MATERI STUDY GUIDE
BLOK EMERGENCY
2016
LECTURE 13 :
Trauma and Non Trauma Conditions
which Potentially Disabling and Life
Threatening
LECTURE 13 :
Trauma Principles
Jorge Fernandez, MD
Neil Rifenbark, MD
Key Points
•
Assess all trauma patients with a rapid primary survey followed by a more comprehensive
secondary evaluation.
•
Address all emergent l ife threats in a stepwise manner during the primary survey before
progressing to the next stage.
•
Treat hemodynamically unstable patients as hemorrhagic shock until proven otherwise.
•
Initiate aggressive volume resuscitation in all unstable patients while concurrently searching
for active sources of hemorrhage.
INTRODUCTION
Trauma is currently the fourth leading cause of death in the United States across all age
groups and the leading cause of death in patients under the age of 44 years. It is responsible for
more deaths in patients under the age of 19 years than all other causes combined.
Approximately 40% of all emergency department (ED) visits are for trauma-related complaints,
and the annual costs exceed $400 billion. Adding to these costs, permanent disability is actually
3 times more likely than death in this cohort.
Trauma is broadly classified by mechanism into blunt and penetrating varieties, with the
former more than twice as common as the latter. Regardless of mechanism, victims of
significant trauma present with a wide range of complex problems, and their proper care
necessitates a multidisciplinary approach, including emergency physicians, trawna surgeons,
and the appropriate subspecialties. Most trawna care delivery systems follow the Advanced
Trawna Life Support guidelines developed and maintained by the American College of
Surgeons.
The mortality rates for trawnatic injuries typically follow a trimodal distribution. Certain
injury patterns including major vascular injuries and high cervical cord disruption with
secondary apnea result in near immediate death. The second cohort of injuries, including
conditions
such as pnewnothorax and pericardia! tamponade, typically evolve over a duration
of minutes to hours and are generally responsive to aggressive emergent intervention.
Septicemia and multisystem organ failure account for the third peak of fatalities and typically
occur weeks to months after injury.
CLINICAL PRESENTATION
History
about the number of shots heard and how many times the patient felt himself or herself get
shot.
Obtain a brief medical history using the AMPLE mnemonic. Ask about any known drug
allergies, current medication use,past medical history, last oral intake, and the immediate
events leading up to the injury. Keep in
mind that regardless of past history, elderly patients
have less physiologic reserve and are prone to higher rates of morbidity and mortality. In
females of childbearing age, always ask about the last menstrual period and assume that they
are pregnant until proven otherwise. Pregnant patients are at higher risk for domestic violence
and warrant unique considerations such as placental abruption, uterine rupture, the supine
hypotensive syndrome, and fetal distress or demise. Even apparently minor injuries including
falls and low-speed motor vehicle accidents can induce preterm labor or placental abruption.
Always ask about any evolving symptoms and identify the exact locations of pain, as this
will guide your physical exam. Patients with altered mental status should be treated as having a
traumatic brain injury until proven otherwise. Shortness of breath may indicate an underlying
pneumothorax ( PTX) , pulmonary contusion, or pericardia! tamponade. Chest pain may
indicate an underlying fracture of the ribs or sternum, hemothorax (HTX) , or traumatic aortic
injury (TAl) . Assume that patients with abdominal pain, hematemesis, or rectal bleeding have
an intra-abdominal visceral injury until proven otherwise. Patients complaining of hematuria
should be considered at a high risk for injury to the genitourinary (GU) tract. Neurologic
complaints including weakness and paresthesias may indicate an underlying spinal cord injury
or vascular dissection.
Physical Examination
The physical exam in major trauma patients is very systematic and can be divided into
primary and secondary surveys.
Primary Survey
The primary survey is a very brief and focused exam meant to identify and address
emergent life threats. It should proceed in a stepwise approach outlined by the ABCDE
mnemonic. Always treat any encountered abnormalities before proceeding to the next step in
the survey. If a patient decompensates at any point during his or her clinical course, return to
the beginning of the primary survey and reassess. Assume an unstable cervical spine injury in all
major t rauma victims until proven otherwise and immediately immobilize on presentation.
Assess the airway for patency. Signs of potential airway compromise include pooling
pharyngeal secretions, intraoral foreign bodies, stridulous or gurgling respirations, obvious
oropharyngeal burns, significant midface, mandibular, and laryngeal fractures, and expanding
neck hematomas.
Evaluate the patient's breathing and ventilation. Expose the chest and look for any signs
of asymmetrical orparadoxical chest wall movement, obvious deformities or open wounds,
tracheal deviation, and jugular venous distention. Auscultate the chest to confirm strong
symmetric bilateral breath sounds. The goal is to identify the presence of emergent life threats
including tension PTX, massive HTX, open PTX ( sucking chest wound), and flail c hest.
otherwise. Other findings concerning for hemorrhagic shock include pale, cool, and mottled
extremities and thready peripheral pulses. Auscultate the heart to detect distant heart tones
suggestive of an underlying pericardia! effusion. Identify all sources of active bleeding and
control with the application of direct pressure.
Perform a rapid neurologic exam, noting any evidence of disability or deficits. Document
the patient's level of consciousness; note the size, symmetry, and reactivity of the pupils; and
assess for any focal numbness or weakness. Perform a rectal exam to ensure adequate rectal
tone and determine the patient's Glasgow Coma Scale (GCS ) .
Completely expose the patient t o ensure that all potential life threats have been accounted for.
Carefully log-roll the patient to examine the back and rule out any occult penetrating injuries.
Once complete, immediately cover the patient with warm blankets to prevent the development
of hypothermia.
Secondary Survey
The secondary survey is a complete head- to-toe examination that should be performed
once the patient has been stabi lized. Examine the scalp, noting any lacerations, c ontusions,
and deformities. Check the visual a cuity, visual fields, extraocular movements, and pupil size
and reactivity. Assess the globe for penetrating injuries, lacerations, or proptosis. Examine the
mid-face, looking for evidence of fracture, lacerations, epistaxis, or septal hematomas. Look for
signs of basilar skull fracture such as hemotympanum, periorbital (raccoon eyes) or
retroauricular ecchymoses (Battle sign) , and cerebrospinal fluid (CSF) rhinorrhea or otorrhea.
CSF rhinorrhea can be detected with the use of a bedside "halo-test." Check for dental injuries
or evidence of mandibular fracture, including point tenderness, malocclusion, and sublingual
hematomas.
Inspect the neck, noting any signs of obvious tracheal deviation, laryngeal fracture,
subcutaneous emphysema, or expanding hematoma. Carefully palpate the cervical spine to
detect any point tenderness or bony step-offs. Re-inspect the chest, noting any signs of
contusions, asymmetry, paradoxical movement, or penetrating injury. Palpate the ribs and
sternum, checking for point tenderness, soft tissue crepitus, and bony deformity. Repeat
auscultation of the lungs and heart and document any abnormalities. Inspect the abdomen for
any signs of distention, contusions, or penetrating injury. Palpate all 4 quadrants to elicit any
tenderness, guarding, or rebound. Carefully assess the pelvis for signs of an unstable fracture by
gently compressing the iliac crests.
Inspect the perineum for lacerations, contusions, and hematomas. Perform vaginal and
rectal examinations to assess for gross blood or mucosal trauma. Note the rectal tone and
check the prostate for signs of displacement. Findings
consistent with urethral injury include
scrotal hematomas, blood at the urethral meatus, and a high-riding prostate.
