Hus s e in I. Abde l Shafy, We rne r He ge mann and Caro la Güldne r Fate o f he avy me tals via c he mic al-bio lo gic al upgrading o f se wage tre atme nt plant Enviro nme ntal Manage me nt and He alth
7 / 3 [1 9 9 6 ] 2 8 –3 6
• Aer a tion ta n k : volu m e: 2,280m3; loa din g: a t 0.15k g BOD5/ k g MLSSxd; SVl: a t 250/ k g; slu dge a ge: 10d.
• Two secon da r y sedim en ta tion ta n k s: volu m e: 2 ×1,280m3; r eten tion tim e: 3h .
• Slu dge tr ea tm en t: two open ta n k s: volu m e: 2 ×4,200m3; r eten tion tim e: 60d.
• Slu dge polder : r eten tion tim e: sever a l m on th s.
• Slu dge h ydr olysis: volu m e: 80; r eten tion tim e: 0.7-2d; pH a r ou n d 6.0-6.5.
Grit c hambe r
Lime -Fe Cl3
additio n COD/ BOD5 re mo val
P-re mo val
Influe nt Sc re e n
Sludge
Fo uling wate r
Primary s e dime ntatio n
Primary s ludge
Primary s ludge tank
Two s e ptic tanks Sludge po lde rs (drying be ds )
Sludge Partial nitrific atio n
Ae ratio n tank
To the anae ro bic re ac to r (hydro lys is )
Se c o ndary s e dime ntatio n
Efflue nt
(Havel) Unc o ntro lle d wild-nitrific atio n
Figure 1
Se wage wate r tre atme nt plant with quic k-lime additio n de vic e at Oranie nburg
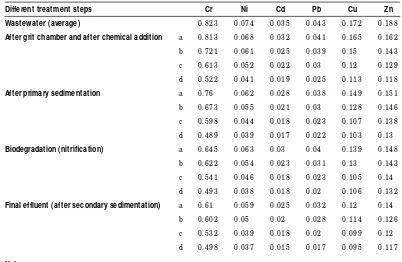
Table I
Le ve l o f he avy me tals in the was te wate r o f the s e wage via diffe re nt tre atme nt pro c e s s e s us ing c he mic al c o agulants (mg/ l)
Different treatment steps Cr Ni Cd Pb Cu Zn
Wastewater (average) 0 .8 2 3 0 .0 7 4 0 .0 3 5 0 .0 4 3 0 .1 7 2 0 .1 8 8
After grit chamber and after chemical addition a 0 .8 1 3 0 .0 6 8 0 .0 3 2 0 .0 4 1 0 .1 6 5 0 .1 6 2
b 0 .7 2 1 0 .0 6 1 0 .0 2 5 0 .0 3 9 0 .1 5 0 .1 4 3
c 0 .6 1 3 0 .0 5 2 0 .0 2 2 0 .0 3 0 .1 2 0 .1 2 9
d 0 .5 2 2 0 .0 4 1 0 .0 1 9 0 .0 2 5 0 .1 1 3 0 .1 1 8
After primary sedimentation a 0 .7 6 0 .0 6 2 0 .0 2 8 0 .0 3 8 0 .1 4 9 0 .1 5 1
b 0 .6 7 3 0 .0 5 5 0 .0 2 1 0 .0 3 0 .1 2 8 0 .1 4 6
c 0 .5 9 8 0 .0 4 4 0 .0 1 8 0 .0 2 3 0 .1 0 7 0 .1 3 8
d 0 .4 8 9 0 .0 3 9 0 .0 1 7 0 .0 2 2 0 .1 0 3 0 .1 3
Biodegradation (nitrification) a 0 .6 4 5 0 .0 6 3 0 .0 3 0 .0 4 0 .1 3 9 0 .1 4 8
b 0 .6 2 2 0 .0 5 4 0 .0 2 3 0 .0 3 1 0 .1 3 0 .1 4 3
c 0 .5 4 1 0 .0 4 6 0 .0 1 8 0 .0 2 3 0 .1 0 5 0 .1 4
d 0 .4 9 3 0 .0 3 8 0 .0 1 8 0 .0 2 0 .1 0 6 0 .1 3 2
Final effluent (after secondary sedimentation) a 0 .6 1 0 .0 5 9 0 .0 2 5 0 .0 3 2 0 .1 2 0 .1 4
b 0 .6 0 2 0 .0 5 0 .0 2 0 .0 2 8 0 .1 1 4 0 .1 2 6
c 0 .5 3 2 0 .0 3 9 0 .0 1 8 0 .0 2 0 .0 9 9 0 .1 2
d 0 .4 9 8 0 .0 3 7 0 .0 1 5 0 .0 1 7 0 .0 9 5 0 .1 1 7
Notes:
Results presented here are the average of 1 2 weekly suc c essive samples a: No c hemic als were added (c ontrol)
b: Addition of FeCl3only (8 .4 g/ m3)
Hus s e in I. Abde l Shafy, We rne r He ge mann and Caro la Güldne r Fate o f he avy me tals via c he mic al-bio lo gic al upgrading o f se wage tre atme nt plant Enviro nme ntal Manage me nt and He alth
7 / 3 [1 9 9 6 ] 2 8 –3 6
• Dose of lim e = 120 g/ m3(20 per cen t); dose of FeCl3= 8.4g Fe+3/ m3[3.4gla s FeCl
3/ m3] a t 14/ d.
