Brain Research 881 (2000) 208–211
www.elsevier.com / locate / bres
Short communication
Noncompetitive inhibition of the glycine receptor-mediated current by
melatonin in cultured neurons
a ,
*
a bFong-Sen Wu
, Ying-Chen Yang , Jing-Jane Tsai
a
Department of Physiology, College of Medicine, National Cheng Kung University, Tainan 70101, Taiwan
b
Department of Neurology, College of Medicine, National Cheng Kung University, Tainan 70101, Taiwan
Accepted 2 August 2000
Abstract
The effect of melatonin on the glycine receptor-mediated response was studied in cultured chick spinal cord neurons using the whole-cell voltage-clamp recording technique. Melatonin rapidly and reversibly inhibited the glycine-induced current in a dose-dependent fashion, with an EC50 of 934 mM and a maximal inhibition of 100%. Furthermore, melatonin noncompetitively inhibited the glycine response by an agonist-independent mechanism that was distinct from that of an open-channel blocker. 2000 Elsevier Science B.V. All rights reserved.
Theme: Neurotransmitters, modulators, transporters and receptors
Topic: Other neurotransmitters
Keywords: Melatonin; Glycine receptors; Cultured spinal neurons; Whole-cell recordings; Noncompetitive inhibition
The pineal hormone melatonin regulates many biologi- explore the mechanism of action of melatonin on the cal functions including circadian rhythm, sleep, mood, sex glycine receptor-mediated response in cultured chick spinal maturation, and immune responses [3]. In particular, cord neurons by using the whole-cell voltage-clamp re-melatonin has been shown to inhibit the excitability of the cording technique.
central nervous system [1]. Radioligand binding assays and Cultures of spinal cord neurons were prepared from behavioral studies suggest that melatonin may exert its 7-day chick embryos and were used 2–4 weeks after central effect by positively modulating the GABAA re- plating as described in detail [12]. Whole-cell currents ceptor in neuronal cells [2,6,9]. Indeed, this idea is were recorded by the whole-cell variant of the patch clamp confirmed by our recent electrophysiological studies, dem- technique [10]. Electrode resistance was 3–5 MV when onstrating that melatonin potentiates the GABA receptor-A filled with an intracellular solution containing (in mM): mediated current in cultured neurons [14]. Interestingly, 140 KCl, 3 NaCl, 1 MgCl , 4 MgATP, 11 EGTA, and 102 melatonin fails to enhance the glycine-induced current, HEPES (pH adjusted to 7.2 with KOH). In experiments in producing instead an inhibition of the glycine response. which high concentrations of glycine were used, the The mechanism underlying the effect of melatonin on the intracellular solution was replaced with a low chloride (20 GABAA receptor has been studied [14]. In contrast, mM) pipet solution. This solution contained the following relatively little is known of interaction of melatonin with (in mM): 130 potassium gluconate, 20 KCl, 3 sodium the glycine receptor. In the present study, we further gluconate, 4 MgATP, 11 EGTA, and 10 HEPES (pH adjusted to 7.2 with KOH). The bath solution contained (in mM): 150 NaCl, 4 KCl, 1 CaCl , 1 MgCl and 10 HEPES2 2 (pH adjusted to 7.2 with NaOH). All experiments were *Corresponding author. Tel.:1886-6-235-3535, ext. 5455; fax:1
886-performed at room temperature (23–258C). 6-236-2780.
E-mail address: [email protected] (F.-S. Wu). Recordings were made using an Axopatch-1D patch
F.-S. Wu et al. / Brain Research 881 (2000) 208 –211 209
clamp amplifier. Cells with series resistance greater than external buffer or melatonin solution, followed by a 10-s 10 MV were rejected. Only cells with resting membrane application of glycine or glycine plus melatonin, followed potential more negative than250 mV and input resistance by a 20-s pulse of external buffer solution. A period of 3–4 in excess of 150 MVwere used. All recordings were made min was allowed between successive applications of with the cell membrane potential clamped at 270 mV. glycine.