Look for any signs of blunt or penetrating trauma to the extremities. Document any
open wounds, point tenderness, or obvious deformities. Range all joints, looking for abnormal
movement. Palpate all muscle compartments to detect any signs of developing tension. Roll the
patient and palpate the entire spine, noting any point tenderness or bony step-offs.
hematomas, corresponding peripheral nerve deficits, or delayed capillary refill. Patients with
soft signs for arterial injury require the measurement of the arterial pressure index (API ) . The
API can be calculated by dividing the systolic pressure of the affected extremity by the systolic
pressure of the contralateral unaffected limb. An API <0.9 is considered abnormal and
suggestive of arterial injury.
Finally, perform a comprehensive motor and sensory examination, reevaluate the pupils
and mental status, and recalculate the GCS.
DIAGNOSTIC STUDIES
Laboratory
Check a STAT bedside capillary blood glucose level in all patients with an abnormal
mental status as hypoglycemia can mimic a traumatic brain injury. Check a complete blood
count to assess an initial hematocrit and follow serially to assess for occult hemorrhage and
responsiveness to therapy. If available, obtain a bedside serum base deficit and lactate level
and follow serially to gauge responsiveness to therapy. Send a type and screen on all trauma
patients and crossmatch blood as necessary for patients likely to require transfusions or
operative intervention. Obtain a urinalysis to rule out gross hematuria, a bedside urine
pregnancy test in all female patients of childbearing age, and a urine toxicology screen in
patients with an abnormal level of consciousness. Check coagulation studies in all patients with
a clotting disorder (eg, patients on warfarin ) .
Imaging
Portable plain radiography is readily available a t most institutions for the rapid bedside
evaluation of trauma patients. Plain films are useful for diagnosing bony fractures including
unstable pelvic and spinal injuries; determining the trajectory of penetrating projectiles;
identifying HTX, PTX, or an abnormal mediastinum; and detecting the presence of
intraperitoneal free air.
Bedside ultrasonography provides a quick, highly sensitive, noninvasive,
and readily repeatable modality to detect occult hemorrhage. Perform a FAST exam to look for
signs of pericardial tamponade, PTX/HTX, and intraperitoneal bleeding.
Computed tomography ( CT) imaging has revolutionized the care of trauma patients.
That said, this modality does expose the patient to increased health care costs, potential
contrast reactions, and harmful ionizing radiation, so every effort should be made to limit its
use to patients whose condition truly warrants it. Furthermore, CT imaging should be pursued
only in patients who are stable enough to safely leave the resuscitation area for an extended
period of time. CT imaging of the head has become invaluable for the evaluation and treatment
of patients with traumatic brain injury. CT imaging of the chest is now the preferred modal ity
to evaluate patients with potential intrathoracic vascular emergencies ( eg, TAl) and evolving
pulmonary contusions. CT of the abdomen and pelvis can simultaneously detect solid viscus
injury (eg, liver and spleen) and intraperitoneal hemorrhage and determine the severity of
pelvic injuries. Finally, CT angiography has rapidly become the preferred means to exclude
vascular injuries in patients whose c ondition warrants some form of radiographic imaging (eg,
patients with "soft signs" for arterial injury) .
should be limited only to stable patients who can afford prolonged excursions outside of a
resuscitation arena.
PROCEDURES
The coordinated resuscitation of a critically ill trauma patient may require a multitude of
simultaneous interventions. The following procedures are described in detail in the
corresponding chapters: central line placement and volume resuscitation, needle thoracostomy
and chest tube insertion, emergent airway management, pericardiocentesis and ED
thoracotomy, and diagnostic peritoneal lavage.
Perform a retrograde urethrogram and cystogram in all patients with suspected urethral
and bladder injuries. Indications include straddle injuries, pelvic fractures, scrotal hematomas,
high-riding prostates, and blood at the urethral meatus. A urethrogram is performed by
injecting intravenous ( IV) dye into the urethral meatus while simultaneously capturing a pelvic
radiograph to detect any signs of urethral disruption (ie, contrast extravasation ) . Avoid the
insertion o f a Foley catheter into any patient with a demonstrated urethral injury without GU
consultation. For patients with an intact urethra, insert a catheter and distend the bladder with
up to 300 mL of diluted IV contrast while simultaneously capturing a pelvic radiograph
(cystogram) to detect any evidence of bladder rupture.
MEDICAL DECISION MAKING
Treatment
Evaluation and treatment should coincide during the primary survey. All life-threatening
conditions must be stabilized before further evaluation. Secure the airway in any patient with
signs of impending compromise.
Patients with a GCS
8 require endotracheal intubation to guard against obstruction
and/or aspiration. Examine the thoracic wall to identify any open PTX and cover with a 3 -sided
occlusive dressing to restore normal respiratory mechanics. Perform immediate needle
thoracos to my in all patients with signs of tension PTX, and place a chest tube in all patients
with evidence of traumatic PTX or HTX.
Patients with evidence of impaired circulation require large-bore IV access and
aggressive volume resuscitation (Lactated Ringer's or normal saline) . Attempt to determine the
class of hypovolemic shock to guide fluid resuscitation and identify the need for packed red
blood cell transfusion. Concurrently attempt to identify the source of hemorrhage to determine
the need for surgical intervention. Unstable patients with either clinical evidence ( hypotension,
distant heart sounds, j ugular venous distention) or ultrasonographic confirmation of
pericardia! tamponade require emergent pericardiocentesis.
DISPOSITION
Admission
The majority of blunt trauma victims require admission for observation to rule out
occult injuries not detected on either the primary and secondary surveys or CT imaging.
Hemodynamically unstable patients with positive focused assessment with sonography for
trauma (FAST) or CT imaging typically require operative intervention. Victims of penetrating
trauma generally require admission and operative intervention when the implements clearly
violate significant body cavities or injure vital anatomical structures.
Discharge
Blunt trauma patients with minor injuries who remain hemodynamically stable on serial
assessments can be
safely discharged. Penetrating trauma patients may be discharged provided
that the path of the implement clearly does not violate any significant body cavities nor
approach any vital anatomical structures. Always ensure that the patient is able to ambulate
and tolerate oral intake before discharge.
Reference
LECTURE 13 :
ABSTRACT
Crush Syndrome remains rare in European practice. It is however comm-on in areas of civil disorder and where the normal structures of society have given way to civil war or natural disaster. Western Doctors are becoming increasingly involved in such situations and there is no reason to believe that instances due to more conventional causes, such as collapse in the elderly or road traffic accidents will cease. For all these reasons it is important that clini-cians who deal infrequently with crush syndrome have access to appropriate guidelines. This consensus report seeks to provide such advice.
Key Words:Crush Syndrome, Renal Failure, Natural Disasters.
Introduction
Crush injuries and crush syndrome were first described in the English Language literature by Bywaters and Beall in 1941 (1), after several patients who had been trapped under rubble of buildings bombed in the Blitz subsequently died of acute renal failure. It has been described in numerous settings since, most commonly after natural disasters such as earthquakes, in war, and after buildings have collapsed as a result of ex-plosion. Crush syndrome is also seen foll-owing industrial incidents such as mining accidents and road traffic accidents. How-ever, crush syndrome is not confined to traumatic aetiologies, and has also been described following periods of crush by patients’ own body weight, after stroke or intoxication (2).
