1
The Growth of
Chryseobacterium indologenes
ID 6016 on Filtrate’s
Culture of
Enterococcus faecalis
ID 6017 Which Continuously Growing
on Orange II Containing Medium
C. indologenes have been reported to grow in the medium which contained degrading orange II by E. faecalis. This phenomena generates a hypothesis that C. indologenes could grow and use intermediate products of orange II degradation as their growing medium. To verify this hypothesis, this research was done. Objective of this research was to determine the growth of C. indologenes ID 6016 on filtrate’s culture of E. faecalis ID 6017 which continuously growing on orange II containing medium. Research was done by inoculating C. indologenes on the rest medium from growth of E. faecalis on orange II containing medium. This medium was assumed to contain sulphanilic acid and 1-amino-2-naphtol. From the results, we concluded that C. indologenes was able to grow on filtrate’s culture of E. faecalis
ID 6017 which contained degraded product of orange II decolorization and to decrease or degrade the content of sulphanilic acid and 1-amino-2-naphtol. The biomass production was increased according to the decrease of sulphanilic acid, 1-amino-2-naphtol and COD. The specific growth rate (μ) varied from 0.0092 – 0.0129. Suphanilic acid, 1-amino-2-napthol, and COD consumption were 33 - 37, 24 – 28.9, and 68 – 100.27 mg.l-1, respectively.
Key words: decolorization of orange II, Sulphanilic acid, 1-amino-2-naphtol, the growth of C. indologenes
INTRODUCTION
Development of textile industry in Indonesia gives negative impacts for environmental
quality. One of these negative impacts is effluent of dyeing process, especially caused by
synthetic dye which relatively resistant to microbial degradation. During dyeing process,
approximately 10-15% of the dyes are released into the environment through effluent of
wastewater treatment (WWT) (Supaka et al. 2004). So, the existence of these dyes in water
ecosystem could inhibit light and create problems to photosynthetic aquatic plants and algae.
In addition to their visual effect and their adverse impact in terms of chemical oxygen
demand, many synthetic dyes are toxic, mutagenic and carcinogenic (Donlon et al., 1997).
Orange II is one of synthetic azo dyes which could be decolorized by Enterococcus
faecalis ID 6016 and produce intermediate products identified as sulphanilic acid and
amino-naphthol (Meitiniarti et al., 2007). Although they are colorless, they are toxic and mutagenic.
Chryseobacterium as one of flavobacteria could grow in the medium contained aromatic
compound as sole carbon source and consume it (Mitchell, 1978). Several researchers
2
reported that C. indologenes could grow in the medium contained suphanilic acid, even until
500 mg/l sulphanilic acid in the medium (Vifian, 2005; Febrilia, 2007). There is no information
about the growth of C. indologenes in medium contained decolorized azodye. Therefore,
objectives of this research are to study the growth of C. indologenes in several cell
concentrations in filtrate’s culture of E. faecalis which continuously growing on orange II
containing medium.
MATERIALS AND METHODS
Microorganism and Composition of Growth Medium
C. indologenes ID6016 was obtained from Laboratory of Microbiology, Faculty of
Biology, Satya Wacana Christian University, Salatiga, Indonesia. C. indologenes ID6016 was
maintained in basal medium which contained 0.250 g/l sulphanilic acid and agar 20 g/l.
Composition of 1 l basal medium was K2HPO4 7.095, KH2PO4 3.574, (NH4)2SO4 1.980,
MgSO4.7H2O 0.250 g, and 1 ml of trace element (composed of CuSO4.5H2O 0.234,
ZnSO4.7H2O 0.11, MnCl2.4H2O 0.35, FeSO4.7H2O 0.63 g/100ml). For growing medium, we
used fíltrate culture of E. faecalis which was continuously growing in orange II containing
medium. Outlet of continuously culture of E. faecalis was filtration using 0,2μm membrane
filter and put into 250 ml sterile flaks. The filtrate culture of E. faecalis was collected when the
growth of E. faecalis raised steady state condition and orange II was decolorized.
Culture condition
The 48 hours slant culture of C. indologenes was inoculated into 200 ml semisynthetic
medium as preculture and incubated in shaker incubator with 120rpm during 24 hours.
Concentration of inoculums tested were 10%, 20% and 40%. Mediums were inoculated
aseptically by precultures according to the treatments. Then, C. indologenes was grown by
batch system and agitated. Five ml of bacterial culture were taken by sterile syringe every 24
hours. It started at the injection of preculture into growth media, and stopped after the
bacterial growth reached stationary phase.
Analytical methods
Samples were centrifuged at 3326 g for 30 min to separate supernatant and cell
mass. The supernatant was used for determining sulfanilic acid, 1-amino-2-naphthol, and
COD concentrations. The concentration of sulfanilic acid and 1-amino-2-naphthol were
determined by HPLC (Haug et al. 1991; Chang et al. 2001). The concentration of COD was
determined by colorimetric method using a standard curve at 600 nm (Kruis, 1995). Cell
3
absorbance at 500 nm against cell mass. All measurement of absorbance was done in a
Shimadzu UV–Vis 1201 Spectrophotometer.
RESULTS AND DISCUSSION
The results showed that C. indologenes could grow on filtrate’s culture of E. faecalis.
