misted trees showed the highest net decrease in organic P, whereas soils under double-dose acid-misted trees showed almost no variation in organic P pools. However, the effect of acid-mist dose did not achieved significance at thea0:05 level.
4. Discussion
Excluding plant P uptake, and P inputs to and outputs from the forest ecosystem, P availability is the net result of inorganic P solubilisation, P immobilisation and organic P mineralisation in the soil. In this study, we have tested the hypothesis that changes in soil phosphatase activity and changes in net P transformation rates resulting in a lower P supply to the soil solution are causal mechanisms for the induction of P deficiency and reduced tree-diameter growth that had been previously reported (Sheppard et al., 1995; Carreira et al. 1997) in a sitka-spruce plantation subjected to the application of acidified mist to the forest canopy.
We found that potential phosphatase activity (mg pNP g21 dw soil) at the soil pH was significantly lower in soils under acid-misted than under non-acid-misted trees. The pattern of enzyme activity inhibition with increas-ing acid-mist dose applied to the forest canopy is even more
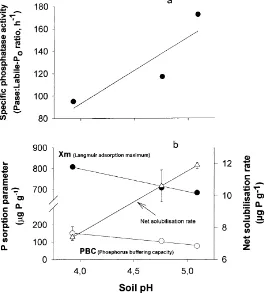
pronounced when expressed in terms of specific phospha-tase activity (Pase:labile Poratio) (Fig. 5), indicating that the
availability of natural sources of substrate is not acting as a limiting factor for the amount of phosphatase enzymes in the acidified soils. Potential enzyme activity rates measured at saturating substrate concentration (as in the phosphatase assay conditions used here) are equivalent to the enzyme
Vmax and, thus, can be considered as indicative of the
enzyme concentration in the soil (Tabatabai, 1994). On the other hand, phosphatase enzyme production is known to be enhanced by low availability of inorganic P in the soil (Stevenson, 1986). However, the smaller size of the most-easily available inorganic P pool in soils under acid-misted trees was not paired by a higher potential phosphatase activ-ity in such soils. We also found that soils under acid-misted trees showed lower respiration rates and an attenuated CO2
release increment in response to increases in the moisture content of the soils. Soil respiration can be used as a measure of the activity of soil microbes (Zibilske, 1994). All this suggests that soil acidification induced by acid-mist-ing treatments have affected the role of phosphatase enzymes in the soil P cycling not only by pH-induced changes in their activity but also by lowering the rate of enzyme production by the microbial biomass.
Changes in phosphatase activity can result in changes on P mineralisation only if P mineralisation is limited by an insufficient quantity of phosphatase (otherwise, the potential decrease in the rate of P mineralisation by a lower phospha-tase activity might be compensated by a higher amount of phosphatase enzyme). This explains why some studies have shown no correlation between phosphatase activity and net P mineralisation rates (e.g. Trasar et al., 1991), whereas others have shown otherwise (e.g. Harrison, 1982; this study). We found that gross P mineralisation rates showed a decreasing trend with increasing acid-mist dose, that was not compensated by changes in the P immobilisation rates. These findings support the hypothesis that the combined effect of soil acidification on the activity and size of the phosphatase enzyme pool, and on the microbial activity, may affect P availability by slowing the supply of P to the soil solution resulting from the net balance between P mineralisation and immobilisation.
Changes in the net balance between P fixation to and P solubilisation from the mineral phase in soils may also alter the supply of P to the soil solution. We found that net P chemical solubilisation rates were significantly higher in non-acid-misted than in acidified soils. This is consistent with previous results which showed an increase in P sorp-tion-fixing capacity in soils under acid-misted trees, as shown by the Langmuir’s adsorption maxima and the phos-phorus buffering capacity (Carreira et al., 1997) (Fig. 5). Increases in soil P sorption capacity induced by increasing soil acidification in areas where the initial soil pH was already slightly acid (the Glencorse soils had a mean pH value of 5.09) has been found elsewhere (Pare´ and Bernier, 1989b). The overall combination of changes in P
transfor-mation rates reported here demonstrates that acidic-mist inputs may induce a decrease in soil P availability through the synergic effect of soil acidification in slowing the rate of processes involved in both the geochemical and the biochemical P cycle.