Most commonly in traumatic crush, the legs are affected, and less frequently the arms. Many authors believe that crush injury of the head and torso significant enough to
cause the syndrome is incompatible with life due to the inherent internal organ damage, but there are a few reported cases of such instances (3).
Crush syndrome bears many similarities to, but is distinct from, the syndrome caused by heat illness.
Definition
Following a search of the literature, it was felt that a definition of crush injury and crush syndrome was required.
Consensus view
“A crush injury is a direct injury resulting from crush. Crush syndrome is the systemic manifestation of muscle cell damage resulting from pressure or crushing”.
The severity of the condition is related to the magnitude and duration of the com-pressing force, and the bulk of muscle aff-ected. The definition is not, however, dep-endent on the duration of the force applied.
Examples of this relationship are firstly a patient whose legs are run over by the wheels of a truck. In this case the force is large, but the duration is very short. At the other extreme, there is the elderly patient who has suffered a stroke, falls, and lies in the same position for hours, sustaining a crush injury to the areas of the body on which they are lying. In this case, the force is relatively small, but crush syndrome may develop as a result of the prolonged period of pressure. Similar cases to this are described as a result of drug overdose (4).
Pathogenesis and clinical
features
The typical clinical features of crush syn-drome are predominantly a result of traumatic rhabdomyolysis and subsequent release of muscle cell contents. The mech-anism behind this in crush syndrome is the leakiness of the sarcolemmal membrane Lt Col I Greaves
Surg Lt Cdr JE Smith MBBS MSc MRCP RN
J R Army Med Corps 2003;149:255-259
Consensus Statement On The Early Management Of Crush
Injury And Prevention Of Crush Syndrome
I Greaves, K Porter, JE Smith
This paper reports the findings of a consensus meeting on Crush Injury and Crush Syndrome held in Birmingham on 31 May 2001, and co-ordinated by the Faculty of Pre-Hospital Care of the Royal College of Surgeons of Edinburgh.
The Voluntary Aid Societies The Ambulance Service Association The British Association for Immediate Care British Association for Emergency Medicine Faculty of Accident and Emergency Medicine The Royal College of Anaesthetists
The Royal College of Physicians
The Royal College of Surgeons of Edinburgh The Intensive Care Society
The Royal College of Nursing The Military
The Faculty of Pre-hospital Care
caused by pressure or stretching. As the sarcolemmal membrane is stretched, sod-ium, calcium and water leak into the sarcoplasm, trapping extracellular fluid inside the muscle cells. In addition to the influx of these elements into the cell, the cell releases potassium and other toxic substances such as myoglobin, phosphate and urate into the circulation (5).
The end result of these events is shock (discussed below), hyperkalaemia (which may precipitate cardiac arrest), hypocalc-aemia, metabolic acidosis, compartment syndrome (due to compartment swelling), and acute renal failure (ARF). The ARF is due to a combination of hypovolaemia with subsequent renal vasoconstriction, metabolic acidosis and the insult of nephrotoxic substances such as myoglobin, urate and phosphate.
Shock
Haemodynamic instability secondary to crush syndrome is multi-factorial. Firstly, many patients have other injuries, such as fractures of the pelvis or lower limbs, sufficient in themselves to cause hypovol-aemia. The sequestration of fluid into the affected muscle compartments has already been described, resulting in fluid shift from the intravascular to the intracellular comp-artments. This may cause hypovolaemia, as the intravascular volume is depleted. Elec-trolyte imbalances such as hyperkalaemia, hypocalcaemia and a metabolic acidosis will have a negatively inotropic effect, and there is also evidence that there is direct myocardial depression from other factors released when muscle cells are damaged (6).
Approach to treatment
Treatment of the crushed patient can be divided into two phases. The initial pre-hospital phase may, depending on the mechanism of injury, involve a prolonged extrication period. The second phase comm-ences on reaching a definitive medical care facility. In the case of prolonged on-scene time, or delay in transfer due to geographical reasons, some of the second phase guidelines may be employed in the pre-hospital environment.
Consensus view
Safety is the first priority when approaching an accident scene, and this is particularly relevant to situations where patients may have suffered crush injuries, as there may be danger from falling debris or risk of further building collapse.
Once the scene has been declared safe, in cases of mass casualties, a triage system (such as the triage sieve – Major Incident Medical Management & Support (MIMMS) (7)) should be used to prioritise casualties and assess the need for further treatment. For each individual casualty, an assessment of
Airway, Breathing and Circulation is the next priority. Attention must be given in trauma to the possibility of spinal injury and full spinal precautions should be maintained. Admin-istration of high flow oxygen by mask should be a priority in treatment, as should the arrest of any obvious external haemorrhage and the splinting of limb injuries.The patient should be exposed as necessary to assess and manage injuries. In a hostile environment, or where there is a risk of hypothermia, exposure should be as limited as possible. Assessment of distal neurovascular status is essential if exposure is to be kept to a minimum.
The patient should be released as quickly as possible, irrespective of the length of time trapped.
Fluid resuscitation
Once the initial primary survey has been performed, intravenous access should be obtained. If limb crush injury has occurred, and there is a likelihood of the patient developing crush syndrome, the following fluid guidelines should be followed. In the presence of life-threatening thoraco-abdominal injury, fluid resuscitation should be performed according to the Faculty’s previously published guidelines (8).
Consensus view
An initial fluid bolus of 2 litres of crystalloid should be given intravenously.This should be followed by 1-1.5 litres per hour.The fluid of choice is normal saline, warmed if possible, as this is established as the fluid carried by the majority of pre-hospital vehicles in the United Kingdom. Hartmann’s solution contains potassium and has a theoretical disadvantage of exacerbating hyperkalaemia. If possible, fluid should be started prior to extrication, however, gaining intravenous access and the administration of fluid should not delay extrication and transport to a definitive care facility. Early catheterisation should be considered, especially if there is a prolonged extrication or evacuation phase.
Once the patient reaches hospital, 5% dextrose should be alternated with normal saline to reduce the potential sodium load.
Analgesia
Consensus view
The use of medical teams including paramedics, nurses and doctors should be considered at an early stage, and appropriate analgesia should be given. This may involve the use of Entonox® initially, but most
patients will require intravenous analgesia such as an opiate, titrated against response. The use of ketamine, with or without the concomitant use of a benzodiazepine, is also an effective means of relieving pain, and may aid extrication.
First responders may give oral analgesia in the absence of senior clinical support.
256 Consensus Statement On Crush Injury
group.bmj.com
on August 18, 2016 - Published by
http://jramc.bmj.com/
Triage
Consensus view
Patients with crush injuries should be taken to a hospital with an intensive care facility and the equipment and expertise necessary to provide renal support therapy such as haemofiltration or dialysis.
Tourniquets
The use of tourniquets has a theoretical role in the management of these patients. If the release into the circulation of the contents of crushed muscle cells can be avoided, possibly with the use of a tourniquet, it may be of benefit. However, there is currently no available evidence to support this.
Consensus view
The use of tourniquets should be reserved for otherwise uncontrollable life threatening haemorrhage. There is no evidence at the moment to support the use of tourniquets in the prevention of reperfusion injury following extrication, or in the prevention of washing of the products of rhabdomyolysis into the circulation.
Amputation
Another theoretically advantageous measure is amputation of a crushed limb to prevent crush syndrome.