Week ly sa m ples of th e w a stew a ter a n d/ or slu dge wer e collected for a per iod of 12 su cces-sive week s. Th e w a stew a ter sa m ples wer e fr esh ly filter ed th r ou gh Wh a tm a n No. 4 equ iva len t filter pa per, a cidified to below pH 2.0 u sin g a n a lytica l r ea gen t (AR) of n itr ic a cid. Th e slu dge sa m ples wer e oven dr ied a t 105°C for 24 h ou r s. A k n ow n weigh t of ea ch slu dge sa m ple w a s a cid digested u sin g AR n itr ic a cid followed by AR h ydr ogen per oxide, a ccor din g to US E nvir on m en ta l P r otection Agen cy[11].
Meta l con cen tr a tion s in th ese w a stew a ter a n d slu dge sa m ples wer e deter m in ed u sin g Va r ia n a tom ic a bsor ption spectr om eter, m odel Spectr AA-400 equ ipped w ith GA 1-96 gr a ph ite tu be a tom izer, a tta ch ed by IBM
per son a l com pu ter AT (pr ogr a m m ed for Va r ia n -Spectr AA-300/ 400). Th e stu died m eta ls a r e Cr, Ni, Cd, P b, Cu a n d Zn .
E a ch r esu lt of ea ch sa m ple is th e aver a ge of ten sequ en tia l r ea din gs. As a n in str u m en t a n d pr ocedu r e: bla n k of dou ble distilled w a ter th a t digested u sin g th e pr ocedu r e pr e-viou sly descr ibed w a s em ployed. Th e a ll-over r esu lts pr esen ted h er e a r e th e aver a ge of a ll th e stu died sa m ples.
Results and discussion
Th e level of h eavy m eta ls in th e r aw w a ste-w a ter of th e seste-w a ge befor e tr ea tm en t a n d a fter gr it-ch a m ber (ch em ica l a ddition ), pr i-m a r y sedii-m en ta tion , a er a tion a s n itr ifica tion a n d fin a lly a fter th e secon da r y sedim en ta tion su ccessively a r e sh ow n in Ta ble I. Th e ch em i-ca ls wer e a dded a s 8.4g/ m3FeCl
3or 120g/ m3
5 0
4 0
3 0
2 0
1 0
0
To tal re mo val (pe r c e nt)
(a) Afte r grit-c hambe r (afte r c he mic al additio n)
Cr Ni Cd Pb Cu Z n
6 0
5 0
4 0
3 0
2 0
1 0
0
To tal re mo val (pe r c e nt)
(b) After primary sedimentation
Cr Ni Cd Pb Cu Z n
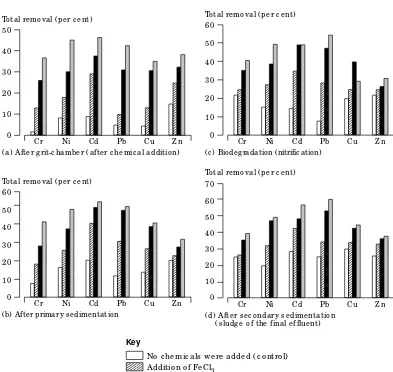
Figure 2
The pe rc e ntage o f me tals e liminatio n in the was te wate r s e wage via diffe re nt tre atme nt pro c e s s e s us ing c he mic al c o agulants
7 0
6 0
5 0
4 0
3 0
2 0
1 0
0
To tal re mo val (pe r c e nt)
(d) Afte r s e c o ndary s e dime ntatio n (s ludge o f the final e fflue nt)
Cr Ni Cd Pb Cu Z n
6 0
5 0
4 0
3 0
2 0
1 0
0
To tal re mo val (pe r c e nt)
(c ) Biodegradation (nitrific ation)
Cr Ni Cd Pb Cu Z n
Additio n o f lime o nly (1 2 0 g/ m3) Key
No c he mic als we re adde d (c o ntro l) Additio n o f Fe Cl3
Hus s e in I. Abde l Shafy, We rne r He ge mann and Caro la Güldne r Fate o f he avy me tals via c he mic al-bio lo gic al upgrading o f se wage tre atme nt plant Enviro nme ntal Manage me nt and He alth