The appearance of P deficiencies in forest stands receiv-ing high ‘natural’ acidifyreceiv-ing atmospheric inputs has been reported both in North America and Europe. Bernier and Brazeau (1988) reported P deficiencies in sugar maple stands in the Quebec area. Mohren et al. (1986) and Houdijk and Roelofs (1993) found generalised P deficiencies in forest stands in the Netherlands, especially in Douglas fir stands. Harrison et al. (1999) reported a general link between ‘thin’ canopy condition and phosphorus stress in beech, Scots pine and Sitka spruce stands growing on a range of comparable soils in the UK. Several causal mechanisms were proposed to explain the association between the appearance of P deficiencies in forest trees and high atmospheric acidifying inputs (Fig. 1). On the one hand, soil acidification is hypothesised to cause (i) increase in the inorganic P sorption capacity of the soils and concomitant decreases in P solubility (Van Breemen et al., 1983; Pare´ and Bernier, 1989b); and (ii) decreases in the rate of organic P mineralisation (Harrison, 1982; Pare´ and Bernier, 1989a). On the other hand, the induction of N saturation associated to high N-containing, atmospheric, acidic inputs, may enhance the tree demand for other nutri-ents (especially P), potentially resulting in nutritional imbal-ances (Nihlgard, 1985; Binkley et al., 1989). However, the establishment of clear linkages between observed P defi-ciency symptoms and any of the proposed causal mechan-isms has been difficult in the above-cited, field-based studies describing ‘natural’ variability.
stress detected on acid-mist treated trees also results, at least partially, from enhanced demand of P by the trees due to the presence of N in the applied acid-mist cannot be fully discounted.
Acknowledgements
We wish to thank L. Sheppard, A. Crossley, N. Cape and F. Harvey (I.T.E., Bush, Scotland) for providing access to the Glencorse Field Experiment, as well as information about the experimental design, results and general conclu-sions from their previous studies at the site. This research was made possible by a bursary (ref. EV5V-CT-94-5229) for J. Carreira from the ENVIRONMENT programme of the Commission of the European Communities.
References
Bernier, B., Brazeau, M., 1988. Nutrient deficiency symptoms associated with sugar maple dieback and decline in the Quebec Appalachians. Canadian Journal of Forest Research 18, 762–767.
Binkley, D., Driscoll, C.T., Allen, H.L., Schoeneberger, P., McAvoy, D., 1989. Acidic Deposition and Forest Soils: Context and Case Studies of the Southeastern United States. Springer, New York.
Carreira, J.A., Harrison, A.F., Sheppard, L.J., Woods, C., 1997. Reduced soil P availability in a Sitka spruce (Picea sitchensis(Bong.) Carr) plantation induced by applied acid-mist: significance in forest decline. Forest Ecology and Management 92, 153–166.
Crossley, A., Wilson, D.B., Sheppard, L.J., Leith, I.D. Cape, J.N. 1991. Effects of acid mist on mature Sitka spruce, Interim report, Department of the Environment, UK, 29pp.
Foy, C.D., Chaney, R.L., White, M.C., 1978. The phisiology of metal toxicity. Annual Reviews Plant Physiology 29, 511–566.
Harrison, A.F., 1982. Labile organic phosphorus mineralization in relation-ship to soil properties. Soil Biology & Biochemistry 14, 343–351. Harrison, A.F. 1989. Phosphorus distribution and cycling in European
forest ecosystems. In: Tiessen, H. (Ed.), Phosphorus Cycles in Terres-trial and Aquatic Ecosystems. Regional Workshop I: Europe, SCOPE-UNEP, pp. 42–76.
Harrison, A.F., Bocock, K.L., 1981. Estimation of soil bulk-density from loss-on-ignition. Journal of Applied Ecology 18, 919–927.
Harrison, A.F., Helliwell, D.R., 1979. A bioassay for comparing phos-phorus availability in soils. Journal of Applied Ecology 16, 497–505. Harrison, A.F., Carreira, J.A., Poskitt, J.M., Robertson, S.M.C., Smith, R.,
Hall, J., Hornung, M., Lindley, D.K., 1999. Impacts of acidifying pollu-tant inputs on forest canopy condition in the UK: possible role of P limitations. Forestry 72, 367–377.