Consensus view
There is no evidence to support the use of amputation as a prophylactic measure to prevent crush syndrome. Reports from the literature suggest that even severely crushed limbs can recover to full function. If the limb is literally hanging on by a thread, or if the patient’s survival is in danger due to entrapment by a limb, amputation should be considered and appropriate expert advice sought.
Immediate in-hospital care
Consensus view
Patients should be assessed following normal Advanced Trauma Life Support (ATLS) guidelines (9). Baseline blood tests should be taken.These will include full blood count, urea and electrolytes, creatinine kinase, amylase, liver function tests, clotting screen and group and save (cross match if deemed appropriate). The patient should be catheterised and hourly urine measurements commenced. Central venous pressure and invasive arterial monitoring should be considered.
The use of solute-alkaline
diuresis
The development of acute renal failure in these patients significantly decreases the chances of survival (10). Every effort must be made, therefore, to prevent its occurrence. Alkalinisation of urine and the
use of a solute alkaline diuresis is accepted to be protective against the development of acute renal failure (11,12).
Consensus view
It is recommended that urine pH is measured, and kept above 6.5 by adding 50mmol aliquots of bicarbonate (50mls 8.4% sodium bicarbonate) to the intravenous fluid regime. Solute diuresis is affected by administering mannitol at a dose of 1-2g/kg over the first four hours as a 20% solution, and further mannitol should be given to maintain a urine output of at least 8 litres per day (300mls per hour). Fluid requirements are high, usually of the order of 12 litres per day, due to the sequestration of fluid in muscle tissue. Fluid should be given at approximately 500 mls/hour, but regular review of clinical parameters such as central venous pressure and urine output should dictate exact amounts of fluid given.
The maximum daily dose of mannitol is 200g, and it should not be given to patients who are in established anuria.
Children
Consensus view
There is very little evidence in the literature to guide the treatment of children suffering from crush injuries. In young children the difference in body proportions, namely the reduced contribution to the total percentage made by the limbs, may influence the incidence of crush syndrome. The fluid resuscitation guidelines from Advanced Paediatric Life Support (APLS) (13) of an initial bolus of 20mls per kg should be followed in these patients.
The elderly and patients with
co-morbidity
Consensus view
In the elderly, and those with pre-existing medical conditions such as cardiac failure, fluid replacement must be tailored to requirements and given with caution. Close monitoring of the clinical state of the patient, and regular review of fluid requirements is essential in these patients.
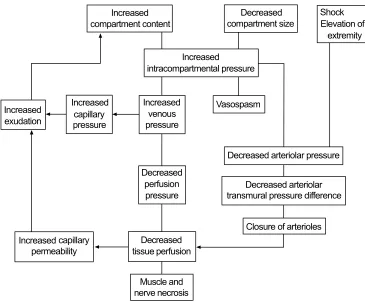
Compartment syndrome
The development of compartment syndrome in crush injury is due to the uptake of fluid into damaged muscle tissue contained within the restricted compart-ment. Once compartment pressure exceeds capillary perfusion pressure at about 30
-40mm Hg, the tissue inside the
compartment becomes ischaemic, and compartment syndrome develops.
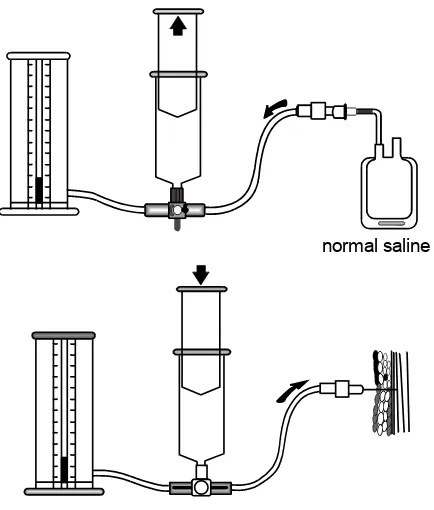
The traditional treatment of compartment syndrome is fasciotomy (4), but there is now evidence that initial treatment with mannitol may decompress compartment syndrome and avoid the need for surgery (5,12).
I Greaves, K Porter, JE Smith 257
group.bmj.com
on August 18, 2016 - Published by
http://jramc.bmj.com/
Consensus view
In patients with compartment syndrome due to crush injury, in the absence of neuro-vascular compromise, a trial of mannitol therapy should be instigated, but a specialist opinion should be sought early.
Hyperbaric oxygen therapy
There is theoretical and limited experimental evidence that hyperbaric oxygen therapy may improve wound healing and reduce the need for multiple surgical procedures in crush injury (14).
High concentrations of O2cause systemic
vasoconstriction but continue to deliver adequate O2 delivery. In a similar fashion,
nitric oxide synthase inhibitors may also have a role in preventing excessive vasodilatation in the crushed muscle and the consequent increase in third space fluid losses (15).
Consensus view
Logistically hyperbaric oxygen treatment has limited application. Patients with no sig-nificant co-morbidity, and who can be man-aged in a hyperbaric chamber where the fac-ility is available, may be treated with hyper-baric oxygen therapy. It is recommended that treatment options are discussed with the local hyperbaric unit. This is not recomm-ended as first line treatment. Patients should, however, receive high flow oxygen, unless there is a specific contra-indication.
Further management
Consensus view
In many cases, intensive care support will be required for the complications of crush syndrome. If the patient becomes oligo- or anuric, it is likely that they will require haemofiltration or dialysis.
Multiple casualties
Consensus view
In the civilian environment in the United Kingdom, there will be a huge strain on intensive care facilities if there are multiple crushed casualties. A policy should be drawn up to prepare for the dispersal of these casualties on both a National and Inter-national level should an incident occur. Further information is available in Better’s review of 1999 (5).
Areas identified for future
research
Use of tourniquets
Is there a role for the tourniquet post or pre extrication of the crush injury casualty? The use of an animal model of crush injury was suggested, to assess the suitability of tourniquet administration. Comparison of tourniquet placement versus no tourniquet in delayed intravenous fluid administration was suggested as a further research option. Are there any further deleterious effects due
to the increased ischaemia times involved in application of a tourniquet?
Could cooling the limb be used in order to slow cellular respiration and consequently decrease oedema, compartment syndrome and improve limb viability?
Tourniquet effectiveness was highlighted as a potential shortfall in their use. There is a requirement to perform a literature search into tourniquet usage, in particular regarding their use in Biers blocks, in order to determine the effectiveness of certain types of tourniquet and the leakage rates of drugs, past the tourniquet. This may assist in establishing the likelihood of potassium leaking into the systemic circulation.
Fluid administration
Types of fluid currently used for admini-stration include: normal saline, Hartmann's, Dextran or starches. What is the correct amount of fluid to be giving? Should we be looking at urine output, absolute volume intake or acidity of urine as a guide to fluid administration? Oedema occurring is secondary to massive fluid administration and may be detrimental. At what stage do we need to worry about this? What effect does this have on compartment syndrome?
Prognostic indicators
Creatinine kinase, myoglobinaemia and amy-lase have been suggested as prognostic indicators, although it is not clear that they can predict outcome at an early enough stage to allow effective intervention. The use of microalbuminuria as a prognostic indicator of crush syndrome was suggested.
Hyperbaric oxygen therapy
Use of the Institute of Naval Medicine was suggested in order to evaluate the merits of this treatment modality. In view of the scarcity of this resource around the country it did not meet with a great deal of support.