7 / 3 [1 9 9 6 ] 2 8 –3 6
lim e a n d/ or com bin a tion of both FeCl3a n d lim e togeth er. Resu lts obta in ed sh ow th a t a n oticea ble decr ea se in th e level of m eta ls w a s r ecor ded wh en th e ch em ica l coa gu la n ts wer e
a dded to th e w a stew a ter in cor r ela tion w ith th e con tr olled on e. Mea n wh ile, th e u se of th e com bin ed coa gu la n ts, n a m ely FeCl3a n d lim e, w a s m or e effective in r edu cin g th e level of
Table III
Le ve l o f he avy me tals in the pre tre atme nt s ludge via the s uc c e s s ive tre atme nt pro c e s s (mg/ kg dry we ight)
M etals (mg/ kg dry weight)
Successive treatment processes Cr Ni Cd Pb Cu Zn
Sludge of the raw sewage Average 3 0 5 3 3 7 .2 1 4 0 1 3 1 8 3 7
(before chemical addition) Maximum 3 1 9 4 6 1 0 .3 3 6 1 1 7 6 8 6 6
Minimum 3 0 1 2 2 1 .0 4 1 8 6 4 7 9 1
After grit chamber a 1 9 5 2 2 2 .7 3 3 0 1 1 1 7 1 9
(after chemical addition) b 2 1 5 3 2 3 .1 1 4 2 1 2 3 7 9 0
c 2 4 0 4 1 4 .8 2 5 5 1 3 1 8 0 3
d 2 8 2 5 8 5 .1 3 6 8 1 3 3 8 2 7
After primary sedimentation a 9 2 3 1 1 .3 2 3 8 1 0 8 8 6 6
b 4 8 2 9 1 .1 4 3 7 9 7 7 5 4
c 3 4 2 5 1 .0 7 3 4 8 2 6 8 3
d 3 1 2 0 0 .5 1 3 3 8 0 6 6 2
Aeration as biodegradation (nitrification) a 3 0 2 3 2 7 .0 1 3 8 1 2 5 8 7 1
b 2 1 4 3 1 2 .2 4 3 7 1 1 3 8 5 2
c 1 5 3 2 4 1 .3 3 3 1 1 0 4 7 3 0
d 1 0 2 2 0 0 .7 1 2 9 1 0 0 6 9 4
After secondary sedimentation a 1 3 7 3 0 1 .9 6 4 2 1 0 8 8 5 4
(sludge of the final effluent) b 1 1 8 2 7 1 .0 5 3 3 9 2 8 4 3
c 9 1 2 3 0 .7 4 2 9 8 3 6 6 9
d 8 8 2 1 0 .5 8 2 8 7 7 5 9 4
Guideline 9 0 0 2 0 0 1 0.0 0 9 0 0 8 0 0 2 ,5 0 0
Notes:
Results presented here are the average of 1 2 weekly suc c essive samples a: No c hemic als were added (c ontrol)
b: Addition of FeCl3 only (8 .4 g/ m3)
c : Addition of lime only (1 2 0 g/ m3) d: Addition of FeCl3and lime Table II
Ave rage tre atme nt pe rfo rmanc e be fo re and during the lime phas e to the s e wage wate r at Oranie nburg
Jan Feb M ar Apr M ay Jun Jul
Unit Without lime addition Lime addition
BOD5 – precipitation after sedimentation % 2 3 2 8 1 1 1 4 2 1 4 8 4 9
Total BOD5-degradation % 9 4 9 3 9 6 9 3 9 4 9 8 9 4
COD-precipitation after sedimentationa % 2 6 3 0 2 6 2 9 3 2 4 9 5 0
Total COD-degradationa % 8 7 8 4 8 5 8 4 8 5 8 5 8 5
NH4-N in effluent (mg/ I) – 3 4 – 3 2 3 6 1 7 1 7
Inorganic N in effluent (mg/ I) – 3 6 – 3 3 3 7 2 7 2 9
Total P in effluent (mg/ I) – 7 .1 – 3 .4 3 1 .7 1 .6
Note:
aCOD-measurement with potassium permanganate
Hus s e in I. Abde l Shafy, We rne r He ge mann and Caro la Güldne r Fate o f he avy me tals via c he mic al-bio lo gic al upgrading o f se wage tre atme nt plant Enviro nme ntal Manage me nt and He alth
7 / 3 [1 9 9 6 ] 2 8 –3 6