Hedley, M.J., Stewart, J-W.B., Chauhan, B.S., 1982. Chamges in inorganic and organic soil phosphorus fractions induced by cultivation practices and by laboratory incubations. Soil Science Society of America Journal 46, 970–976.
Hendershot, W.H., Duquette, M., 1986. A simple barium chloride method for determining cation exchange capacity and exchangeable cations. Soil Science Society of America Journal 50, 605–608.
John, M.K., 1970. Colorimetric determination of phosphorus in soil and plant materials with ascorbic acid. Soil Science 109, 214–220. Kazda, M., 1990. Indications of unbalanced nitrogen nutrition of Norway
spruce stands. Plant and Soil 128, 97–101.
McLean, E.O., et al., 1982. Soil pH and Lime Requirement. In: Page, A.L., et al. (Eds.). Methods of Soil Analysis, Part 2, Chemical and Micro-biological Properties, 2nd ed. Book series no 9, Soil Science Society of America, Madison, MI, pp. 199–223.
Mohren, G.M., Van der Burg, J., Burger, F.W., 1986. Phosphorus defi-ciency induced by nitrogen input in Douglas fir in the Netherlands. Plant and Soil 95, 191–200.
Nihlgard, B., 1985. The ammonium hypothesis: an additional explanation to the forest dieback in Europe. Ambio 14, 2–8.
Pare´, D., Bernier, B., 1989a. Origin of the phosphorus deficiency observed in declining sugar maple stands in the Quebec Appalachians. Canadian Journal of Forest Research 19, 24–34.
Pare´, D., Bernier, B., 1989b. Phosphorus-fixing potential of Ah and H horizons subjected to acidification. Canadian Journal of Forest Research 19, 132–134.
Schulze, E.D., 1989. Air pollution and forest decline in a spruce (Picea abies) forest. Science 244, 776–783.
Sheppard, L.J., Leith, I.D., Cape, J.N., 1994. Effects of acid mist on mature grafts of Sitka spruce. Part I. Frost hardiness and foliar nutrient concen-trations. Environmental Pollution 85, 229–238.
Sheppard, L.J., Crossley, A., Harvey, F.J., Cape, J.N., Fowler, D. 1995. Long term effects of field exposure to acid mist on the performance of a single Sitka spruce clone. Final report, Department of the Environment, UK, 75pp.
Stevenson, F.J., 1986. Cycles of Soil: Carbon, Nitrogen, Phosphorus, Sulfur, Micronutrients. Wiley, New York (380pp).
Tabatabai, M.A., 1994. Soil Enzymes. In: Weaver, R.W. (Ed.). Methods of Soil Analysis, Part 2, Microbiological and Biochemical Properties, Book Series no 5, Soil Science Society of America, Madison, WI, pp. 775–833.
Tomlinson, G.H., 1990. Effects of acid deposition on the forests of Europe and North America. CRC Press, Boca Raton, FL (281pp).
Trasar, M.C., Gil, F., Carballas, T., 1991. Liming and the phosphatase activity and mineralization of phosphorus in an andic soil. Soil Biology & Biochemistry 23, 209–215.
Ulrich, B., Mayer, R., Khanna, P.K., 1980. Chemical changes due to acid precipitation in a loess-derived soil in central Europe. Soil Science 130, 193–199.
Van Breemen, N., Mulder, J., Driscoll, C.T., 1983. Acidification and alka-linization of soils. Plant and Soil 75, 283–308.
Van Dick, H.F.G., Roelofs, J.G.M., 1988. Effects of excessive ammonium deposition on the nutritional status and condition of pine needles. Physiologia Plantarum 73, 494–501.
Wolf, D.C., Skipper, H.D., 1994. Soil Sterilization. In: Weaver, R.W. (Ed.). Methods of Soil Analysis, Part 2, Microbiological and Biochemical Properties, Soil Science of America, Madison, WI, pp. 41–49. Zibilske, L.M., 1994. Carbon Mineralization. In: Weaver, R.W. (Ed.).
Methods of Soil Analysis, Part 2, Microbiological and Biochemical Properties, Book series no 5, Soil Science of America, Madison, WI, pp. 835–863.