Bicarbonate administration
Early administration of bicarbonate intra-venously is thought to decrease metabolic acidosis and promote alkalisation of urine which decreases the precipitation of myo-globin in the renal tubules. Administration of bicarbonate immediately post-extrication, in anticipated metabolic acidosis, was dis-cussed. Has this been shown to be beneficial? Are there any detrimental features? What would be the appropriate and safe doses to use? Is there a role for the combined use of acetazolamide in order to prevent metabolic alkalosis following bicarbonate admini-stration?
Mannitol and compartment syndrome There is anecdotal evidence in the literature that due to the high complication rate in performing fasciotomy for compartment syndrome in crushed patients, they are best
258 Consensus Statement On Crush Injury
group.bmj.com
on August 18, 2016 - Published by
http://jramc.bmj.com/
managed with mannitol alone. It is suggested that there is a noticeable difference in dia-meter and symptoms of the lower leg within 40 minutes of administration of IV mannitol (5). Fasciotomy should be reserved for refractory cases. The use of an animal model of a compartment syndrome is questioned due to the anatomical differences from hum-ans. Many animals that are commonly used as models for humans, such as pigs, sheep and dogs do not have fascial compartments. Primates share similarities but, ethically, would be more difficult to justify. Further information on existing animal experiment-ation relating to compartment syndrome is required prior to planning any further projects.
References
1. Bywaters EGL, Beall D. Crush injuries with impairment of renal function.BMJ1941;1: 427. 2. Michaelson M. Crush injury and crush syndrome.
World J Surg 1992;16: 899-903.
3. Hiraide A, Ohnishi M, Tanaka H et al. Abdominal and lower extremity crush syndrome.Injury1997;
28(9-10): 685-6.
4. Shaw AD, Sjolin SU, McQueen MM. Crush syndrome following unconsciousness: need for urgent orthopaedic referral.BMJ1994;309: 857-9.
5. Better OS. Rescue and salvage of casualties
suffering from the crush sydrome after mass disasters.Mil Med1999;164: 366-9.
6. Rawlins M, Gullichsen E, Kuttila K, Peltola O, Niinikoski J. Central hemodynamic changes in experimental muscle crush injury in pigs.Eur Surg Res1999;31: 9-18.
7. Major Incident Medical Management & Support (MIMMS) 2002. BMJ Bookshops, London. 8. Greaves I, Porter KM, Revell MP. Fluid
Resuscitation in Pre-Hospital Trauma Care: A Consensus View.J.R. Coll. Surg. Edinb. 47;2; 451-457.
9. Advanced Trauma Life Support for Doctors. American College of Surgeons 1997 Chicago. 10. Ward MM. Factors predictive of acute renal failure
in rhabdomyolysis. Arch Intern Med 1988; 148: 1553-1557.
11. Better OS. The crush syndrome revisited (1940-1990). Nephron 1990; 55: 97-103.
12. Better OS, Zinman C, Reis ND et al. Hypertonic mannitol ameliorates intracompartmental tamponade in model compartment syndrome in the dog.Nephron1991;58: 344-6.
13. Advanced Paediatric Life Support 2nd Edition 2001. BMJ Bookshops.
14. Bouachour G, Cronier P, Gouello JP, Toulemonde JL, Talha A, Alquier P. Hyperbaric Oxygen therapy in the management of crush injuries; a randomized double-blind placebo-controlled clinical trial. J Trauma1996;41(2): 333-9.
15. Rubinstein I, Abassi Z, Coleman R, Milman F, Winaver J, Better OS. Involvement of nitric oxide system in experimental muscle crush injury.J Clin Invest1998;101(6): 1325-33.
I Greaves, K Porter, JE Smith 259
group.bmj.com
on August 18, 2016 - Published by
http://jramc.bmj.com/
Of Crush Syndrome
Management Of Crush Injury And Prevention
Consensus Statement On The Early
I Greaves, K Porter and JE Smith
doi: 10.1136/jramc-149-04-02
2003 149: 255-259 J R Army Med Corps
http://jramc.bmj.com/content/149/4/255
Updated information and services can be found at:
These include:
service
Email alerting
box at the top right corner of the online article.
Receive free email alerts when new articles cite this article. Sign up in the
Notes
http://group.bmj.com/group/rights-licensing/permissions To request permissions go to:
http://journals.bmj.com/cgi/reprintform To order reprints go to:
http://group.bmj.com/subscribe/ To subscribe to BMJ go to:
group.bmj.com
on August 18, 2016 - Published by
http://jramc.bmj.com/
Page 1 of 8
Consensus Statement On The Early Management Of
Crush Injury And Prevention Of Crush Syndrome
*I Greaves, K Porter, JE Smith
Abstract
Crush Syndrome remains rare in European practice. It is however common in areas of civil disorder and where the normal structures of society have given way to civil war or natural disaster. Western Doctors are becoming increasingly involved in such situations and there is no reason to believe that instances due to more conventional causes, such as collapse in the elderly or road traffic accidents will cease. For all these reasons it is important that clinicians who deal infrequently with crush syndrome have access to appropriate guidelines. This consensus report seeks to provide such advice.
Key Words: Crush Syndrome, Renal Failure, Natural Disasters
Introduction
Crush injuries and crush syndrome were first described in the English Language literature by Bywaters and Beall in 1941 (1), after several patients who had been trapped under rubble of buildings bombed in the Blitz subsequently died of acute renal failure. It has been described in numerous settings since, most commonly after natural disasters such as earthquakes, in war, and after buildings have collapsed as a result of
explosion. Crush syndrome is also seen following industrial incidents such as mining accidents and road traffic accidents. However, crush syndrome is not confined to traumatic aetiologies, and has also been described following periods of crush by patients’ own body weight, after stroke or intoxication (2).
Most commonly in traumatic crush, the legs are affected, and less frequently the arms. Many authors believe that crush injury of the head and torso significant enough to cause the syndrome is incompatible with life due to the inherent internal organ damage, but there are a few reported cases of such
instances (3).
Crush syndrome bears many similarities to, but is distinct from, the syndrome caused by heat illness.
Definition
Following a search of the literature, it was felt that a definition of crush injury and crush syndrome was required.
Consensus view
Page 2 of 8
The severity of the condition is related to the magnitude and duration of the compressing force, and the bulk of muscle affected. The definition is not, however, dependent on the duration of the force applied. Examples of this relationship are firstly a patient whose legs are run over by the wheels of a truck. In this case the force is large, but the duration is very short. At the other extreme, there is the elderly patient who has suffered a stroke, falls, and lies in the same position for hours, sustaining a crush injury to the areas of the body on which they are lying. In this case, the force is relatively small, but crush syndrome may develop as a result of the prolonged period of pressure. Similar cases to this are described as a result of drug
overdose (4).
Pathogenesis and clinical features
The typical clinical features of crush syndrome are predominantly a result of traumatic rhabdomyolysis and subsequent release of muscle cell contents. The mechanism behind this in crush syndrome is the leakiness of the sarcolemmal membrane caused by pressure or stretching. As the sarcolemmal membrane is
stretched, sodium, calcium and water leak into the sarcoplasm, trapping extracellular fluid inside the muscle cells. In addition to the influx of these elements into the cell, the cell releases potassium and other toxic substances such as myoglobin, phosphate and urate into the circulation (5).