m eta ls followed by th e u se of lim e th a n FeCl3. F u r th er m or e, th e pr im a r y a n d secon da r y sedim en ta tion wer e a lso sligh tly effective in m eta ls elim in a tion . Th e per cen ta ges of m eta ls elim in a tion in th e w a stew a ter via th ese su ccessive tr ea tm en t pr ocesses u sin g ch em ica l coa gu la n ts a r e pr esen ted in F igu r e 2. Su ch decr ea se in th e level of m eta ls is a ttr ibu ted m a in ly to th e in cr ea se of pH-va lu e to 9.0-9.2 wh ich in du ces th e decr ea se of m eta ls solu bility[6,7,10,12]. As th e pH in cr ea se, th e co-pr ecipita tion of m eta l h ydr oxide
in cr ea ses[6], bu t cer ta in a m ph oter ic m eta ls w ill r e-dissolve a t h igh er pH-va lu es[6,7]. Th e pr eviou s stu dy on th e sa m e pla n t pr oved th a t
lim e pr ecipita tion a t pH between 9.0-9.2 h a s a lso im pr oved th e pr e-tr ea tm en t per for m a n ce by @20 per cen t com pa r ed to th e zer o-ph a se (Ta ble II)[5].
F u r th er stu dy w a s ca r r ied ou t on th e level of m eta ls in th e sew a ge slu dge via th e sa m e su ccessive tr ea tm en t pr ocesses u sin g lim e a n d/ or FeCl3(Ta ble III). Th e given r esu lts sh ow th a t th e ch em ica l coa gu la n t in cr ea sed th e level of m eta ls in th e slu dge a s a r esu lt of th e co-pr ecipita tion effect, i.e. conver tin g th e solu ble for m s of m eta l to th e in solu ble for m in th e slu dge. Aga in th e u se of th e com bin ed coa gu la n t in cr ea sed th e co-pr ecipita tion r a te. However, th e u se of lim e w a s m or e effective
Table IV
Ge rman guide line re gulatio n fo r the le ve l o f he avy me tals in the s ludge (mg/ kg dry we ight)
M etal Ag Cr Ni Cd Pb Cu Zn
Guideline level 8 9 0 0 2 0 0 1 0 9 0 0 8 0 0 2 ,5 0 0
Source:[1 3 ]
(d) Afte r s e c o ndary s e dime ntatio n (s ludge o f the final e fflue nt) (a) Afte r grit-c hambe r (afte r c he mic al additio n)
7 0 6 0 5 0 4 0 3 0 2 0 1 0 0
Me tals e liminatio n (pe r c e nt)
Cr Ni Cd Pb Cu Z n
1 0 0
8 0
6 0
4 0
2 0
0
Me tals e liminatio n (pe r c e nt)
(c ) Biodegradation (nitrific ation)
Cr Ni Cd Pb Cu Z n
1 0 0
8 0
6 0
4 0
2 0
0
Me tals e liminatio n (pe r c e nt)
Cr Ni Cd Pb Cu Z n
1 0 0
8 0
6 0
4 0
2 0
0
Me tals e liminatio n (pe r c e nt)
(b) After primary sedimentation
Cr Ni Cd Pb Cu Z n
Key
No c he mic als we re adde d (c o ntro l) Additio n o f lime o nly (1 2 0 g/ m3)
Additio n o f Fe Cl3
Additio n o f lime and Fe Cl3
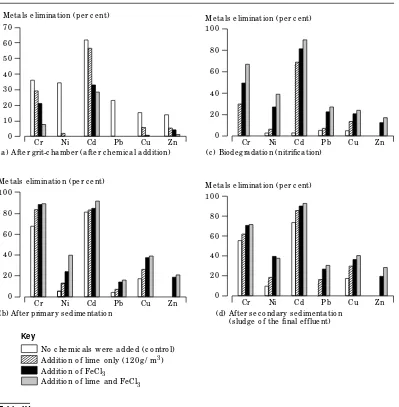
Figure 3
Hus s e in I. Abde l Shafy, We rne r He ge mann and Caro la Güldne r Fate o f he avy me tals via c he mic al-bio lo gic al upgrading o f se wage tre atme nt plant Enviro nme ntal Manage me nt and He alth
7 / 3 [1 9 9 6 ] 2 8 –3 6