Currents were filtered at 1 kHz using an eight-pole Bessel At a holding potential of 270 mV, glycine produced filter and digitized (4 ms / point) using an on-line data inward currents, which reversed at or near 0 mV in
2 2
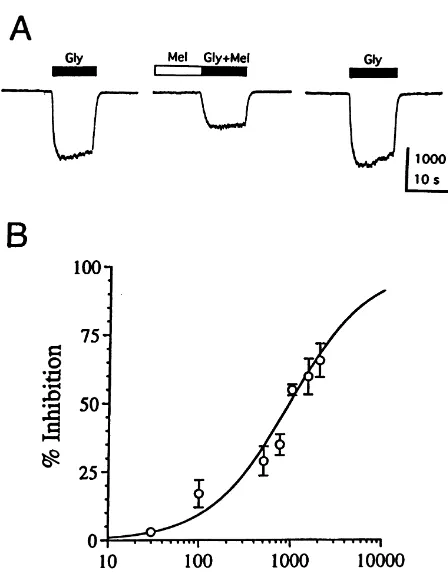
acquisition system (pClamp, Axon Instruments). symmetrical Cl solutions, as expected for Cl -mediated Drug solutions were applied to single neurons by currents (data not shown). In agreement with the previous pressure ejection (15 psi) from seven-barrel pipets [4,5]. work [13], the current induced by 50 mM glycine slowly All drugs were obtained from Sigma, with the exception of desensitized and did not decrease with repeated glycine melatonin (Acros). Stock solution of melatonin was pre- applications. The effect of melatonin on the current pared in dimethyl sulfoxide (final concentration, 0.5%, induced by 50mM glycine is illustrated in Fig. 1. Pressure v / v). To obviate the possible effect of dimethyl sulfoxide application of 1 mM melatonin reversibly inhibited the on the glycine-induced current, all other drug solutions, glycine response by 5562.0% (n54) (Fig. 1A). To including glycine and external buffer (in the pressure quantitatively evaluate the potency and efficacy of pipet), also contained 0.5% dimethyl sulfoxide. In all melatonin for glycine receptors, pooled data were used to experiments, neurons received a 10-s prepulse of either construct the dose–response curve for inhibition of the 50
mM glycine response by melatonin. As shown in Fig. 1B, melatonin produced a dose-dependent inhibition of the glycine-induced current, and curve-fit analysis revealed an EC50 of 934 mM and a maximal inhibition of 100%. Inhibition of the glycine response by melatonin was evident over roughly the same concentration range as enhancement of the GABA response, although melatonin was somewhat less potent at the glycine receptor than at the GABA receptor (ECA 505766mM) [14]. In both cases, Hill slopes were close to one, consistent with a single class of non-interacting sites.
Fig. 1. Melatonin inhibits the whole-cell current induced by glycine. (A) Melatonin (Mel) (1 mM) inhibits the current induced by 50mM glycine. Horizontal bar above each trace, period of drug application. (B) Dose– response curve for inhibition of the glycine (50 mM) response by melatonin. Data points, percentage change in peak current in the presence of melatonin (mean of 4–6 experiments). Error bars, standard errors. Error bars are not indicated when smaller than the size of the circle.
Fig. 2. Antagonism of the glycine response by melatonin is noncompeti-Melatonin dose–response curve is fitted with the logistic equation [8]. (%
nH nH nH tive. Inhibition of the glycine-induced current by 1 mM melatonin is not
inhibition) /(% inhibition)max5[Mel] /([Mel] 1EC50) where [Mel] is
significantly reduced by increasing glycine from 100mM to 300mM or to the concentration of melatonin, and nH is the Hill coefficient. Curve-fit
1 mM. Error bars, standard errors. Number of cells is indicated in analysis reveals an EC50of 934mM, a maximal inhibition of 100%, and
210 F.-S. Wu et al. / Brain Research 881 (2000) 208 –211
Fig. 3. Inhibition of the glycine response by melatonin is not agonist-dependent. Melatonin (1 mM) inhibits rapidly and reversibly the current induced by 50mM glycine in the absence of melatonin prepulse.
To investigate the mechanism of melatonin action on the latter three compounds is competitive [7,11,13], indicating glycine receptor, we examined the effect of 1 mM that melatonin and these drugs do not act through a melatonin on currents induced by higher concentrations of common site. Like melatonin, antagonism of the glycine
2
glycine. In this experiment, a low Cl (20 mM) pipet response by progesterone is noncompetitive [12]. Whether solution was used to reduce the amplitude of the glycine- melatonin acts through the progesterone modulatory site to induced current when the cell was held at 270 mV. As inhibit the glycine response needs further investigation. illustrated in Fig. 2, the percentage of inhibition by
melatonin of the glycine-induced current was not sig-nificantly attenuated by increasing glycine from 100 mM
Acknowledgements (5163.2%, n54) to 300mM (4666.1%, n54) or to 1 mM
(5466.9%, n55) (P.0.05, unpaired t-test). Because 1
Research support was provided by the National Health mM is a saturating concentration of glycine [13], this
Research Institutes (DOH88-HR-830) of Taiwan. suggests that antagonism of the glycine response by
melatonin is noncompetitive in nature and that melatonin inhibits the glycine response by acting through a site
References distinct from the glycine recognition site.