The end result of these events is shock (discussed below), hyperkalaemia (which may precipitate cardiac arrest), hypocalcaemia, metabolic acidosis, compartment syndrome (due to compartment swelling), and acute renal failure (ARF). The ARF is due to a combination of hypovolaemia with subsequent renal
vasoconstriction, metabolic acidosis and the insult of nephrotoxic substances such as myoglobin, urate and phosphate.
Shock
Haemodynamic instability secondary to crush syndrome is multi-factorial. Firstly, many patients have other injuries, such as fractures of the pelvis or lower limbs, sufficient in themselves to cause hypovolaemia. The sequestration of fluid into the affected muscle compartments has already been described, resulting in fluid shift from the intravascular to the intracellular compartments. This may cause hypovolaemia, as the intravascular volume is depleted. Electrolyte imbalances such as hyperkalaemia, hypocalcaemia and a metabolic acidosis will have a negatively inotropic effect, and there is also evidence that there is direct myocardial depression from other factors released when muscle cells are damaged (6).
Approach to treatment
Treatment of the crushed patient can be divided into two phases. The initial prehospital phase may, depending on the mechanism of injury, involve a prolonged extrication period.The second phase commences on reaching a definitive medical care facility. In the case of prolonged on-scene time, or delay in transfer due to geographical reasons, some of the second phase guidelines may be employed in the pre-hospital environment.
Consensus view
Safety is the first priority when approaching an accident scene, and this is particularly relevant to situations where patients may have suffered crush injuries, as there may be danger from falling debris or risk of further building collapse.
Page 3 of 8
in treatment, as should the arrest of any obvious external haemorrhage and the splinting of limb injuries. The patient should be exposed as necessary to assess and manage injuries. In a hostile environment, or where there is a risk of hypothermia, exposure should be as limited as possible. Assessment of distal neurovascular status is essential if exposure is to be kept to a minimum.
The patient should be released as quickly as possible, irrespective of the length of time trapped.
Fluid resuscitation
Once the initial primary survey has been performed, intravenous access should be obtained. If limb crush injury has occurred, and there is a likelihood of the patient developing crush syndrome, the following fluid guidelines should be followed. In the presence of life-threatening thoracoabdominal injury, fluid
resuscitation should be performed according to the Faculty’s previously published guidelines (8).
Consensus view
An initial fluid bolus of 2 litres of crystalloid should be given intravenously.This should be followed by 1-1.5 litres per hour.The fluid of choice is normal saline, warmed if possible, as this is established as the fluid carried by the majority of pre-hospital vehicles in the United Kingdom. Hartmann’s solution contains potassium and has a theoretical disadvantage of exacerbating hyperkalaemia. If possible, fluid should be started prior to extrication, however, gaining intravenous access and the administration of fluid should not delay extrication and transport to a definitive care facility. Early catheterisation should be considered, especially if there is a prolonged extrication or evacuation phase. Once the patient reaches hospital, 5% dextrose should be alternated with normal saline to reduce the potential sodium load.
Analgesia
Consensus view
The use of medical teams including paramedics, nurses and doctors should be considered at an early stage, and appropriate analgesia should be given. This may involve the use of Entonox® initially, but most patients will require intravenous analgesia such as an opiate, titrated against response. The use of ketamine, with or without the concomitant use of a benzodiazepine, is also an effective means of relieving pain, and may aid extrication.
First responders may give oral analgesia in the absence of senior clinical support.
Triage
Consensus view
Patients with crush injuries should be taken to a hospital with an intensive care facility and the equipment and expertise necessary to provide renal support therapy such as haemofiltration or dialysis.
Tourniquets
Page 4 of 8
Consensus view
The use of tourniquets should be reserved for otherwise uncontrollable life threatening haemorrhage. There is no evidence at the moment to support the use of tourniquets in the prevention of reperfusion injury following extrication, or in the prevention of washing of the products of rhabdomyolysis
into the circulation.
Amputation
Another theoretically advantageous measure is amputation of a crushed limb to prevent crush syndrome.
Consensus view
There is no evidence to support the use of amputation as a prophylactic measure to prevent crush syndrome. Reports from the literature suggest that even severely crushed limbs can recover to full function. If the limb is literally hanging on by a thread, or if the patient’s survival is in danger due to entrapment by a limb, amputation should be considered and appropriate expert advice sought.
Immediate in-hospital care
Consensus view
Patients should be assessed following normal Advanced Trauma Life Support (ATLS) guidelines (9). Baseline blood tests should be taken. These will include full blood count, urea and electrolytes, creatinine kinase, amylase, liver function tests, clotting screen and group and save (cross match if deemed appropriate). The patient should be catheterised and hourly urine measurements commenced. Central venous pressure and invasive arterial monitoring should be considered.
The use of solute-alkaline diuresis
The development of acute renal failure in these patients significantly decreases the chances of survival (10). Every effort must be made, therefore, to prevent its occurrence. Alkalinisation of urine and the use of a solute alkaline diuresis is accepted to be protective against the development of acute renal failure (11,12).
Consensus view
It is recommended that urine pH is measured, and kept above 6.5 by adding 50mmol aliquots of icarbonate (50mls 8.4% sodium bicarbonate) to the intravenous fluid regime. Solute diuresis is affected by
dministering mannitol at a dose of 1-2g/kg over the first four hours as a 20% solution, and further mannitol should be given to maintain a urine output of at least 8 litres per day (300mls per hour). Fluid requirements are high, usually of the order of 12 litres per day, due to the sequestration of fluid in muscle tissue. Fluid should be given at approximately 500 mls/hour, but regular review of clinical parameters such as central venous pressure and urine output should dictate exact amounts of fluid given. The maximum daily dose of mannitol is 200g, and it should not be given to patients who are in established anuria.
Children
Consensus view
There is very little evidence in the literature to guide the treatment of children suffering from crush injuries. In young children the difference in body proportions, namely the reduced contribution to the total
Page 5 of 8
The elderly and patients with comorbidity
Consensus view
In the elderly, and those with pre-existing medical conditions such as cardiac failure, fluid replacement must be tailored to requirements and given with caution. Close monitoring of the clinical state of the patient, and regular review of fluid requirements is essential in these patients.
Compartment syndrome
The development of compartment syndrome in crush injury is due to the uptake of fluid into damaged muscle tissue contained within the restricted compartment. Once compartment pressure exceeds
capillary perfusion pressure at about 30 - 40mm Hg, the tissue inside the compartment becomes ischaemic, and compartment syndrome develops.
The traditional treatment of compartment syndrome is fasciotomy (4), but there is now evidence that initial treatment with mannitol may decompress compartment syndrome and avoid the need for surgery (5,12).
Consensus view
In patients with compartment syndrome due to crush injury, in the absence of neurovascular compromise, a trial of mannitol therapy should be instigated, but a specialist opinion should be sought early.
Hyperbaric oxygen therapy
There is theoretical and limited experimental evidence that hyperbaric oxygen therapy may improve wound healing and reduce the need for multiple surgical procedures in crush injury (14).
High concentrations of O2 cause systemic vasoconstriction but continue to deliver adequate O2 delivery. In a similar fashion, nitric oxide synthase inhibitors may also have a role in preventing excessive vasodilatation in the crushed muscle and the consequent increase in third space fluid losses (15).
Consensus view
Logistically hyperbaric oxygen treatment has limited application. Patients with no significant co-morbidity, and who can be managed in a hyperbaric chamber where the facility is available, may be treated with hyperbaric oxygen therapy. It is recommended that treatment options are discussed with the local hyperbaric unit. This is not recommended as first line treatment. Patients should, however, receive high flow oxygen, unless there is a specific contra-indication.