During its growth, biomass concentration increased and sulfanilic acid and
1-amino-2-naphthol concentration decreased (Fig. 1). The increasing of biomass concentration was
followed by decreasing of COD concentration too. The decreasing of COD concentration
which indicated organic content of medium has a significant relationship with consumption of
organic matters. From growth curve of C. indologenes (Fig. 1), we could see that in the all
treatments of inoculum concentration, decreasing of sulfanilic acid and 1-amino-2-naphthol
content will followed by decreasing of COD concentration. These results answer assessment
of previously experiment (Vifian 2005; Febrilia, 2007) which presumed C. indologenes could
use intermediate products of orange II degradation as their growing substrate.
a) b)
c)
Fig. 1. The change of cell biomass of C. indologenes ID 6016 ( ), COD (X), sulphanilic acid
( ), and 1-amino-2-naphthol ( ) concentration on the medium with 10 (a), 20 (b), and 40 (c) % inoculum
Commonly, bacterial growth will refer lag, logaritmic, and stationary phase, but in the
growth of C. indologenes relatively didn’t show lag phase (Fig. 2). These phenomenon might
4
they didn’t need long time to grow. Lag phase of growth only occurred in 40% inoculum
culture caused there was take palace competition in the high cell density. In the high cell
density population will occurre competition for resources (Mitchell, 1978).
Fig. 2. The growth of C. indologenes ID 6016 in the medium with 10 ( ), 20 ( ), and 40 ( ) % inoculum
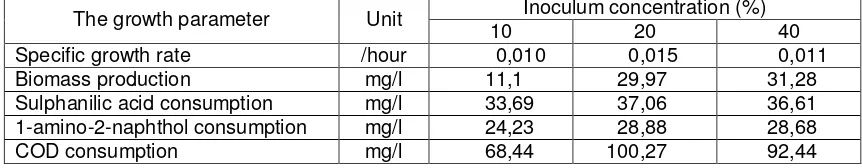
The results showed that C. indologenes showed the optimum growth in medium with
20% inoculum. In this culture, specific growth rate of C. indologenes was the highest, i.e
0.015/hour (Table 1). In this culture, sulphanilic acid and 1-amino-2-naphtol consumption
were the highest. So, this inoculum concentration could give best condition for growth of C.
indologenes and consumption of intermediet products of orange II degradation.
Table 1. The parameter of C. indologenes growth in culture with several inoculum concentrations
The growth parameter Unit Inoculum concentration (%)
10 20 40
Specific growth rate /hour 0,010 0,015 0,011 Biomass production mg/l 11,1 29,97 31,28 Sulphanilic acid consumption mg/l 33,69 37,06 36,61 1-amino-2-naphthol consumption mg/l 24,23 28,88 28,68 COD consumption mg/l 68,44 100,27 92,44
Existence of intermediate products consumption give evidence ability of C.
indologenes to use intermediate products of orange II decolorization for bacterial growth,
despite of these intermediate products were resistant to microbial degradation. There was
assessment that C. indologenes used sulphanilic acid and 1-amino-2-naphthol as their
carbon and nitrogen sources. It was proven to be decreasing of sulphanilic acid and
1-amino-2-naphthol concentration during cell growth phase. Several researchers also reported that
Sphingomonas sp. and Pseudomonaspaucimobilis could use sulphanilic acid as sole carbon
5
CONCLUSIONS
Based on experimental results, we concluded that C. indologenes could growth on
filtrate’s culture of E. faecalis which continuously growing on orange II containing medium.
The inoculum concentration which gave the best growth of C. indologenes was 20%. It was
caused by in this inoculum concentration bacterial growth faster and consumes intermediate
products higher than 10 and 40 % inoculum concentration.
REFERENCES
1. Supaka, N., Juntongjin, K., Damronglerd, S., Delia, M.L., Strehaiano, P. 2004. Microbial decolorization of reactive azo dyes in a sequential anaerobic-aerobic system. Chemistry Engineering Journal 99, 169-176
2. Donlon, B., Razo-Flores, E., Luijten, M., Swarts, H., Lettinga, G and Field, J., 1997. Detoxification and Pertial Mineralization of The azo Dye Mordan Orange I in a Continous up Flow Anaerobic Sludge-Blanket Reactor. Appl Microbiol Biotechnol, 47: 83-90.
3. Meitiniarti, VI, Sutarto, ES, Timotius, KH, Sugiharto, E. 2007. Product of Orange II biodegradation by Enterococcus faecalis ID6017 and Chryseobacterium indologenes
ID6016. Micobiol. Indones. 1 (2): 51-54
4. Mitchell,R. 1978. Water Pollution Microbiology. Vol II. John Willey and Sons. New York. 5. Vifian, N. 2005. Pertumbuhan Chryseobacterium indologenes ID 6016 pada Medium
yang Mengandung Asam Sulfanilat, Anilin dan 1Amino-2Naphtol. Tesis Magister Biologi. UKSW. Salatiga.
6. Febrilia, S. 2007. Pertumbuhan Chryseobacterium indologenes ID 6016 pada Medium yang Mengandung Asam Sulfanilat. Sripsi Fakultas Biologi. UKSW. Salatiga.
7. Haug, W., A. Schmidt, B. nortemann, D.C. Hempel, A. Stolz, and H.-J. Knackmuss. 1991. Mineralization of the sulfonated azo dye mordant yellow 3 by 6-aminonapthalene-2-sulfonate-degrading bacterial consortium. Appl. Environ. Microbiol. 57 (11): 3144-3149 8. Chang, J.-S., Chien, C., Lin, Y.-C., Lin, P.-J., Ho, J.-Y. 2001 Kinetic characteristics of
bacterial azo dye decolorization by Pseudomonas luteola. Water Research 35, 2841-2850
9. Kruis, F. 1995. Environmental chemistry selected analytical methods. Laboratory manual. IHE Deflt.
10. Coughlin, M. F., B. K. Kinkle and P. L. Bishop, 2003. Degradation of Acid Orange 7 in an Aerobic Biofilm. Chemorphere 37:2757-2763.