th a n FeCl3. Th e per cen ta ge of m eta ls elim in a -tion in th e slu dge in cor r ela -tion to th e or igi-n a l coigi-n ceigi-n tr a tioigi-n iigi-n th e r aw w a stew a ter is illu str a ted in F igu r e 3. Th e pr im a r y a n d sec-on da r y sedim en ta tisec-on ta n k s w ith th e th r ee
h ou r s’ r eten tion tim e sh owed h igh efficien cy in tr a ppin g th e pr ecipita ted m eta ls. Th e level of m eta ls in th e pr im a r y slu dge wer e fa r below th e Ger m a n per m issible level (Ta ble IV)[13]. In a ddition , th e for m er stu dies
7 0 0
6 0 0
5 0 0
4 0 0
3 0 0
2 0 0
1 0 0
0
Capillary s uc tio n time (s e c o nds )
Solids c ontent (per c ent)
1 2 3 4 5 6 7 8
Fas te r filte ring
Source: [5 ]
Key
Be fo re c he mic al additio n Lime additio n
Lime and Fe Cl3 additio n Figure 4
Co rre latio n be twe e n the c apillary s uc tio n time and the s o lids c o nte nt
Table V
Le ve l o f he avy me tals in the s ludge be fo re and afte r the anae ro bic re ac to r pro c e s s (mg/ kg dry we ight)(pH 6 .0 -6 .5 )
M etals (mg/ kg dry weight)
Type of treatment Cr Ni Cd Pb Cu Zn
After primary sedimentation a 9 6 3 1 1 .3 2 3 8 1 0 8 8 6 6
(influent to the anaerobic reactor) b 4 8 2 9 1 .1 4 3 7 9 7 7 6 0
c 3 4 2 5 1 .0 7 3 4 8 2 6 8 3
d 3 1 2 0 0 .5 1 3 3 8 0 6 6 2
Average 4 6 2 5 0 .9 5 3 1 9 2 7 5 7
After secondary sedimentation a 1 3 7 3 0 1 .9 6 4 2 1 0 8 8 5 4
(influent to the anaerobic reactor) b 1 1 8 2 7 1 .0 5 3 3 9 2 9 4 3
c 1 0 2 3 0 .7 4 2 9 8 3 6 6 9
d 8 8 2 1 0 .5 8 2 8 7 7 5 9 4
Average 1 0 3 2 8 1 .0 7 3 0 9 1 8 4 5
Final effluent of the anaerobic reactor Average 9 9 3 9 1 .9 2 4 9 1 5 7 8 7 6
Maximum 1 1 7 3 6 2 .1 1 5 4 1 8 7 9 5 1
Minimum 8 1 2 4 0 .3 8 2 2 7 0 4 7 2
Guideline 9 0 0 2 0 0 1 0.0 0 9 0 0 8 0 0 2 ,5 0 0
Notes:
Results presented here are the average of 1 2 weekly suc c essive samples Average = average metal c onc entration of the four different sewage sludges a: No c hemic als were added (c ontrol)
b: Addition of FeCl3only (8 ,4 g/ m3)
Hus s e in I. Abde l Shafy, We rne r He ge mann and Caro la Güldne r Fate o f he avy me tals via c he mic al-bio lo gic al upgrading o f se wage tre atme nt plant still below th e per m issible level (Ta ble IV)[13]. Cor r ela tion between th e level of
Afte r primary s e dime ntatio n (influe nt to re ac to r) influe nts (name ly afte r primary s e dime ntatio n and afte r the s e c o ndary s e dime ntatio n) to the
Effluent of the anaerobic reac tor
Figure 6
Hus s e in I. Abde l Shafy, We rne r He ge mann and Caro la Güldne r Fate o f he avy me tals via c he mic al-bio lo gic al upgrading o f se wage tre atme nt plant Enviro nme ntal Manage me nt and He alth
7 / 3 [1 9 9 6 ] 2 8 –3 6