To determine whether block of the glycine response is
[1] D. Acuna-Castroviejo, G. Escames, M. Macias, A. Munoz Hoyos, agonist-dependent, we tested the effect of 1 mM melatonin
A. Molina Carballo, M. Arauzo, R. Montes, F. Vives, Cell protective on the current induced by 50mM glycine in the absence of
role of melatonin in the brain, J. Pineal Res. 19 (1995) 57–63. melatonin prepulse. Under this condition, the first
applica-[2] T.E. Albertson, S.L. Peterson, L.G. Stark, M.L. Lakin, W.D. Winters, tion of melatonin in the presence of glycine produced an The anticonvulsant properties of melatonin on kindled seizures in immediate blockade of both peak and plateau responses, rats, Neuropharmacology 20 (1981) 61–66.
[3] A. Brzezinski, Melatonin in humans, N. Engl. J. Med. 336 (1997) and recovery of the glycine response after washout of
186–195. melatonin was rapid (Fig. 3), suggesting that melatonin
[4] C.Y. Chan, D.H. Farb, Modulation of neurotransmitter action: inhibition of the glycine response is not agonist-dependent.
control of the g-aminobutyric acid response through the benzo-The present study demonstrates that inhibition of the diazepine receptor, J. Neurosci. 5 (1985) 2365–2373.
glycine response by melatonin is noncompetitive and [5] D.W. Choi, G.D. Fischbach, GABA conductance of chick spinal cord and dorsal root ganglion neurons in cell culture, J. Neurophysiol. 45 agonist-independent. There are a number of potential sites
(1981) 605–620.
at which melatonin could exert its blocking action includ- 3
[6] F.M. Coloma, L.P. Niles, Melatonin enhancement of [ H]-g -amino-ing: (1) competitive inhibition at the glycine binding site, 3
butyric acid and [ H]muscimol binding in rat brain, Biochem. (2) blockade of the glycine receptor channel, or (3) Pharmacol. 37 (1988) 1271–1274.
noncompetitive inhibition or allosteric modulation at a [7] D.R. Curtis, L. Hosli, G.A.R. Johnston, A pharmacological study of the depression of spinal neurones by glycine and related amino distinct site. Firstly, inhibition by melatonin of the glycine
acids, Exp. Brain Res. 6 (1968) 1–18. response is not reduced by increasing concentrations of
[8] A.P. De Lean, P.J. Munson, D. Rodbard, Simultaneous analysis of glycine (Fig. 2), suggesting that the blocking action of
families of sigmoidal curves: application to bioassay, radioligand melatonin is noncompetitive. Secondly, inhibition of the assay, and physiological dose–response curves, Am. J. Physiol. 235 glycine-induced current by melatonin is not agonist-depen- (1978) E97–E102.
dent (Fig. 3), arguing against the possibility that melatonin [9] D.A. Golombek, D. Fernandez Duque, M.G. De Brito Sanchez, L. Burin, D.P. Cardinali, Time-dependent anticonvulsant activity of acts as an open-channel blocker.
melatonin in hamsters, Eur. J. Pharmacol. 210 (1992) 253–258. In addition to melatonin, the glycine receptor is also
[10] O.P. Hamill, A. Marty, E. Neher, B. Sakmann, F.J. Sigworth, negatively modulated by a number of compounds includ- Improved patch-clamp techniques for high-resolution current record-ing progesterone, pregnenolone sulfate, strychnine, and ing from cells and cell-free membrane patches, Pflugers Arch. 391¨
21
F.-S. Wu et al. / Brain Research 881 (2000) 208 –211 211
[11] B. Laube, J. Kuhse, N. Rundstrom, J. Kirsch, Modulation by zinc [13] F.-S. Wu, S.-C. Chen, J.-J. Tsai, Competitive inhibition of the ions of native rat and recombinant human inhibitory glycine glycine-induced current by pregnenolone sulfate in cultured chick receptors, J. Physiol. 483 (1995) 613–619. spinal cord neurons, Brain Res. 750 (1997) 318–320.
[12] F.-S. Wu, T.T. Gibbs, D.H. Farb, Inverse modulation ofg-amino- [14] F.-S. Wu, Y.-C. Yang, J.-J. Tsai, Melatonin potentiates the GABAA