Further management
Consensus view
In many cases, intensive care support will be required for the complications of crush syndrome. If the patient becomes oligo- or anuric, it is likely that they will require haemofiltration or dialysis.
Multiple casualties
Consensus view
Page 6 of 8
Areas identified for future research
Use of tourniquets
Is there a role for the tourniquet post or pre extrication of the crush injury casualty? The use of an animal model of crush injury was suggested, to assess the suitability of tourniquet administration. Comparison of tourniquet placement versus no tourniquet in delayed intravenous fluid administration was suggested as a further research option. Are there any further deleterious effects due to the increased ischaemia times involved in application of a tourniquet?
Could cooling the limb be used in order to slow cellular respiration and consequently decrease oedema, compartment syndrome and improve limb viability?
Tourniquet effectiveness was highlighted as a potential shortfall in their use. There is a requirement to perform a literature search into tourniquet usage, in particular regarding their use in Biers blocks, in order to determine the effectiveness of certain types of tourniquet and the leakage rates of drugs, past the tourniquet. This may assist in establishing the likelihood of potassium leaking into the systemic circulation.
Fluid administration
Types of fluid currently used for administration include: normal saline, Hartmann's, Dextran or starches. What is the correct amount of fluid to be giving? Should we be looking at urine output, absolute volume intake or acidity of urine as a guide to fluid administration? Oedema occurring is secondary to massive fluid administration and may be detrimental. At what stage do we need to worry about this? What effect does this have on compartment syndrome?
Prognostic indicators
Creatinine kinase,myoglobinaemia and amylase have been suggested as prognostic indicators, although it is not clear that they can predict outcome at an early enough stage to allow effective intervention. The use of microalbuminuria as a prognostic indicator of crush syndrome was suggested.
Hyperbaric oxygen therapy
Use of the Institute of Naval Medicine was suggested in order to evaluate the merits of this treatment modality. In view of the scarcity of this resource around the country it did not meet with a great deal of support.
Bicarbonate administration
Early administration of bicarbonate intravenously is thought to decrease metabolic acidosis and promote alkalisation of urine which decreases the precipitation of myoglobin in the renal tubules. Administration of bicarbonate immediately post-extrication, in anticipated metabolic acidosis, was discussed. Has this been shown to be beneficial? Are there any detrimental features? What would be the appropriate and safe doses to use? Is there a role for the combined use of acetazolamide in order to prevent metabolic alkalosis following bicarbonate administration?
Mannitol and compartment syndrome
There is anecdotal evidence in the literature that due to the high complication rate in performing
Page 7 of 8
minutes of administration of IV mannitol (5). Fasciotomy should be reserved for refractory cases. The use of an animal model of a compartment syndrome is questioned due to the anatomical differences from
humans. Many animals that are commonly used as models for humans, such as pigs, sheep and dogs do not have fascial compartments. Primates share similarities but, ethically, would be more difficult to justify. Further information on existing animal experimentation relating to compartment syndrome is required prior to planning any further projects.
This paper reports the findings of a consensus meeting on Crush Injury and Crush Syndrome held in Birmingham on 31 May 2001, and co-ordinated by the Faculty of Pre-Hospital Care of the Royal College of Surgeons of Edinburgh.
Organisations represented
The Voluntary Aid Societies
The Ambulance Service Association The British Association for Immediate Care British Association for Emergency Medicine Faculty of Accident and Emergency Medicine The Royal College of Anaesthetists
The Royal College of Physicians
The Royal College of Surgeons of Edinburgh The Intensive Care Society
The Royal College of Nursing The Military
The Faculty of Pre-hospital Care
*Lt Col I Greaves;RAMC
Visiting Professor of Emergency Medicine Education Centre, The James Cook University Hospital, Marton Road, Middlesbrough, TS4 3BW
K Porter
Consultant Trauma Surgeon, University Hospital, Birmingham
Surg Lt Cdr JE Smith;MBBS MSc MRCP RN
Page 8 of 8
References
1. Bywaters EGL, Beall D. Crush injuries with impairment of renal function. BMJ 1941; 1: 427. 2. Michaelson M. Crush injury and crush syndrome. World J Surg 1992; 16: 899-903.
3. Hiraide A, Ohnishi M,Tanaka H et al. Abdominal and lower extremity crush syndrome. Injury 1997;
28(9-10): 685-6.
4. Shaw AD, Sjolin SU, McQueen MM. Crush syndrome following unconsciousness: need for urgent orthopaedic referral. BMJ 1994; 309: 857-9.
5. Better OS. Rescue and salvage of casualties suffering from the crush sydrome after mass disasters. Mil Med 1999; 164: 366-9.
6. Rawlins M, Gullichsen E, Kuttila K, Peltola O, Niinikoski J. Central hemodynamic changes in experimental muscle crush injury in pigs. Eur Surg Res 1999; 31: 9-18.
7. Major Incident Medical Management & Support (MIMMS) 2002. BMJ Bookshops, London. 8. Greaves I, Porter KM, Revell MP. Fluid Resuscitation in Pre-Hospital Trauma Care: A Consensus
View. J.R. Coll. Surg. Edinb. 47; 2; 451-457.
9. Advanced Trauma Life Support for Doctors. American College of Surgeons 1997 Chicago.
10. Ward MM. Factors predictive of acute renal failure in rhabdomyolysis. Arch Intern Med 1988; 148: 1553-1557.
11. Better OS. The crush syndrome revisited (1940-1990). Nephron 1990; 55: 97-103.
12. Better OS, Zinman C, Reis ND et al. Hypertonic mannitol ameliorates intracompartmental tamponade in model compartment syndrome in the dog. Nephron 1991; 58: 344-6. 13. Advanced Paediatric Life Support 2nd Edition 2001. BMJ Bookshops.
14. Bouachour G, Cronier P, Gouello JP,Toulemonde JL,Talha A, Alquier P. Hyperbaric Oxygen therapy in the management of crush injuries; a randomized double-blind placebo-controlled clinical trial. J Trauma 1996; 41(2): 333-9.
Clinical
Reviews
CRUSH SYNDROME: A CASE REPORT AND REVIEW OF THE LITERATURE
Alissa Genthon,MD*†and Susan R. Wilcox,MD†§
*Department of Emergency Medicine, Brigham and Women’s Hospital, Boston, Massachusetts, †Department of Emergency Medicine, Massachusetts General Hospital, Boston, Massachusetts, and §Department of Anesthesia, Critical Care, and Pain Medicine, Massachusetts
General Hospital, Boston, Massachusetts
Reprint Address:Susan R. Wilcox,MD, Departments of Emergency Medicine and Anesthesia, Critical Care, and Pain Medicine, Massachusetts General Hospital, 55 Fruit Street, Boston, MA 02115
,Abstract—Background: Crush trauma to the
extrem-ities, even if not involving vital organs, can be life threat-ening. Crush syndrome, the systemic manifestation of the breakdown of muscle cells with release of contents into the circulation, leads to metabolic derangement and acute kid-ney injury. Although common in disaster scenarios, emer-gency physicians also see the syndrome in patients after motor-vehicle collisions and patients ‘‘found down’’ due to intoxication. Objective: The objectives of this review are to discuss the pathophysiology of crush syndrome, report on prehospital and emergency department treatment, and discuss the relationship between crush syndrome and compartment syndrome. Discussion: We present the case of a young man found down after an episode of intoxication, with compartment syndrome of his lower extremity and crush syndrome. Although he eventually required an ampu-tation, aggressive fluid resuscitation prevented further kidney injury and metabolic derangement. Conclusions: Early, aggressive resuscitation in the prehospital setting, before extrication if possible, is recommended to reduce the complications of crush syndrome. Providers must be aware of the risk of hyperkalemia shortly after extrication. Ongoing resuscitation with i.v. fluids is the mainstay of treat-ment. Compartment syndrome is a common complication, and prompt fasciotomies should be performed when compartment syndrome is present. Ó2014 Elsevier Inc.