th e over loa ded pla n t[5]. Th e decr ea se of m eta ls con cen tr a tion in th e w a stew a ter w a s du e to th e in cr ea se of pH-va lu es a s a r esu lt of lim e a ddition . As th e pHin cr ea se, th e for m a -tion of m eta l h ydr oxide in cr ea ses[6,7,12]. Th is r ela tion sh ip is expr essed by th e follow in g sta bility pr odu ct equ a tion :
(M+2)(OM)2= K sp(solu bility con sta n t) Th e solu bility pr odu ct con sta n ts for a n u m -ber of m eta ls a r e well docu m en ted a n d a lr ea dy pu blish ed[13]. However, beca u se of pr ecipita tion a gein g, in com plete solid sepa r a -tion or th e co-pr ecipita -tion a n d a dsor p-tion effects in w a stew a ter solu bility pr odu cts pr ovide on ly a gen er a l gu ide to r esidu a l m eta l con cen tr a tion to be expected in pr a ctice[10].
Th e level of m eta ls in th e pr e-tr ea tm en t slu dge is fa r below th e Ger m a n r estr iction lim its[13]. It h a s been r epor ted th a t th is pr e-tr ea tm en t slu dge is im pr oved con sider a bly in ter m s of th ick en in g a n d dr a in in g qu a lities[5]. Despite th e in cr ea se in th e solid con ten t, on ly sligh t in cr ea se in th e slu dge volu m e w a s r epor ted[5].
Th e h ydr olysis a n d a cidifica tion of th e pr im a r y slu dge exh ibited con sider a ble in cr ea se in th e level of m eta ls du e to th e decr ea se of pH-va lu e fr om 9.0-9.2 to 6.0-6.5. However, th e vola tile or ga n ic a cids (C2-C6) for m ed du r in g th e pr ocess ca n be u sed a s a sou r ce of ca r bon for th e den itr ifica tion [14-16].
Th e level of Ni in th e tr ea ted slu dge of th e dr yin g bed r ea ch ed 317m g/ k g. Th is h igh level of Ni is du e to th e disch a r ge of cer ta in in du s-tr ia l w a stew a ter to th e sewer system .
Tr ea tm en t of th is in du str ia l w a stew a ter sh ou ld be ca r r ied ou t befor e disch a r gin g to th e sewer system . Accor din g to th e Ger m a n r egu la tion Ni sh ou ld n ot exceed 200m g/ k g a s dr y weigh t. Th er efor e, su ch slu dge ca n n ot be u sed a s a m a n u r e in a gr icu ltu r e[17]. On th e oth er h a n d, su ch u pgr a din g of th e sew a ge pla n t im pr oved th e per for m a n ce of tr ea tm en t a n d th is r edu ces sew a ge ta x. Mea n wh ile, th e h ydr olysis of th e pr e-tr ea tm en t slu dge ca n im pr ove th e per for m a n ce of th e den itr ifica -tion pr ocess a n d th er efor e it ca n save en er gy. F u r th er m or e, th e lim e a pplica tion for th e u pgr a din g of th e sew a ge pla n t ca n be a su bsti-tu tion for en la r gin g th e pla n t itself.
It is wor th n oticin g h er e th a t th e level of Zn in th e dr yin g bed slu dges r a n ges fr om 2,175 to 2,985 w ith th e aver a ge of 2,451m g/ k g dr y weigh t (Ta ble VI). Th e gu idelin e[5] is 2,500m g/ k g.
Th e pr esen t gu idelin e lim its for th e level of m eta l in slu dge is su fficien t for th e envi-r on m en ta l penvi-r otection . Th e settin g of lim its a t levels lower th a n n ecessa r y ca n be cou n ter -pr odu ctive a n d lea d to illega l discon tin u ou s disch a r ges a n d la ck of co-oper a tion between th e en for cem en t body a n d th e fir m s. Th is ca n lea d to excessive costs in con tr ol a n d m or e fr equ en t fa ilu r e to m eet th e r equ ir ed sta n da r d.
References
1 Ru dolph , D., “Rech tsgr u n dla gen der Abw a sser ein leitu n g in den n eu en
Table VI
Charac te ris tic s and the le ve l o f me tals in the primary and the tre ate d s ludge (s e ptic tanks and s ludge po lde r)
M etals (mg/ kg dry weight) Dry %
Type of sludge Cr Ni Cd Pb Cu Zn residue % ovm
Before the sludge treatment Average 1 0 4 2 5 0 .9 3 3 3 .6 1 1 1 7 3 0 6 .3 9 3 8 .4 6
(primary sludge) Maximum 1 5 1 3 7 2 .0 1 4 3 .5 1 7 8 9 9 7
Minimum 7 8 1 9 0 .3 2 5 .4 6 9 4 9 6
Septic tank (No. 1) Average 2 1 7 1 8 7 2 .2 1 1 1 8 .2 2 1 4 1 ,9 9 3 4 .1 8 5 7 .8 3
Maximum 2 9 5 2 1 9 7 .9 2 2 1 8 .4 3 5 1 2 ,5 0 1
Minimum 1 8 7 7 6 0 .7 9 9 7 .3 1 7 9 7 0 5
Septic tank (No. 2) Average 1 9 6 3 0 8 2 .5 4 1 2 5 .8 2 0 4 2 ,0 1 3 3 .4 4 5 9 .8 6
Maximum 2 5 7 3 9 5 5 .9 3 2 1 6 .8 3 1 2 2 ,7 0 4
Maximum 1 3 4 1 1 2 0 .5 1 9 0 .8 1 6 3 8 1 2
Sludge polder (drying beds) Average 3 2 2 3 1 7 3 .1 9 1 2 6 .8 2 3 9 2 ,4 5 1 9 .2 2 6 1 .0 8
Maximum 5 1 7 3 8 6 6 .9 8 2 4 7 .3 3 5 2 2 ,9 8 5 1 2 .5 6 6 2 .8 7
Maximum 2 3 9 1 7 5 0 .7 9 9 7 .8 1 9 3 2 ,1 7 5 5 .9 8 5 9 .6 5
Guideline 9 0 0 2 0 0 1 0.1 0 9 0 0 .0 8 0 0 2 ,5 0 0
Note:
Hus s e in I. Abde l Shafy, We rne r He ge mann and Caro la Güldne r Fate o f he avy me tals via c he mic al-bio lo gic al upgrading o f se wage tre atme nt plant Enviro nme ntal Manage me nt and He alth
7 / 3 [1 9 9 6 ] 2 8 –3 6
Bu n deslä n der n ”, gw f Wa sser / A b w a sser, Vol. 132 No. 4, 1991, pp. 193-6.
2 Hen ze, M. a n d Ha r r em oes, P., “Ch em ica l-biologica l n u tr ien t r em ova l – Th e HYP RO con cept”, in Ha h n , H.H. a n d Klu te, R. (E ds), Ch em ica l Wa ter a n d Wa stew a ter T rea tm en t, Spr in ger -Ver la g, Heidelber g, Ber lin , 1990, pp. 499-509.
3 Pesch en , N. a n d Sch u ster, G., “Steiger u n g der Rein igu n gsleistu n g u n d Sta bilisier u n g des Klä r pr ozesses bei m ech a n isch -biologisch en Klä r a n la gen du r ch Ka lk fä llu n g”, Kor resp on -d en z A b w a sser, Vol. 30, 1983, pp. 18-22. 4 Bisch ofsber ger, W., Ru f, M., Over a th , H. a n d
Hegem a n n , W., “An wen du n g von Fä llu n gsver -fa h r en zu r Ver besser u n g der Leistu n gs-fä h igk eit biologisch er An la gen a u s
Wa sser gü tew ir tsch a ft u n d Gesu n dh eitsin ge-n ieu r wesege-n ”, T U M ü n ch en, No. 13, 1976. 5 Gü ldn er, C., Hegem a n n , W., Pesch en , N. a n d
Iter, K.S., “Use of lim e for th e u pgr a din g of existin g w a stew a ter tr ea tm en t system ”, 2n d in ter n a tion a l specia lized con fer en ce: u pgr a d-in g of w a stew a ter tr ea tm en t pla n ts, Ber ld-in , 21-24 Septem ber 1993.