,Keywords—rhabdomyolysis; crush syndrome; renal failure; resuscitation; hyperkalemia
INTRODUCTION
Crush trauma to the extremities, even if not involving vi-tal organs, can be life threatening. The termcrush injury
refers to the damage resulting directly from the crushing force. Conversely, crush syndrome, also known as trau-matic rhabdomyolysis, is the systemic manifestation of the breakdown of muscle cells with release of contents into the circulation (1,2). Crush syndrome leading to acute kidney injury (AKI) is one of the few life-threatening complications of crush injuries that can be prevented or reversed(3).
Crush syndrome was first described after the Battle of London by Bywaters and Beall in 1941. Patients pulled from the rubble initially appeared to be unharmed, but then these patients developed progressive limb swelling and shock and died of renal failure a few days later(2). Postmortem examination revealed muscle necrosis and brown pigment casts in the renal tubules (4). Crush in-juries are common in natural disasters such as earth-quakes, but emergency physicians more commonly see the syndrome in patients after motor-vehicle collisions, especially with prolonged extrications, as well as in vic-tims of assault (5,6). Crush syndrome also occurs in patients who compress a part of their own body, such as patients ‘‘found down’’ due to a stroke, intoxication, or mental illness(1). Any condition that results in prolonged
RECEIVED: 23 February 2013; FINAL SUBMISSION RECEIVED: 19 May 2013;
ACCEPTED: 15 August 2013
313
The Journal of Emergency Medicine, Vol. 46, No. 2, pp. 313–319, 2014 CopyrightÓ2014 Elsevier Inc. Printed in the USA. All rights reserved 0736-4679/$ - see front matter
immobility can result in a crush injury(4,7). In the United States, heroin is a common etiology and alcohol has been found to be the most common etiology of crush syndrome, compartment syndrome, and rhabdomyolysis in many industrialized countries (5,7–12). Patients might regain consciousness within several hours, but due to pain in limbs are unable to get up off the floor, leading to ongoing compression.
CASE REPORT
A 23-year-old male with a history of bipolar disorder and polysubstance abuse was brought into the emergency department (ED) by emergency medical services (EMS) after being found down at home. EMS reported that no one had seen the patient for nearly 24 h when his mother came home and found him lying on the floor in the kitchen. He was lethargic and confused with a Glasgow Coma Scale score of 13. He was lying on his left side, with his left lower extremity curled underneath his body. He was boarded, collared, and brought to the ED.
In the ED, he complained of pain in his left leg, but he was unable to provide any history. His vital signs were a temperature of 37.2C, heart rate of 150 beats/min, blood
pressure of 150/70 mm Hg, respiratory rate of 16 breaths/ min, and an O2saturation of 99% on room air. His
phys-ical examination was notable for ecchymosis around his left orbit and numerous areas of skin breakdown on his left chest and abdomen. His left lower extremity had a noncircumferential macerated disruption to the skin on the posterior-lateral aspect, where it had been in contact with the floor, with surrounded blistering that appeared similar to a burn. The leg was cold with mottling, and the compartments of the lower leg were all tight to palpa-tion. No pulses or capillary refill could be appreciated.
His laboratory results were notable for a white blood cell count of 26,000 cells/mL, hemoglobin of 19.4 g/dL,
hematocrit of 59.3%, and platelets of 183,000/mm3. His sodium was 132 mmol/L, potassium was 5.4 mmol/L, chloride was 105 mmol/L, bicarbonate was 14 mmol/L, blood urea nitrogen was 22 mmol/L, creatinine was 1.4
mmol/L, glucose was 128 mmol/L, and lactate was 2.8
mmol/L. The patient’s toxicology screens, including ethanol, were negative. The patient’s initial creatinine kinase (CK) was 41,669 IU/L. His head computed tomog-raphy (CT) and C-spine CT were negative for any acute injury or pathology.
The trauma team placed two large-bore i.v. lines and the patient was started on 2 L of 0.9% saline boluses. He was taken emergently to the operating room from the ED for fasciotomies of his left lower extremity.
After the procedure, the patient remained intubated and was admitted to the intensive care unit. After the fas-ciotomies, his left lower extremity pulses returned and the
leg was monitored closely by the surgery department. He continued to receive aggressive fluid hydration at 200–500 mL/h to maintain a urine output of at least 200 mL/h, and he required a norepinephrine infusion to main-tain a mean arterial pressure > 65 mm Hg. The following day, the patient’s CK peaked at 50,867 IU/L. His urine myoglobin was checked, with a level of 32.9 mg/mL
(reference range: < 0.025 mg/mL). With continued
aggressive i.v. fluids his creatinine trended down during the following 3 days to 0.64mmol/L. On hospital day 3,
while he continued to receive high-volume fluid resusci-tation, pulmonary edema developed. The i.v. fluids were reduced to 100 mL/h, mannitol and Lasix were added to maintain his urine output, and his ventilator was managed with low tidal volume ventilation for lung protection. The pulmonary edema resolved over the following day.
Despite the initial return of pulses with fasciotomy, the patient’s left lower extremity suffered extensive soft-tissue damage and ischemia. He underwent a below-the-knee amputation (BKA) on hospital day 4. Shortly after the BKA, his CK dropped markedly, the shock improved, and aggressive fluid resuscitation was stopped. He was extubated the following day. The patient was not able to recall how he came to be lying on the floor, but did recall drinking a significant amount of alcohol the night before. The patient was discharged to a rehabilitation hospital on post-injury day 20. He has since followed up in surgi-cal clinic and has been doing well in physisurgi-cal therapy, learning to ambulate with his prosthesis.
DISCUSSION
The mechanism of injury and cell death in crush syn-drome comes from the compression of the muscle fibers. In addition to the direct trauma of the compression, the tissue is deprived of blood flow and becomes ischemic, with both mechanisms causing lysis of muscle cells, lead-ing to significant metabolic imbalance and eventual organ failure(13). The times to cellular injury and death vary with the crushing force involved. Skeletal muscle can generally tolerate up to 2 h of ischemia without perma-nent injury. However, at 4–6 h, tissue necrosis develops
(13). At the cellular level, a crush insult opens stretch-activated channels in the muscle cell membrane and dis-rupts the Na/K transporter, allowing calcium to move freely into the cell. The increased intracellular calcium stimulates the activity of intracellular proteases, leading to eventual breakdown of the cell (5). Restoration of circulation to the damaged area results in ischemia-reperfusion injury. The post-ischemic tissues have high concentrations of neutrophil chemoattractants, leading to activation of neutrophils with release of proteolytic en-zymes and generation of free radical superoxide anions once perfusion is restored(5,13).