6 Abdel-Sh a fy, H.I., “P r ecipita tion of Ni, Cu a n d Mn fr om in du str ia l w a stew a ter ”, B u lletin N a tion a l R esea rch Cen tre, Vol. 17 No. 3, 1992, pp. 153-60.
7 Abu -E l-Wa fa , O., Abdel-Sh a fy, H.I. a n d E l-Ga m a l, I.M., “Stu dies on h eavy m eta ls r em ova l via ch em ica l tr ea tm en t”, T h e 1st n a tion a l con feren ce – In stitu te of E n v ir on m en -ta l S tu d ies a n d R esea rch, Ca ir o, 31 J a n u a r y-4 Febr u a r y 1988, pp. 757-65.
8 Leon h a r d, K. a n d Hegem a n n , W., “Th e effect of copper a n d zin c in sew a ge slu dge”, B er ich te a u s Wa ssergü tew ir tsch a ft u n d Gesü n d h eitsin ge-n ieu r w esege-n, T U M ü n ch en, No. 62, 1985, pp. 1-55. 9 E l Ga m a l, I.M. a n d Abdel-Sh a fy, H.I., “Role of
la n d ir r iga tion by liqu id sew a ge on th e u pta k e
of m eta ls a n d n u tr ien t elem en ts by pla n ts”, B iom a ss a n d B ioen erg y – A n In ter n a tion a l J ou r n a l, Vol. 1 No. 5, 1991, pp. 275-80. 10 E l-Goh a r y, F.A., La sh een , M.R. a n d
Abdel-Sh a fy, H.I., “Tr a ce m eta ls r em ova l fr om w a ste-w a ter via ch em ica l tr ea tm en t”, Heavy Meta ls in th e E nvir on m en t – Th e In ter n a tion a l Con -fer en ce, Lon don , Septem ber 1979.
11 US E nvir on m en ta l P r otection Agen cy, M eth od s for Ch em ica l A n a lysis of Wa ter a n d
Wa stew a ter, Office of Tech n ology Tr a n sfer, Wa sh in gton , DC, 1974.
12 Feitk n ech t, W. a n d Sch in dler, P., “Solu bility con sta n ts of m eta l oxides, m eta l h ydr oxides a n d m eta l h ydr oxide sa lts in a qu eou s solu -tion s”, Pu re A p p lied Ch em istr y, Vol. 6, 1963, p. 130.
13 Klä r sch la m m ver or dn u n g (AbfKlä V), B u n d es-gesetz bla tt, J a h rga n g 1992, Teil I, No. 15, Apr il 1992, p. 3.
14 Hegem a n n , W., “Übu n gsk u r s Siedlu n gsw a sser -w ir tsch a ft a n der Tech n isch en Un iver sitä t Ber lin ”, Ku r steil P h osph a telim in a tion , 27 Febr u a r y 1991, u n pu blish ed.
15 Hoffm a n n , E . a n d Klu te, R., “Im pr ovin g th e den itr ifica tion poten tia l in biologica l w a ste-w a ter tr ea tm en t by dosin g ca r bon fr om slu dge h ydr olysis”, in Ha h n , H.H. a n d Klu te, R. (E ds), Ch em ica l Wa ter a n d Wa stew a ter T rea tm en t, Spr in ger -Ver la g, Ber lin -Heidelber g, 1990. 16 Ka r lsson , I., Gör a n sson , J. a n d Rin del, K.,
“Use of in ter n a l ca r bon fr om slu dge h ydr olysis in biologica l w a stew a ter tr ea tm en t”, in Ha h n , H.H. a n d Klu te, R. (E ds), Ch em ica l Wa ter a n d Wa stew a ter T rea tm en t, Spr in ger -Ver la g, Ber lin -Heidelber g, 1990.